
Synopsis
Synopsis
0
USDMF
0
JDMF
0
EU WC
0
KDMF
0
FDA Orange Book
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Panacur
2. Phenbendasol
1. 43210-67-9
2. Panacur
3. Fenbendazol
4. Phenbendasol
5. Fenbendazolum
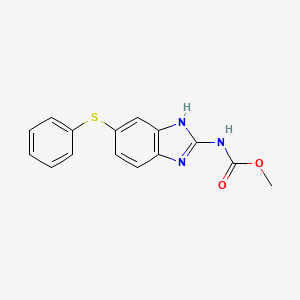
6. Methyl 5-(phenylthio)-2-benzimidazolecarbamate
7. Hoe 881v
8. Safe-guard
9. Safe-quard
10. Hoe-881v
11. 2-(methoxycarbonylamino)-5-(phenylthio)benzimidazole
12. Panacur Aquasol
13. Methyl [5-(phenylsulfanyl)-1h-benzimidazol-2-yl]carbamate
14. Methyl (6-(phenylthio)-1h-benzo[d]imidazol-2-yl)carbamate
15. Methyl N-(6-phenylsulfanyl-1h-benzimidazol-2-yl)carbamate
16. Fenbendazole (panacur)
17. Fenbendazole (usp/inn)
18. Mfcd00144301
19. Nsc-757824
20. Methyl N-[5-(phenylsulfanyl)-1h-1,3-benzodiazol-2-yl]carbamate
21. Carbamic Acid, (5-(phenylthio)-1h-benzimidazol-2-yl)-, Methyl Ester
22. Carbamic Acid, [5-(phenylthio)-1h-benzimidazol-2-yl]-, Methyl Ester
23. Methyl (5-(phenylthio)-1h-benzimidazol-2-yl)carbamate
24. Chembl37161
25. 5-(phenylthio)-2-benzimidazolecarbamic Acid Methyl Ester
26. Fenbendazole For Veterinary Use
27. Mls000069723
28. Hoe-881y
29. 2-benzimidazolecarbamic Acid, 5-(phenylthio)-, Methyl Ester
30. Chebi:77092
31. [5-(phenylthio)-1h-benzimidazol-2-yl]carbamic Acid Methyl Ester
32. Hoe 881
33. 621bvt9m36
34. N-[6-(phenylthio)-1h-benzimidazol-2-yl]carbamic Acid Methyl Ester
35. Carbamic Acid, N-[6-(phenylthio)-1h-benzimidazol-2-yl]-, Methyl Ester
36. Methyl [5-(phenylthio)-1h-benzimidazol-2-yl]carbamate
37. Ncgc00016855-01
38. Smr000058217
39. Cas-43210-67-9
40. Dsstox_cid_20672
41. Dsstox_rid_79532
42. Dsstox_gsid_40672
43. Methyl 5-(phenylthio)-1h-benzo[d]imidazol-2-ylcarbamate
44. Methyl (5-(phenylthio)-1h-benzo[d]imidazol-2-yl)carbamate
45. Methyl N-(5-phenylsulfanyl-1h-benzimidazol-2-yl)carbamate
46. Fenbendazole D3 (methyl D3)
47. Fenbendazol [inn-spanish]
48. Fenbendazolum [inn-latin]
49. Ccris 7309
50. Fenbendazole [usan:ban:inn]
51. Einecs 256-145-7
52. Pancacur
53. Axilur
54. Unii-621bvt9m36
55. Worm-a-rest
56. Prestwick_213
57. Fenbendazole [usan:usp:inn:ban]
58. Panacur (tn)
59. Spectrum_001254
60. Fenbendazole, >=98%
61. Prestwick0_000210
62. Prestwick1_000210
63. Prestwick2_000210
64. Prestwick3_000210
65. Spectrum2_000040
66. Spectrum3_000911
67. Spectrum4_001010
68. Spectrum5_001359
69. Fenbendazole [mi]
70. Fenbendazole [inn]
71. Fenbendazole [usan]
72. Cid_3334
73. Panacur [veterinary] (tn)
74. Oprea1_477843
75. Bspbio_000199
76. Kbiogr_001479
77. Kbioss_001734
78. Methyl [5-(phenylthio)benzimidazol-2-yl]carbamate
79. Mls006010049
80. Divk1c_000257
81. Fenbendazole [usp-rs]
82. Fenbendazole [who-dd]
83. Schembl166352
84. Spectrum1501016
85. Spbio_000139
86. Spbio_002120
87. Bpbio1_000219
88. Hoe 881y
89. Schembl8469702
90. Dtxsid0040672
91. Schembl20796999
92. Bdbm31047
93. Hms500m19
94. Kbio1_000257
95. Kbio2_001734
96. Kbio2_004302
97. Kbio2_006870
98. Kbio3_001882
99. Fenbendazole [green Book]
100. Ninds_000257
101. [5-(phenylthio)benzimidazol-2-yl]carbamic Acid Methyl Ester
102. Hms1568j21
103. Hms1921d03
104. Hms2090g12
105. Hms2092b11
106. Hms2095j21
107. Hms2234p14
108. Hms3371j16
109. Hms3656c04
110. Hms3712j21
111. Pharmakon1600-01501016
112. Zinc402911
113. Bcp22258
114. Hy-b0413
115. Fenbendazole [usp Monograph]
116. Tox21_110649
117. Tox21_301828
118. Ccg-39631
119. Nsc757824
120. S2468
121. Stk378581
122. Akos005448335
123. Akos024462438
124. Tox21_110649_1
125. Ac-8717
126. Ccg-220210
127. Db11410
128. Ks-5042
129. Nsc 757824
130. Fenbendazole 100 Microg/ml In Methanol
131. Idi1_000257
132. Ncgc00016855-02
133. Ncgc00016855-03
134. Ncgc00016855-04
135. Ncgc00016855-05
136. Ncgc00016855-06
137. Ncgc00016855-07
138. Ncgc00016855-08
139. Ncgc00016855-10
140. Ncgc00016855-11
141. Ncgc00022515-03
142. Ncgc00022515-04
143. Ncgc00022515-05
144. Ncgc00255694-01
145. Fenbendazole [ema Epar Veterinary]
146. Smr004701217
147. Sy052602
148. Sbi-0051634.p002
149. Ab00052199
150. F0812
151. Ft-0630630
152. Sw196522-3
153. C21876
154. D04140
155. Ab00052199-13
156. Ab00052199-14
157. Ab00052199_15
158. Ab00052199_16
159. Fenbendazole, Vetranal(tm), Analytical Standard
160. 210f679
161. A826225
162. Methyl 5-(phenyl-thio)-2-benzimidazole-carbamate
163. Methyl 5-phenylthio-1h-benzimidazol-2-ylcarbamate
164. Q908013
165. Sr-01000000195
166. Sr-05000001768
167. Q-201109
168. Sr-01000000195-2
169. Sr-05000001768-1
170. Brd-k51318897-001-04-7
171. Brd-k51318897-001-07-0
172. Brd-k56309460-001-03-0
173. Fenbendazole For Veterinary Use [ep Impurity]
174. Fenbendazole For Veterinary Use [ep Monograph]
175. Methyl 5-(phenylsulfanyl)-1h-benzimidazol-2-ylcarbamate #
176. Oxfendazole Impurity, Fenbendazole- [usp Impurity]
177. Fenbendazole, European Pharmacopoeia (ep) Reference Standard
178. Oxfendazole For Veterinary Use Impurity A [ep Impurity]
179. (5-phenylsulfanyl-1h-benzoimidazol-2-yl)-carbamic Acid Methyl Ester
180. Fenbendazole, United States Pharmacopeia (usp) Reference Standard
181. Panacur; Phenbendasol; Safe-quard; Hoe 881v; Hoe881v; Hoe-881v
182. Fenbendazole, Pharmaceutical Secondary Standard; Certified Reference Material
1. Ofdz
2. Synanthic
3. Systamex
4. Fenbendazole Sulfoxide
5. Oxfendazole
| Molecular Weight | 299.3 g/mol |
|---|---|
| Molecular Formula | C15H13N3O2S |
| XLogP3 | 3.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 299.07284784 g/mol |
| Monoisotopic Mass | 299.07284784 g/mol |
| Topological Polar Surface Area | 92.3 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 363 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
For the treatment and control of gastro-intestinal nematodes in pigs infected with:
- Ascaris suum (adult, intestinal and migrating larval stages);
- Oesophagostomum spp. (adult stages);
- Trichuris suis (adult stages).
For the treatment of gastro-intestinal nematodes in chickens infected with:
- Ascaridia galli (L5 and adult stages);
- Heterakis gallinarum (L5 and adult stages);
Capillaria spp. (L5 and adult stages).
Antinematodal Agents
Substances used in the treatment or control of nematode infestations. They are used also in veterinary practice. (See all compounds classified as Antinematodal Agents.)
QP52AC13
P - Antiparasitic products, insecticides and repellents
P02 - Anthelmintics
P02C - Antinematodal agents
P02CA - Benzimidazole derivatives
P02CA06 - Fenbendazole
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
86
PharmaCompass offers a list of Fenbendazole API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Fenbendazole manufacturer or Fenbendazole supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Fenbendazole manufacturer or Fenbendazole supplier.
A Synanthic manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Synanthic, including repackagers and relabelers. The FDA regulates Synanthic manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Synanthic API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Synanthic manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Synanthic supplier is an individual or a company that provides Synanthic active pharmaceutical ingredient (API) or Synanthic finished formulations upon request. The Synanthic suppliers may include Synanthic API manufacturers, exporters, distributors and traders.
click here to find a list of Synanthic suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Synanthic CEP of the European Pharmacopoeia monograph is often referred to as a Synanthic Certificate of Suitability (COS). The purpose of a Synanthic CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Synanthic EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Synanthic to their clients by showing that a Synanthic CEP has been issued for it. The manufacturer submits a Synanthic CEP (COS) as part of the market authorization procedure, and it takes on the role of a Synanthic CEP holder for the record. Additionally, the data presented in the Synanthic CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Synanthic DMF.
A Synanthic CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Synanthic CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Synanthic suppliers with CEP (COS) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Synanthic as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Synanthic API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Synanthic as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Synanthic and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Synanthic NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Synanthic suppliers with NDC on PharmaCompass.
Synanthic Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Synanthic GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Synanthic GMP manufacturer or Synanthic GMP API supplier for your needs.
A Synanthic CoA (Certificate of Analysis) is a formal document that attests to Synanthic's compliance with Synanthic specifications and serves as a tool for batch-level quality control.
Synanthic CoA mostly includes findings from lab analyses of a specific batch. For each Synanthic CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Synanthic may be tested according to a variety of international standards, such as European Pharmacopoeia (Synanthic EP), Synanthic JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Synanthic USP).

