Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
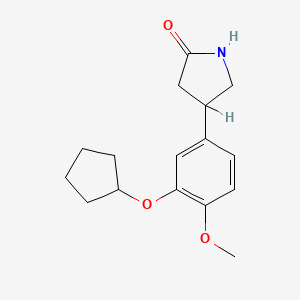

1. Zk 62711
2. Zk-62711
3. Zk62711
1. 61413-54-5
2. (r,s)-rolipram
3. Zk 62711
4. Rolipramum [latin]
5. Adeo
6. (+/-)-rolipram
7. Rolipramum
8. 4-(3-(cyclopentyloxy)-4-methoxyphenyl)pyrrolidin-2-one
9. 4-[3-(cyclopentyloxy)-4-methoxyphenyl]-2-pyrrolidinone
10. 4-[3-(cyclopentyloxy)-4-methoxyphenyl]pyrrolidin-2-one
11. Zk-62711
12. 4-(3-cyclopentyloxy-4-methoxyphenyl)pyrrolidin-2-one
13. 4-(3-(cyclopentyloxy)-4-methoxyphenyl)-2-pyrrolidinone
14. Sb 95952
15. Chembl63
16. Nsc-760125
17. 4-(3-cyclopentyloxy-4-methoxyphenyl)-2-pyrrolidone
18. C16h21no3
19. Mls000069691
20. Zk 62 711
21. 2-pyrrolidinone, 4-[3-(cyclopentyloxy)-4-methoxyphenyl]-
22. Chebi:104872
23. K676nl63n7
24. Sb-95952
25. Smr000058510
26. Zk-62771
27. (4s)-4-[3-(cyclopentyloxy)-4-methoxyphenyl]-pyrrolidin-2-one
28. (r,s)-rolipram;sb 95952;zk 62711
29. Dsstox_cid_24124
30. Dsstox_rid_80104
31. Dsstox_gsid_44124
32. 4-(3-cyclopentyloxy-4-methoxy-phenyl)-pyrrolidin-2-one
33. 2-pyrrolidinone, 4-(3-(cyclopentyloxy)-4-methoxyphenyl)-
34. (4r)-4-[3-(cyclopentyloxy)-4 Methoxyphenyl)pyrrolidin-2-one
35. Rolipram [usan:inn]
36. [3h]rolipram
37. [3h]-rolipram
38. Sr-01000000162
39. Einecs 262-771-1
40. 4-(3-(cyclopentyloxy)-4-methoxyphenyl)-2-pyrrolidinon
41. Brn 1588548
42. Unii-k676nl63n7
43. (plusmn)-rolipram
44. Ncgc00016899-01
45. Cas-61413-54-5
46. Mfcd00270906
47. R-(-)-rolipram
48. Me-3167
49. Rolipram [usan]
50. Rolipram [inn]
51. Rolipram [jan]
52. Rolipram [mi]
53. Opera_id_1749
54. Prestwick0_000924
55. Prestwick1_000924
56. Prestwick2_000924
57. Prestwick3_000924
58. Rolipram [mart.]
59. Rolipram [who-dd]
60. Upcmld-dp110
61. Lopac0_001072
62. Schembl27930
63. Bspbio_000828
64. Bspbio_001356
65. Kbiogr_000076
66. Kbioss_000076
67. Rolipram (jan/usan/inn)
68. Mls000758273
69. Mls000759531
70. Mls001146912
71. Mls001424084
72. Mls006010272
73. Spbio_003007
74. (+)-rolipram; (s)-rolipram
75. Bpbio1_000912
76. Gtpl5260
77. Gtpl5313
78. 4-[3-(cyclopentoxy)-4-methoxy-phenyl]pyrrolidin-2-one
79. Dtxsid3044124
80. Upcmld-dp110:001
81. Upcmld-dp110:002
82. Bdbm14361
83. Kbio2_000076
84. Kbio2_002644
85. Kbio2_005212
86. Kbio3_000151
87. Kbio3_000152
88. Bio2_000076
89. Bio2_000556
90. Hms1361d18
91. Hms1570j10
92. Hms1791d18
93. Hms1989d18
94. Hms2051n15
95. Hms2090j16
96. Hms2097j10
97. Hms2232f06
98. Hms3263g06
99. Hms3267a05
100. Hms3369j08
101. Hms3393n15
102. Hms3402d18
103. Hms3411f08
104. Hms3654p06
105. Hms3655j21
106. Hms3675f08
107. Hms3714j10
108. Pharmakon1600-01505683
109. Bcp21910
110. Bcp23388
111. Tox21_110672
112. Tox21_501072
113. Nsc760125
114. Rolipram, Solid, >=98% (hplc)
115. S1430
116. Rolipram - Cas 61413-54-5
117. Akos015959793
118. Tox21_110672_1
119. Ccg-100965
120. Ccg-205149
121. Cs-1754
122. Db01954
123. Ks-1400
124. Lp01072
125. Nc00215
126. Nsc 760125
127. Sb12922
128. Sdccgsbi-0051042.p003
129. Idi1_033826
130. Ncgc00015898-03
131. Ncgc00015898-04
132. Ncgc00015898-05
133. Ncgc00015898-06
134. Ncgc00015898-07
135. Ncgc00015898-08
136. Ncgc00015898-09
137. Ncgc00015898-11
138. Ncgc00015898-12
139. Ncgc00015898-15
140. Ncgc00015898-31
141. Ncgc00089796-02
142. Ncgc00089796-04
143. Ncgc00089796-05
144. Ncgc00089796-06
145. Ncgc00089796-07
146. Ncgc00089796-08
147. Ncgc00261757-01
148. Ac-32927
149. Hy-16900
150. Sbi-0051042.p002
151. Db-080675
152. Ab00513966
153. Eu-0101072
154. Ft-0738515
155. R0110
156. Sw196583-4
157. D01783
158. R 6520
159. Ab00384360-17
160. Ab00384360_19
161. A847969
162. Q410285
163. 4-[3-(cyclopentyloxy)-4-methoxyphenyl]pyrrolidinone
164. Sr-01000000162-3
165. Sr-01000000162-4
166. Sr-01000000162-5
167. Sr-01000000162-9
168. 4-[3-cyclopentyloxy-4-methoxyphenyl]-2-pyrrolidinone
169. Brd-a34255068-001-04-8
170. Brd-a34255068-001-26-1
171. Sr-01000000162-12
172. 4-(3-cyclopentyloxy-4-methoxy-phenyl)pyrrolidin-2-one
173. 4-(3-cyclopentyloxy-4-methoxyphenyl)-pyrrolidin-2-one
174. 4-(3-cyclopentyloxy-4-methoxyphenyl)pyrrolidone
175. 4-[4-methoxy-3-(cyclopentyloxy)phenyl]-2-pyrrolidinone
176. 2-pyrrolidinone, 4-[3-(cyclopentyloxy)-4-methoxyphenyl]- (9ci)
| Molecular Weight | 275.34 g/mol |
|---|---|
| Molecular Formula | C16H21NO3 |
| XLogP3 | 2.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Exact Mass | 275.15214353 g/mol |
| Monoisotopic Mass | 275.15214353 g/mol |
| Topological Polar Surface Area | 47.6 Ų |
| Heavy Atom Count | 20 |
| Formal Charge | 0 |
| Complexity | 341 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Phosphodiesterase 4 Inhibitors
Compounds that specifically inhibit PHOSPHODIESTERASE 4. (See all compounds classified as Phosphodiesterase 4 Inhibitors.)
Antidepressive Agents
Mood-stimulating drugs used primarily in the treatment of affective disorders and related conditions. Several MONOAMINE OXIDASE INHIBITORS are useful as antidepressants apparently as a long-term consequence of their modulation of catecholamine levels. The tricyclic compounds useful as antidepressive agents (ANTIDEPRESSIVE AGENTS, TRICYCLIC) also appear to act through brain catecholamine systems. A third group (ANTIDEPRESSIVE AGENTS, SECOND-GENERATION) is a diverse group of drugs including some that act specifically on serotonergic systems. (See all compounds classified as Antidepressive Agents.)
ABOUT THIS PAGE
70
PharmaCompass offers a list of Rolipram API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Rolipram manufacturer or Rolipram supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Rolipram manufacturer or Rolipram supplier.
A Rolipram manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Rolipram, including repackagers and relabelers. The FDA regulates Rolipram manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Rolipram API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Rolipram supplier is an individual or a company that provides Rolipram active pharmaceutical ingredient (API) or Rolipram finished formulations upon request. The Rolipram suppliers may include Rolipram API manufacturers, exporters, distributors and traders.
Rolipram Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Rolipram GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Rolipram GMP manufacturer or Rolipram GMP API supplier for your needs.
A Rolipram CoA (Certificate of Analysis) is a formal document that attests to Rolipram's compliance with Rolipram specifications and serves as a tool for batch-level quality control.
Rolipram CoA mostly includes findings from lab analyses of a specific batch. For each Rolipram CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Rolipram may be tested according to a variety of international standards, such as European Pharmacopoeia (Rolipram EP), Rolipram JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Rolipram USP).

