
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Aclin
2. Apo Sulin
3. Apo-sulin
4. Arthrobid
5. Arthrocine
6. Chibret
7. Clinoril
8. Copal
9. Kenalin
10. Klinoril
11. Mk 231
12. Mk-231
13. Mk231
14. Novo Sundac
15. Novo-sundac
16. Nu Sulindac
17. Nu-sulindac
18. Sulindal
1. Clinoril
2. 38194-50-2
3. Arthrocine
4. Artribid
5. Sulindaco
6. Sulindacum
7. Aflodac
8. Sulreuma
9. Sulindac Sulfoxide
10. (z)-2-(5-fluoro-2-methyl-1-(4-(methylsulfinyl)benzylidene)-1h-inden-3-yl)acetic Acid
11. Mobilin
12. Cis-5-fluoro-2-methyl-1-((p-methylsulfinyl)benzylidene)indene-3-acetic Acid
13. Chebi:9352
14. (z)-5-fluoro-2-methyl-1-((p-(methylsulfinyl)phenyl)methylene)-1h-indene-3-acetic Acid
15. Cis-5-fluoro-2-methyl-1-((4-(methylsulfinyl)phenyl)methylene)-1h-indene-3-acetic Acid
16. Mk 231
17. Mk-231
18. Nsc-757344
19. 184sns8vuh
20. Algocetil
21. Citireuma
22. Clisundac
23. Imbaral
24. Reumofil
25. Sulinol
26. Chembl15770
27. Sudac
28. 1h-indene-3-acetic Acid, 5-fluoro-2-methyl-1-((4-(methylsulfinyl)phenyl)methylene)-, (z)-
29. 2-[(1z)-5-fluoro-1-[(4-methanesulfinylphenyl)methylidene]-2-methyl-1h-inden-3-yl]acetic Acid
30. 2-[(3z)-6-fluoro-2-methyl-3-[(4-methylsulfinylphenyl)methylidene]inden-1-yl]acetic Acid
31. Ncgc00015970-02
32. Cas-38194-50-2
33. Dsstox_cid_3624
34. Sulindacum [inn-latin]
35. Dsstox_rid_77117
36. Dsstox_gsid_23624
37. Sulindaco [inn-spanish]
38. Cis-sulindac
39. Sulindac (clinoril)
40. (s,z)-2-(5-fluoro-2-methyl-1-(4-(methylsulfinyl)benzylidene)-1h-inden-3-yl)acetic Acid
41. (z)-2-(5-fluoro-2-methyl-1-(4-(methylsulfinyl)-benzylidene)-1h-inden-3-yl)acetic Acid
42. 1h-indene-3-acetic Acid, 5-fluoro-2-methyl-1-[[4-(methylsulfinyl)phenyl]methylene]-, (1z)-
43. Clinoril (tn)
44. Ccris 3305
45. {(1z)-5-fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1h-inden-3-yl}acetic Acid
46. Sr-01000075237
47. 32004-68-5
48. Einecs 253-819-2
49. Unii-184sns8vuh
50. Brn 2951842
51. Moblilin
52. Mfcd00599589
53. (z)-2-[5-fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1h-inden-3-yl]acetic Acid
54. 1h-indene-3-aceticacid, 5-fluoro-2-methyl-1-[[4-(methylsulfinyl)phenyl]methylene]-, (1z)-
55. 1h-indene-3-acetic Acid, 5-fluoro-2-methyl-1-((4-(methylsulfinyl)phenyl)methylene)-, (1z)-
56. Einecs 250-893-8
57. Einecs 256-402-3
58. Sulindac [usan:usp:inn:ban:jan]
59. Tocris-1707
60. Sulindac [inci]
61. Sulindac [usan]
62. Sulindac [inn]
63. Sulindac [jan]
64. Sulindac [mi]
65. Sulindac [vandf]
66. Prestwick3_000073
67. Spectrum5_001024
68. Lopac-s-8139
69. Sulindac [mart.]
70. Sulindac [usp-rs]
71. Sulindac [who-dd]
72. S 8139
73. Sulindac, >=98.0%
74. Schembl4202
75. Schembl4203
76. 5-fluoro-2-methyl-1-((4-(methylsulphinyl)phenyl)methylene)-1h-indene-3-acetic Acid
77. Lopac0_001070
78. Bspbio_000285
79. Bspbio_002890
80. Mls001056554
81. Spectrum1500556
82. Sulindac (jp17/usp/inn)
83. Sulindac [orange Book]
84. Bpbio1_000315
85. Gtpl5425
86. Sulindac [ep Monograph]
87. Dtxsid4023624
88. Sulindac [usp Monograph]
89. Chebi:93811
90. Hms501o03
91. Cmap_000021
92. Hms1921c11
93. Hms2092k15
94. Hms2095o07
95. Hms2231n24
96. Hms3259k06
97. Hms3263e22
98. Hms3414n11
99. Hms3649p19
100. Hms3678n09
101. Hms3712o07
102. Hms3884k18
103. Pharmakon1600-01500556
104. Hy-b0008
105. Tox21_110270
106. Tox21_301418
107. Tox21_501070
108. Bdbm50012899
109. Bdbm50103504
110. Ccg-39264
111. Nsc757344
112. S2007
113. Akos015895412
114. Cis-5-fluoro-2-methyl-1-((p-methylsulfinyl)benzylidenyl)indene-3-acetic Acid
115. Tox21_110270_1
116. Cs-0569
117. Db00605
118. Ks-5153
119. Lp01070
120. Nc00540
121. Nsc 757344
122. Sdccgsbi-0051040.p005
123. (z)-(1)-5-fluoro-2-methyl-1-((4-(methylsulphinyl)phenyl)methylene)-1h-indene-3-acetic Acid
124. (z)-5-fluoro-2-methyl-1-[[4-(methyl-sulfinyl)phenyl]methylene]-1h-indene-3-acetic Acid
125. Idi1_000601
126. Sulindac 100 Microg/ml In Acetonitrile
127. Ncgc00015970-01
128. Ncgc00015970-03
129. Ncgc00015970-04
130. Ncgc00015970-05
131. Ncgc00015970-06
132. Ncgc00015970-07
133. Ncgc00015970-08
134. Ncgc00015970-11
135. Ncgc00025268-01
136. Ncgc00025268-02
137. Ncgc00025268-03
138. Ncgc00094349-01
139. Ncgc00094349-02
140. Ncgc00255143-01
141. Ncgc00261755-01
142. Ncgc00381714-10
143. Bp-30208
144. Bs166205
145. Ls-14757
146. Smr000326718
147. Sbi-0051040.p004
148. Sulindac, Meets Usp Testing Specifications
149. Ab00513800
150. Eu-0101070
151. Sw219748-1
152. U0099
153. C01531
154. D00120
155. Ab00052105-07
156. Ab00052105_08
157. Ab00052105_09
158. Q963093
159. J-008554
160. J-012337
161. Sr-01000075237-2
162. Sr-01000075237-3
163. Sr-01000075237-5
164. Sr-01000075237-7
165. Brd-a13946108-001-04-9
166. Brd-a13946108-001-08-0
167. Sr-01000075237-13
168. Sulindac, European Pharmacopoeia (ep) Reference Standard
169. Sulindac, United States Pharmacopeia (usp) Reference Standard
170. (z)-5-fluoro-2-methyl-1-[(p-methylsulfinyl)benzylidene]-1h-indene-3-acetic Acid
171. (z)-5-fluoro-2-methyl-1-[[4-(methylsulfinyl)-phenyl]methylene]-1h-indene-3-acetic Acid
172. {(1z)-5-fluoro-2-methyl-1-[4-(methylsulfinyl)benzylidene]-1h-indene-3-yl}acetic Acid
173. 2-[(3z)-6-fluoro-2-methyl-3-[(4-methylsulfinylphenyl)methylene]inden-1-yl]acetic Acid
174. Sulindac
175. (1z)-5-fluoro-2-methyl-1-[[4-(methylsulfinyl)phenyl]methylene]-1h-indene-3-acetic Acid
| Molecular Weight | 356.4 g/mol |
|---|---|
| Molecular Formula | C20H17FO3S |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Exact Mass | 356.08824374 g/mol |
| Monoisotopic Mass | 356.08824374 g/mol |
| Topological Polar Surface Area | 73.6 Ų |
| Heavy Atom Count | 25 |
| Formal Charge | 0 |
| Complexity | 616 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Clinoril |
| PubMed Health | Sulindac (By mouth) |
| Drug Classes | Analgesic, Antirheumatic, Central Nervous System Agent, Musculoskeletal Agent |
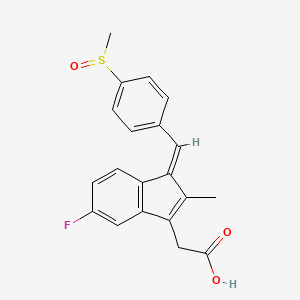
| Drug Label | Sulindac is a non-steroidal, anti-inflammatory indene derivative designated chemically as (Z)-5-fluoro-2-methyl-1- [[p-(methylsulfinyl)phenyl]methylene]-1H-indene-3-acetic acid. It is not a salicylate, pyrazolone or propionic acid derivative. Sulinda... |
| Active Ingredient | Sulindac |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg |
| Market Status | Prescription |
| Company | Merck |
| 2 of 4 | |
|---|---|
| Drug Name | Sulindac |
| PubMed Health | Sulindac (By mouth) |
| Drug Classes | Analgesic, Antirheumatic, Central Nervous System Agent, Musculoskeletal Agent |
| Active Ingredient | Sulindac |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 150mg |
| Market Status | Prescription |
| Company | Epic Pharma; Watson Labs; Mutual Pharm; Mylan; Heritage Pharms |
| 3 of 4 | |
|---|---|
| Drug Name | Clinoril |
| PubMed Health | Sulindac (By mouth) |
| Drug Classes | Analgesic, Antirheumatic, Central Nervous System Agent, Musculoskeletal Agent |
| Drug Label | Sulindac is a non-steroidal, anti-inflammatory indene derivative designated chemically as (Z)-5-fluoro-2-methyl-1- [[p-(methylsulfinyl)phenyl]methylene]-1H-indene-3-acetic acid. It is not a salicylate, pyrazolone or propionic acid derivative. Sulinda... |
| Active Ingredient | Sulindac |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg |
| Market Status | Prescription |
| Company | Merck |
| 4 of 4 | |
|---|---|
| Drug Name | Sulindac |
| PubMed Health | Sulindac (By mouth) |
| Drug Classes | Analgesic, Antirheumatic, Central Nervous System Agent, Musculoskeletal Agent |
| Active Ingredient | Sulindac |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 150mg |
| Market Status | Prescription |
| Company | Epic Pharma; Watson Labs; Mutual Pharm; Mylan; Heritage Pharms |
For acute or long-term use in the relief of signs and symptoms of osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, acute painful shoulder (acute subacromial bursitis/supraspinatus tendinitis), and acute gouty arthritis.
FDA Label
Sulindac is a non-steroidal anti-inflammatory indene derivative, also possessing analgesic and antipyretic activities.
Anti-Inflammatory Agents, Non-Steroidal
Anti-inflammatory agents that are non-steroidal in nature. In addition to anti-inflammatory actions, they have analgesic, antipyretic, and platelet-inhibitory actions. They act by blocking the synthesis of prostaglandins by inhibiting cyclooxygenase, which converts arachidonic acid to cyclic endoperoxides, precursors of prostaglandins. Inhibition of prostaglandin synthesis accounts for their analgesic, antipyretic, and platelet-inhibitory actions; other mechanisms may contribute to their anti-inflammatory effects. (See all compounds classified as Anti-Inflammatory Agents, Non-Steroidal.)
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Cyclooxygenase Inhibitors
Compounds or agents that combine with cyclooxygenase (PROSTAGLANDIN-ENDOPEROXIDE SYNTHASES) and thereby prevent its substrate-enzyme combination with arachidonic acid and the formation of eicosanoids, prostaglandins, and thromboxanes. (See all compounds classified as Cyclooxygenase Inhibitors.)
M - Musculo-skeletal system
M01 - Antiinflammatory and antirheumatic products
M01A - Antiinflammatory and antirheumatic products, non-steroids
M01AB - Acetic acid derivatives and related substances
M01AB02 - Sulindac
Absorption
Approximately 90% absorbed in humans following oral administration.
Route of Elimination
Sulindac is excreted in rat milk; concentrations in milk were 10 to 20% of those levels in plasma. It is not known if sulindac is excreted in human milk. Approximately 50% of the administered dose of sulindac is excreted in the urine with the conjugated sulfone metabolite accounting for the major portion. Hepatic metabolism is an important elimination pathway.
Clearance
Renal cl=68.12 +/- 27.56 mL/min [NORMAL (19-41 yrs)]
Undergoes two major biotransformations: reversible reduction to the sulfide metabolite, and irreversible oxidation to the sulfone metabolite. Sulindac and its sulfide and sulfone metabolites undergo extensive enterohepatic circulation. Available evidence indicates that the biological activity resides with the sulfide metabolite. Side chain hydroxylation and hydration of the double bond also occur.
The mean half-life of sulindac is 7.8 hours while the mean half-life of the sulfide metabolite is 16.4 hours.
Sulindac's exact mechanism of action is unknown. Its antiinflammatory effects are believed to be due to inhibition of both COX-1 and COX-2 which leads to the inhibition of prostaglandin synthesis. Antipyretic effects may be due to action on the hypothalamus, resulting in an increased peripheral blood flow, vasodilation, and subsequent heat dissipation.
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

ABOUT THIS PAGE
49
PharmaCompass offers a list of Sulindac API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sulindac manufacturer or Sulindac supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Sulindac manufacturer or Sulindac supplier.
A Sulindac manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Sulindac, including repackagers and relabelers. The FDA regulates Sulindac manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Sulindac API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Sulindac manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Sulindac supplier is an individual or a company that provides Sulindac active pharmaceutical ingredient (API) or Sulindac finished formulations upon request. The Sulindac suppliers may include Sulindac API manufacturers, exporters, distributors and traders.
click here to find a list of Sulindac suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Sulindac DMF (Drug Master File) is a document detailing the whole manufacturing process of Sulindac active pharmaceutical ingredient (API) in detail. Different forms of Sulindac DMFs exist exist since differing nations have different regulations, such as Sulindac USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Sulindac DMF submitted to regulatory agencies in the US is known as a USDMF. Sulindac USDMF includes data on Sulindac's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Sulindac USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Sulindac suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Sulindac as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Sulindac API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Sulindac as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Sulindac and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Sulindac NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Sulindac suppliers with NDC on PharmaCompass.
Sulindac Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Sulindac GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sulindac GMP manufacturer or Sulindac GMP API supplier for your needs.
A Sulindac CoA (Certificate of Analysis) is a formal document that attests to Sulindac's compliance with Sulindac specifications and serves as a tool for batch-level quality control.
Sulindac CoA mostly includes findings from lab analyses of a specific batch. For each Sulindac CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Sulindac may be tested according to a variety of international standards, such as European Pharmacopoeia (Sulindac EP), Sulindac JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Sulindac USP).

