
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. 7631-90-5
2. Sodium Hydrogen Sulfite
3. Sodium Hydrogensulfite
4. Sodium Bisulphite
5. Sulfurous Acid, Monosodium Salt
6. Sodium Sulhydrate
7. Monosodium Sulfite
8. Sodium Hydrogen Sulphite
9. Monosodic Sulphite
10. Nahso3
11. Liquid Of Sodium Bisulfite
12. Ins No.222
13. Tzx5469z6i
14. Chembl1689285
15. Chebi:26709
16. Ins-222
17. Sulfurous Acid, Sodium Salt (1:1)
18. Nsc-60680
19. E-222
20. Uantax Sbs
21. Sodium Bisulfite, Mixture Of Nahso3 And Na2s2o5
22. Caswell No. 750
23. Hydrogen Sodium Sulfite
24. Sodium Bisulfite [jan]
25. Sodium Hydrosulfite(dot)
26. Mfcd00003530
27. Bisulfite De Sodium [french]
28. Ccris 3950
29. Hsdb 724
30. Sodium Hydrogensulphite
31. Einecs 231-548-0
32. Nsc 60680
33. Epa Pesticide Chemical Code 078201
34. Sodium Bisulfite [jan:nf]
35. Sodiumbisulfit
36. Unii-tzx5469z6i
37. Sodium Hydrogensulphite (aqueous Solution)
38. Ai3-08582
39. Sodium Hydrogen Sulfite, Solution
40. Sodium Bi-sulphite
41. Sodiumhydrogensulfit
42. Saures Natriumsulfit
43. Natriumhydrogensulfit
44. Sodiumhydrogensulphite
45. Sodium Hydogen Sulfite
46. Primaeres Natriumsulfit
47. Sodium Hydrogen-sulfite
48. Sodium;hydrogen Sulfite
49. E222
50. Sodium Bisulfite (tn)
51. Sodium Bisulfite (jp17)
52. Ec 231-548-0
53. Sodium Hydrogen Sulfite Solution (45% Or Less)
54. Dsstox_cid_14902
55. Dsstox_gsid_47218
56. Sodium Bisulfite [ii]
57. Sodium Bisulfite [mi]
58. Sodium Bisulfite, Acs Reagent
59. Sodium Bisulfite [fcc]
60. Sodium Bisulfite [hsdb]
61. Sodium Bisulfite [inci]
62. Dtxsid8034902
63. Sodium Bisulfite [vandf]
64. Sodium Bisulfite [mart.]
65. Sodium Bisulfite [who-dd]
66. Tox21_303826
67. Akos015856800
68. Db14015
69. Ncgc00357103-01
70. Sodium Bisulfite Solution, Purum, ~40%
71. Bp-12336
72. Sodium Bisulfite Aqueous Solution 35-40%
73. Sodium Bisulfite, 37-40% Aqueous Solution
74. Ft-0698943
75. D02059
76. Q407891
77. Sodium Hydrogensulfite, Mixture Of Nahso3 And Na2s2o5
| Molecular Weight | 104.06 g/mol |
|---|---|
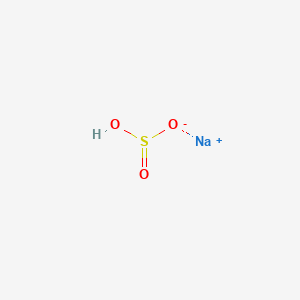
| Molecular Formula | HNaO3S |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Exact Mass | 103.95440935 g/mol |
| Monoisotopic Mass | 103.95440935 g/mol |
| Topological Polar Surface Area | 79.6 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 33.9 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Mutagens; Anti-Infective Agents, Local; Antioxidants
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
DIETARY SUPPLEMENTS
Sax, N.I. and R.J. Lewis, Sr. (eds.). Hawley's Condensed Chemical Dictionary. 11th ed. New York: Van Nostrand Reinhold Co., 1987., p. 1054
Anti-Infective Agents, Local
Substances used on humans and other animals that destroy harmful microorganisms or inhibit their activity. They are distinguished from DISINFECTANTS, which are used on inanimate objects. (See all compounds classified as Anti-Infective Agents, Local.)
Antioxidants
Naturally occurring or synthetic substances that inhibit or retard oxidation reactions. They counteract the damaging effects of oxidation in animal tissues. (See all compounds classified as Antioxidants.)
Mutagens
Chemical agents that increase the rate of genetic mutation by interfering with the function of nucleic acids. A clastogen is a specific mutagen that causes breaks in chromosomes. (See all compounds classified as Mutagens.)
EIGHTY TO 90% OF AN INTRAPERITONEAL DOSE IN RATS CAN BE ACCOUNTED FOR AS URINARY SULFATE IN 4 HR.
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-123
Approx 50% of the ingested bisulfite is absorbed from rat gut.
American Conference of Governmental Industrial Hygienists, Inc. Documentation of the Threshold Limit Values and Biological Exposure Indices. 6th ed. Volumes I, II, III. Cincinnati, OH: ACGIH, 1991., p. 1408
Bisulfite is oxidized in vivo to sulfate, principally by hepatic sulfite oxidase, with lesser amounts metabolized by the kidneys, intestines, heart, & lungs. Some 70 to 95% of the radioactivity assoc with a 50 mg/kg oral bisulfite dose appeared in rodent & monkey urine within 3 days as sulfate. Only a small fraction (8-10%) of the absorbed bisulfite was eliminated intact.
American Conference of Governmental Industrial Hygienists, Inc. Documentation of the Threshold Limit Values and Biological Exposure Indices. 6th ed. Volumes I, II, III. Cincinnati, OH: ACGIH, 1991., p. 1408
Bisulfite is oxidized in vivo to sulfate, principally by hepatic sulfite oxidase, with lesser amounts metabolized by the kidneys, intestines, heart, & lungs.
American Conference of Governmental Industrial Hygienists, Inc. Documentation of the Threshold Limit Values and Biological Exposure Indices. 6th ed. Volumes I, II, III. Cincinnati, OH: ACGIH, 1991., p. 1408
Rat liver contains 10 to 20 times more sulphite oxidase activity than does human liver.
American Conference of Governmental Industrial Hygienists, Inc. Documentation of the Threshold Limit Values and Biological Exposure Indices. 6th ed. Volumes I, II, III. Cincinnati, OH: ACGIH, 1991., p. 1409
Sodium bisulfite is a mutagen that can specifically deaminate more than 96% of the cytosine residues in single-stranded DNA via formation of a 5,6-dihydrocytosine-6-sulfonate intermediate.
PMID:7443525 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC324387 Wang RY et al; Nucleic Acids Res 8 (20): 4777-90 (1980)
Bisulfite concn of 100 mM caused nearly complete inhibition of dNTP incorporation in calf thymus DNA. Preincubation of the various DNA synthesis assay components in 100 mM bisulfite showed that only preincubation of DNA polymerase I caused inhibition of DNA synthesis. There was a 7.5-fold decrease in the fidelity of DNA synthesis. It is hypothesized that sulfitolysis of the one disulfide group in DNA polymerase I by bisulfite might be responsible for reduced polymerase activity and accuracy. Effects of bisulfite on mutation frequency might be mediated by effects on the fidelity of DNA repair systems.
PMID:6352068 Mallon RG, Rossman TG; Chem Biol Interact 46 (1): 101-8 (1983)
Market Place

ABOUT THIS PAGE
83
PharmaCompass offers a list of Sodium Bisulfite API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sodium Bisulfite manufacturer or Sodium Bisulfite supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Sodium Bisulfite manufacturer or Sodium Bisulfite supplier.
A Sodium Bisulfite manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Sodium Bisulfite, including repackagers and relabelers. The FDA regulates Sodium Bisulfite manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Sodium Bisulfite API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Sodium Bisulfite supplier is an individual or a company that provides Sodium Bisulfite active pharmaceutical ingredient (API) or Sodium Bisulfite finished formulations upon request. The Sodium Bisulfite suppliers may include Sodium Bisulfite API manufacturers, exporters, distributors and traders.
click here to find a list of Sodium Bisulfite suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Sodium Bisulfite as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Sodium Bisulfite API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Sodium Bisulfite as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Sodium Bisulfite and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Sodium Bisulfite NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Sodium Bisulfite suppliers with NDC on PharmaCompass.
Sodium Bisulfite Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Sodium Bisulfite GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sodium Bisulfite GMP manufacturer or Sodium Bisulfite GMP API supplier for your needs.
A Sodium Bisulfite CoA (Certificate of Analysis) is a formal document that attests to Sodium Bisulfite's compliance with Sodium Bisulfite specifications and serves as a tool for batch-level quality control.
Sodium Bisulfite CoA mostly includes findings from lab analyses of a specific batch. For each Sodium Bisulfite CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Sodium Bisulfite may be tested according to a variety of international standards, such as European Pharmacopoeia (Sodium Bisulfite EP), Sodium Bisulfite JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Sodium Bisulfite USP).

