
Synopsis
Synopsis
0
JDMF
0
VMF
0
Canada
0
Australia
DRUG PRODUCT COMPOSITIONS
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
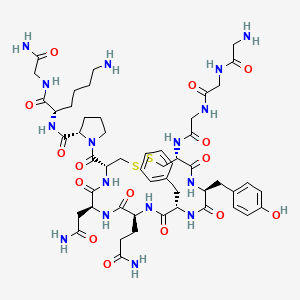

1. Glipressin
2. Gly Gly Gly 8 Lys Vasopressin
3. Gly-gly-gly-8-lys-vasopressin
4. Glycylpressin
5. Glypressin
6. N-(alpha)-glycyl-glycyl-glycyl-8-lysine Vasopressin
7. Remestyp
8. Terlypressin
9. Tglvp
10. Tri-gly-8-lys- Vasopressin
11. Triglycyl Lysine Vasopressin
12. Triglycyl-(8-lysine)vasopressin
13. Triglycylvasopressin
14. Vasopressin, Tri-gly-8-lys-
1. 14636-12-5
2. Terlipressin Acetate
3. Glypressin
4. Glycylpressin
5. Terlipressine [inn-french]
6. Terlipressinum [inn-latin]
7. Terlipressina [inn-spanish]
8. N-(n-(n-glycylglycyl)glycyl)-8-l-lysinevasopressin
9. (2s)-1-[(4r,7s,10s,13s,16s,19r)-19-[[2-[[2-[(2-aminoacetyl)amino]acetyl]amino]acetyl]amino]-7-(2-amino-2-oxoethyl)-10-(3-amino-3-oxopropyl)-13-benzyl-16-[(4-hydroxyphenyl)methyl]-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentazacycloicosane-4-carbonyl]-n-[(2s)-6-amino-1-[(2-amino-2-oxoethyl)amino]-1-oxohexan-2-yl]pyrrolidine-2-carboxamide
10. Ncgc00185754-01
11. Terlipressina
12. Terlipressine
13. Terlipressinum
14. Lucassin
15. Einecs 238-680-8
16. Terlivaz
17. Variquel
18. Unii-7z5x49w53p
19. Terlipressin [usan:inn:ban]
20. Lucassin (tn)
21. Hs-2028
22. Terlipressin (usan/inn)
23. Dsstox_cid_28878
24. Dsstox_rid_83146
25. Dsstox_gsid_48952
26. Schembl22699
27. Chembl2135460
28. Dtxsid7048952
29. Gtpl11241
30. Chebi:135905
31. 7z5x49w53p
32. Amy25370
33. Ex-a3116
34. Glycyl-glycyl-glycyl-l-cysteinyl-l-tyrosyl-l-phenylalanyl-l-glutaminyl-l-asparagyl-l-cysteinyl-l-prolyl-l-lysyl-glycinamide (4->9)-disulfide
35. Tox21_113374
36. Akos015994637
37. Ccg-270662
38. Cs-5769
39. Db02638
40. Hy-12554
41. Cas-14636-12-5
42. 36t125
43. C72780
44. D06672
45. Q324147
46. J-008213
47. (2s)-6-amino-2-{[(2s)-1-{[(4r,7s,10s,13s,16s,19r)-19-{2-[2-(2-aminoacetamido)acetamido]acetamido}-13-benzyl-10-(2-carbamoylethyl)-7-(carbamoylmethyl)-16-[(4-hydroxyphenyl)methyl]-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaazacycloicosan-4-yl]carbonyl}pyrrolidin-2-yl]formamido}-n-(carbamoylmethyl)hexanamide
| Molecular Weight | 1227.4 g/mol |
|---|---|
| Molecular Formula | C52H74N16O15S2 |
| XLogP3 | -5.8 |
| Hydrogen Bond Donor Count | 16 |
| Hydrogen Bond Acceptor Count | 19 |
| Rotatable Bond Count | 25 |
| Exact Mass | 1226.49609806 g/mol |
| Monoisotopic Mass | 1226.49609806 g/mol |
| Topological Polar Surface Area | 563 Ų |
| Heavy Atom Count | 85 |
| Formal Charge | 0 |
| Complexity | 2380 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 8 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Commonly used to stop bleeding of varices in the food pipe (oesophagus).
Terlipressin is a medicine similar to a naturally occurring hormone present in the body, known as antidiuretic hormone (ADH) or vasopressin. ADH has two main effects in the body. Firstly, it causes narrowing of blood vessels (vasoconstriction), thereby limiting blood flow to a particular area of the body. It also acts on receptors in the kidney to retain water in the body, which helps to prevent excessive loss of water in the urine.
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
Vasoconstrictor Agents
Drugs used to cause constriction of the blood vessels. (See all compounds classified as Vasoconstrictor Agents.)
H01BA04
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
H - Systemic hormonal preparations, excl. sex hormones and insulins
H01 - Pituitary and hypothalamic hormones and analogues
H01B - Posterior pituitary lobe hormones
H01BA - Vasopressin and analogues
H01BA04 - Terlipressin
Terlipressin, an analogue of vasopressin, acts on three different receptors, vasopressin receptor V1a (which initiates vasoconstriction, liver gluconeogenesis, platelet aggregation and release of factor VIII), vasopressin receptor V1b (which mediates corticotrophin secretion from the pituitary) and vasopressin receptor V2 which controls free water reabsorption in the renal medullar. The binding of terlipressin to the V2 receptor activates adenylate cyclase which causes the release of aquaporin 2 channels into the cells lining the renal medullar duct. This allows water to be reabsorbed down an osmotic gradient so the urine is more concentrated.
 ChemWerth works in generic API development & supply, non-infringement patent strategy development and regulatory support.
ChemWerth works in generic API development & supply, non-infringement patent strategy development and regulatory support.
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 34391
Submission : 2019-12-06
Status : Active
Type : II

NDC Package Code : 63586-0124
Start Marketing Date : 2019-07-08
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (4g/4g)
Marketing Category : BULK INGREDIENT

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25589
Submission : 2011-12-21
Status : Inactive
Type : II

Certificate Number : R0-CEP 2019-316 - Rev 00
Issue Date : 2021-05-21
Type : Chemical
Substance Number : 2646
Status : Valid

NDC Package Code : 58862-0179
Start Marketing Date : 2022-09-14
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

Registrant Name : Ferring Korea Co., Ltd.
Registration Date : 2005-08-31
Registration Number : 20050831-59-A-113-01
Manufacturer Name : PolyPeptide Laboratories (Sweden) AB
Manufacturer Address : Hogerudsgatan 21 SE-216 13 Limhamn


Certificate Number : R0-CEP 2021-470 - Rev 00
Issue Date : 2022-12-06
Type : Chemical
Substance Number : 2646
Status : Valid
Date of Issue : 2023-02-09
Valid Till : 2026-02-08
Written Confirmation Number : WC-0549
Address of the Firm :

NDC Package Code : 41701-016
Start Marketing Date : 2011-03-18
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (6g/6g)
Marketing Category : BULK INGREDIENT

Registrant Name : Aging Life Science Co., Ltd.
Registration Date : 2014-07-25
Registration Number : 20140725-59-A-289-04
Manufacturer Name : Hemmo Pharmaceuticals Pvt. Ltd.
Manufacturer Address : C-43, MIDC, TTC Industrial Area, Turbhe, Off Thane Belapur Road, Dist. Thane-400613, India



Certificate Number : R0-CEP 2020-319 - Rev 00
Issue Date : 2021-10-28
Type : Chemical
Substance Number : 2646
Status : Valid

NDC Package Code : 14403-0026
Start Marketing Date : 2025-11-12
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

Certificate Number : R0-CEP 2020-111 - Rev 00
Issue Date : 2022-05-23
Type : Chemical
Substance Number : 2646
Status : Withdrawn by Holder

Date of Issue : 2022-06-08
Valid Till : 2025-07-25
Written Confirmation Number : WC-0159nA2
Address of the Firm :


USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39987
Submission : 2024-05-29
Status : Active
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 34391
Submission : 2019-12-06
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25589
Submission : 2011-12-21
Status : Inactive
Type : II

USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39987
Submission : 2024-05-29
Status : Active
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 9995
Submission : 1992-12-10
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
94
PharmaCompass offers a list of Terlipressin Acetate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Terlipressin Acetate manufacturer or Terlipressin Acetate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Terlipressin Acetate manufacturer or Terlipressin Acetate supplier.
A Terlipressin Acetate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Terlipressin Acetate, including repackagers and relabelers. The FDA regulates Terlipressin Acetate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Terlipressin Acetate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Terlipressin Acetate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Terlipressin Acetate supplier is an individual or a company that provides Terlipressin Acetate active pharmaceutical ingredient (API) or Terlipressin Acetate finished formulations upon request. The Terlipressin Acetate suppliers may include Terlipressin Acetate API manufacturers, exporters, distributors and traders.
click here to find a list of Terlipressin Acetate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Terlipressin Acetate DMF (Drug Master File) is a document detailing the whole manufacturing process of Terlipressin Acetate active pharmaceutical ingredient (API) in detail. Different forms of Terlipressin Acetate DMFs exist exist since differing nations have different regulations, such as Terlipressin Acetate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Terlipressin Acetate DMF submitted to regulatory agencies in the US is known as a USDMF. Terlipressin Acetate USDMF includes data on Terlipressin Acetate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Terlipressin Acetate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Terlipressin Acetate suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Terlipressin Acetate Drug Master File in Korea (Terlipressin Acetate KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Terlipressin Acetate. The MFDS reviews the Terlipressin Acetate KDMF as part of the drug registration process and uses the information provided in the Terlipressin Acetate KDMF to evaluate the safety and efficacy of the drug.
After submitting a Terlipressin Acetate KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Terlipressin Acetate API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Terlipressin Acetate suppliers with KDMF on PharmaCompass.
A Terlipressin Acetate CEP of the European Pharmacopoeia monograph is often referred to as a Terlipressin Acetate Certificate of Suitability (COS). The purpose of a Terlipressin Acetate CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Terlipressin Acetate EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Terlipressin Acetate to their clients by showing that a Terlipressin Acetate CEP has been issued for it. The manufacturer submits a Terlipressin Acetate CEP (COS) as part of the market authorization procedure, and it takes on the role of a Terlipressin Acetate CEP holder for the record. Additionally, the data presented in the Terlipressin Acetate CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Terlipressin Acetate DMF.
A Terlipressin Acetate CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Terlipressin Acetate CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Terlipressin Acetate suppliers with CEP (COS) on PharmaCompass.
A Terlipressin Acetate written confirmation (Terlipressin Acetate WC) is an official document issued by a regulatory agency to a Terlipressin Acetate manufacturer, verifying that the manufacturing facility of a Terlipressin Acetate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Terlipressin Acetate APIs or Terlipressin Acetate finished pharmaceutical products to another nation, regulatory agencies frequently require a Terlipressin Acetate WC (written confirmation) as part of the regulatory process.
click here to find a list of Terlipressin Acetate suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Terlipressin Acetate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Terlipressin Acetate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Terlipressin Acetate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Terlipressin Acetate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Terlipressin Acetate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Terlipressin Acetate suppliers with NDC on PharmaCompass.
Terlipressin Acetate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Terlipressin Acetate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Terlipressin Acetate GMP manufacturer or Terlipressin Acetate GMP API supplier for your needs.
A Terlipressin Acetate CoA (Certificate of Analysis) is a formal document that attests to Terlipressin Acetate's compliance with Terlipressin Acetate specifications and serves as a tool for batch-level quality control.
Terlipressin Acetate CoA mostly includes findings from lab analyses of a specific batch. For each Terlipressin Acetate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Terlipressin Acetate may be tested according to a variety of international standards, such as European Pharmacopoeia (Terlipressin Acetate EP), Terlipressin Acetate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Terlipressin Acetate USP).

