
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
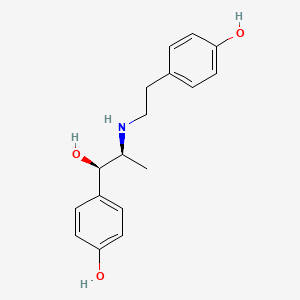

1. Du 21220
2. Du-21220
3. Du21220
4. Hydrochloride, Ritodrine
5. Pre Par
6. Pre-par
7. Prepar
8. Ritodrine Hydrochloride
9. Yutopar
1. Ritodrina [inn-spanish]
2. Ritodrinium [inn-latin]
3. 26652-09-5
4. Du-21220
5. Prepar
6. P-hydroxy-alpha-(1-((p-hydroxyphenethyl)amino)ethyl)benzyl Alcohol
7. Mls002153782
8. Ritodrine (usan/inn)
9. Smr001233166
10. (?)-ritodrine
11. Prestwick0_000349
12. Prestwick1_000349
13. Prestwick2_000349
14. Prestwick3_000349
15. Chembl785
16. 4-[(1r,2s)-1-hydroxy-2-{[2-(4-hydroxyphenyl)ethyl]amino}propyl]phenol
17. Schembl34194
18. Bspbio_000417
19. Cid_31728
20. Spbio_002338
21. Bpbio1_000459
22. Chebi:8872
23. Gtpl7294
24. Dtxsid7048534
25. Bdbm97162
26. Zinc57480
27. Db00867
28. Ncgc00179566-01
29. Ncgc00179566-03
30. C07239
31. D02359
32. Q5577596
33. Brd-k51465424-003-03-6
34. 4-(2-{[(1r,2s)-1-hydroxy-1-(4-hydroxyphenyl)propan-2-yl]amino}ethyl)phenol
| Molecular Weight | 287.35 g/mol |
|---|---|
| Molecular Formula | C17H21NO3 |
| XLogP3 | 2.3 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Exact Mass | 287.15214353 g/mol |
| Monoisotopic Mass | 287.15214353 g/mol |
| Topological Polar Surface Area | 72.7 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 284 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
For the treatment and prophylaxis of premature labour
Beta-2 adrenergic receptors are located at sympathetic neuroeffector junctions of many organs, including uterus. Ritodrine is beta-2 adrenergic agonist. It stimulates beta-2 adrenergic receptor, increases cAMP level and decreases intracellular calcium concentration. The decrease of calcium concentration leads to a relaxation of uterine smooth muscle and, therefore, a decrease in premature uterine contractions.
Sympathomimetics
Drugs that mimic the effects of stimulating postganglionic adrenergic sympathetic nerves. Included here are drugs that directly stimulate adrenergic receptors and drugs that act indirectly by provoking the release of adrenergic transmitters. (See all compounds classified as Sympathomimetics.)
Adrenergic beta-2 Receptor Agonists
Compounds bind to and activate ADRENERGIC BETA-2 RECEPTORS. (See all compounds classified as Adrenergic beta-2 Receptor Agonists.)
Tocolytic Agents
Drugs that prevent preterm labor and immature birth by suppressing uterine contractions (TOCOLYSIS). Agents used to delay premature uterine activity include magnesium sulfate, beta-mimetics, oxytocin antagonists, calcium channel inhibitors, and adrenergic beta-receptor agonists. The use of intravenous alcohol as a tocolytic is now obsolete. (See all compounds classified as Tocolytic Agents.)
G - Genito urinary system and sex hormones
G02 - Other gynecologicals
G02C - Other gynecologicals
G02CA - Sympathomimetics, labour repressants
G02CA01 - Ritodrine
Hepatic, by both the mother and fetus
1.7-2.6 hours
Ritodrine is beta-2 adrenergic agonist. It binds to beta-2 adrenergic receptors on outer membrane of myometrial cell, activates adenyl cyclase to increase the level of cAMP which decreases intracellular calcium and leads to a decrease of uterine contractions.
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 3490
Submission : 1979-03-06
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6264
Submission : 1986-04-14
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 4527
Submission : 1982-04-26
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]About the Company : In the dinamic pharmaceutical field, DEAFARMA is the reference point for primaries Pharmaceutical Laboratories for over twenty years, even in the national and international territo...

About the Company : Welcome To The Most Famous And Reputed Manufacturer And Supplier For Pharmaceutical Products, Chemicals, Pharmaceutical Package Group In China. FENGCHEN GROUP CO., LTD Have Been Se...

About the Company : Koa Shoji is top API import company in Japan. We have 23 years history in Japanese pharmaceutical field. Our corporate slogan is: 'To stably supply API with high quality at reasona...

About the Company : Suzhou Lixin Pharmaceutical Co., Ltd. has focused on developing high-quality APIs and advanced intermediates for global markets. Specializing in anti-tumor, cardiovascular, antivir...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Regulatory Info :
Registration Country : Italy
Brand Name : Miolene
Dosage Form : Ritodrine 50Mg 3 Units' Parenteral Use
Dosage Strength : 3 EV vials 50 mg 5 ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Regulatory Info :
Registration Country : Italy
Brand Name : Miolene
Dosage Form : Ritodrine 50Mg 3 Units' Parenteral Use
Dosage Strength : 3 EV vials 50 mg 5 ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
19
PharmaCompass offers a list of Ritodrine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ritodrine manufacturer or Ritodrine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ritodrine manufacturer or Ritodrine supplier.
A Ritodrine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ritodrine, including repackagers and relabelers. The FDA regulates Ritodrine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ritodrine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ritodrine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Ritodrine supplier is an individual or a company that provides Ritodrine active pharmaceutical ingredient (API) or Ritodrine finished formulations upon request. The Ritodrine suppliers may include Ritodrine API manufacturers, exporters, distributors and traders.
click here to find a list of Ritodrine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Ritodrine DMF (Drug Master File) is a document detailing the whole manufacturing process of Ritodrine active pharmaceutical ingredient (API) in detail. Different forms of Ritodrine DMFs exist exist since differing nations have different regulations, such as Ritodrine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Ritodrine DMF submitted to regulatory agencies in the US is known as a USDMF. Ritodrine USDMF includes data on Ritodrine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Ritodrine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Ritodrine suppliers with USDMF on PharmaCompass.
Ritodrine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ritodrine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ritodrine GMP manufacturer or Ritodrine GMP API supplier for your needs.
A Ritodrine CoA (Certificate of Analysis) is a formal document that attests to Ritodrine's compliance with Ritodrine specifications and serves as a tool for batch-level quality control.
Ritodrine CoA mostly includes findings from lab analyses of a specific batch. For each Ritodrine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ritodrine may be tested according to a variety of international standards, such as European Pharmacopoeia (Ritodrine EP), Ritodrine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ritodrine USP).

