
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
Canada
0
Australia
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Raunervil
2. Raupasil
3. Rausedil
4. Rausedyl
5. Serpasil
6. Serpivite
7. V Serp
8. V-serp
1. 50-55-5
2. Serpalan
3. Serpasil
4. Apoplon
5. Serpivite
6. Hypersil
7. Alserin
8. Sandril
9. Rau-sed
10. Elserpine
11. Hiserpia
12. Raunervil
13. Raupasil
14. Rausedil
15. Rausedyl
16. Rauwasedin
17. Reserpin
18. Serpanray
19. Resine
20. Rivasin
21. Serpate
22. Ascoserpina
23. Austrapine
24. Bioserpine
25. Enipresser
26. Eskaserpine
27. Helfoserpin
28. Hiposerpil
29. Maviserpin
30. Mayserpine
31. Mephaserpin
32. Purserpine
33. Rauserpine
34. Reserpamed
35. Reserpanca
36. Alkarau
37. Apsical
38. Banasil
39. Benazyl
40. Carditivo
41. Carrserp
42. Escaspere
43. Eserpine
44. Eskaserp
45. Hypersine
46. Idsoserp
47. Interpina
48. Lemiserp
49. Loweserp
50. Neoserfin
51. Neoserp
52. Quiescin
53. Raudiford
54. Raudixoid
55. Rauloydin
56. Raumorine
57. Raunorine
58. Raupoid
59. Raurine
60. Rausedan
61. Rauserpol
62. Rausingle
63. Rautrin
64. Rauvlid
65. Rauwilid
66. Rauwipur
67. Rauwoleaf
68. Rawilid
69. Resedrex
70. Reserbal
71. Resercaps
72. Resercen
73. Reserjen
74. Reserlor
75. Reserpal
76. Reserpene
77. Reserpex
78. Reserpil
79. Reserpina
80. Reserpoid
81. Respital
82. Restran
83. Riserpa
84. Roxinoid
85. Kitine
86. Raucap
87. Raugal
88. Raulen
89. Rausan
90. Reserp
91. (-)-reserpine
92. Key-serpine
93. 3p Reserp
94. Crystoserpine
95. Reserpinum
96. Neo-antitensol
97. Rauserpin-alk
98. Deserpine
99. Ryser
100. Reserfia
101. 3,4,5-trimethoxybenzoyl Methyl Reserpate
102. V-serp
103. Carpacil
104. Gilucard
105. Klimanosid
106. Resaltex
107. Resedril
108. Reserpidefe
109. Recipin
110. Serpine
111. Ent 50146
112. Raunova
113. Residin
114. Serpivate
115. Rese-lar
116. Reser-ar
117. Nci-c50157
118. H 520
119. Sedaraupina
120. Temposerpine
121. Eberpine
122. Raudixin
123. Reserpur
124. Residine
125. Resocalm
126. Resperine
127. Rezerpin
128. Sandron
129. Sedaraupin
130. Sedserp
131. Serfolia
132. Serolfia
133. Serpaloid
134. Serpasol
135. Serpazol
136. Serpena
137. Serpentil
138. Serpentina
139. Serpicon
140. Serpiloid
141. Serpogen
142. Serpoid
143. Serpone
144. Sertabs
145. Sertens
146. Sertina
147. Triserpin
148. Unilord
149. Serfin
150. Serpen
151. Roxel
152. Vio-serpine
153. Sk-reserpine
154. Renese R
155. T-serp
156. Nsc-59272
157. Chembl772
158. Nsc-237659
159. Broserpine
160. R-e-s
161. Sederaupin
162. Serpaneurona
163. Tefaserpina
164. Tenserpinie
165. Tepserpine
166. Eberspine
167. Reserpka
168. Resiatric
169. Resperin
170. Rivased
171. Rolserp
172. Roxynoid
173. Serpazil
174. Serpedin
175. Serpentin
176. Serpipur
177. Serpyrit
178. Sertensin
179. Serpil
180. Tempo-reserpina
181. 8b1qwr724a
182. 50-55-5 (free)
183. Mallopress
184. Rcra Waste Number U200
185. Hydropine
186. Hydroserp
187. Rauwita
188. .gamma.-serpine
189. Chebi:28487
190. Hydromox-r
191. Diurese-r
192. Nsc59272
193. Metatensin #2
194. Metatensin #4
195. Ent-50146
196. Component Of Naquival
197. Component Of Regroton
198. Hydropres 25
199. Hydropres 50
200. Hydrosine 25
201. (3beta,16beta,17alpha,18beta,20alpha)-11,17-dimethoxy-18-[(3,4,5-trimethoxybenzoyl)oxy]yohimban-16-carboxylic Acid Methyl Ester
202. Methyl (1s,2r,3r,4as,13br,14as)-2,11-dimethoxy-3-((3,4,5-trimethoxybenzoyl)oxy)-1,2,3,4,4a,5,7,8,13,13b,14,14a-dodecahydroindolo[2',3':3,4]pyrido[1,2-b]isoquinoline-1-carboxylate
203. Usaf Cb-27
204. Component Of Renese R
205. Component Of Metatensin
206. Ncgc00091250-04
207. Hydroserpalan
208. Diupres 250
209. Diupres 500
210. L-carpserp
211. Component Of Butiserpazide
212. Dsstox_cid_1237
213. Hydroserpine Plus
214. Hydroserpine #1
215. Dsstox_rid_76029
216. Dsstox_gsid_21237
217. Chloroserp-250
218. Chloroserp-500
219. Chloroserpin-250
220. Methyl (1r,15s,17r,18r,19s,20s)-6,18-dimethoxy-17-(3,4,5-trimethoxybenzoyl)oxy-1,3,11,12,14,15,16,17,18,19,20,21-dodecahydroyohimban-19-carboxylate
221. Methyl (3beta,16beta,17alpha,18beta,20alpha)-11,17-dimethoxy-18-[(3,4,5-trimethoxybenzoyl)oxy]yohimban-16-carboxylate
222. Yohimban-16-carboxylic Acid,11,17-dimethoxy-18-[(3,4,5-trimethoxybenzoyl)oxy]-, Methyl Ester,(3b,16b,17a,18b,20a)-
223. Serpasil Premix
224. Chloroserpine-500
225. Hydro-reserpine-25
226. Hydro-reserpine-50
227. Hydro-fluserpine #1
228. L -carpserp
229. Caswell No. 722a
230. Serp-afd
231. Reserpina [inn-spanish]
232. 3,5-trimethoxybenzoyl Methyl Reserpate
233. Yohimban-16-carboxylic Acid, 11,17-dimethoxy-18-((3,4,5-trimethoxybenzoyl)oxy)-, Methyl Ester, (3beta,16beta,17alpha,18beta,20alpha)-
234. (1s,2r,3r,4as,13br,14as)-methyl 2,11-dimethoxy-3-((3,4,5-trimethoxybenzoyl)oxy)-1,2,3,4,4a,5,7,8,13,13b,14,14a-dodecahydroindolo[2',3':3,4]pyrido[1,2-b]isoquinoline-1-carboxylate
235. Methyl (3beta,16beta,17alpha,18beta,20alpha)-11,17-bis(methyloxy)-18-({[3,4,5-tris(methyloxy)phenyl]carbonyl}oxy)yohimban-16-carboxylate
236. Smr000059122
237. Apoplon (tn)
238. Ccris 550
239. Serpine (pharmaceutical)
240. Hsdb 213
241. Methyl Reserpate 3,5-trimethyloxybenzoic Acid
242. 11,17-dimethoxy-18-((3,4,5-trimethoxybenzoyl)oxy)yohimban-16-carboxylic Acid Methyl Ester
243. Methyl Reserpate 3,5-trimethoxybenzoic Acid Ester
244. Einecs 200-047-9
245. Rcra Waste No. U200
246. Epa Pesticide Chemical Code 123101
247. Nsc 237659
248. Brn 0102014
249. Unii-8b1qwr724a
250. Ai3-50146
251. Wln: T F6 D5 C666 Em On&&tttj Ho1 Sovr Co1 Do1 Eo1& To1 Uvo1
252. Reserpine [usp:inn:ban:jan]
253. Cas-50-55-5
254. Reserpine,(s)
255. Serpalan (tn)
256. 3-.beta., 18-.beta.-hydroxy-11,17-.alpha.-dimethoxy-,methyl Ester, 3,4,5-trimethoxybenzoate (ester)
257. 3.beta., 18.beta.-hydroxy-11,17.alpha.-dimethoxy- Methyl Ester 3,4,5-trimethoxybenzoate (ester)
258. Benz[g]indolo[2, 1,2,3,4,4a,5,7,8,13,13b,14,14a-dodecahydro-3-hydroxy-2,11-dimethoxy-, Methyl Ester, 3,4,5-trimethoxybenzoate
259. Methyl (3beta,16beta,17alpha,18beta,20alpha)-11,17-dimethoxy-18-{[(3,4,5-trimethoxyphenyl)carbonyl]oxy}yohimban-16-carboxylate
260. Yohimban-16-carboxylic Acid,17-dimethoxy-18-[(3,4,5-trimethoxybenzoyl)oxy]-, Methyl Ester, (3.beta.,16.beta.,17.alpha.,18.beta.,20.alpha.)-
261. Prestwick_147
262. Methyl Reserpate 3,4,5-trimethoxybenzoic Acid Ester
263. Mfcd00005091
264. Reserpine, 99%
265. Spectrum_000109
266. Reserpic Acid Methyl Ester 3,4,5-trimethoxybenzoate (ester)
267. Reserpine [inn]
268. Reserpine [jan]
269. Reserpine [mi]
270. 79 More Names Available
271. Reserpine [hsdb]
272. Reserpine [iarc]
273. Prestwick0_000875
274. Prestwick1_000875
275. Prestwick2_000875
276. Prestwick3_000875
277. Spectrum3_000894
278. Spectrum4_000989
279. Spectrum5_001415
280. Reserpine [vandf]
281. Reserpinum [hpus]
282. Reserpine [mart.]
283. R 0875
284. Reserpine [usp-rs]
285. Reserpine [who-dd]
286. Reserpine [who-ip]
287. Schembl2589
288. Lopac0_000073
289. Bspbio_000949
290. Kbiogr_001397
291. Kbioss_000549
292. 4-25-00-01319 (beilstein Handbook Reference)
293. Methyl 18beta-hydroxy-11,17alpha-dimethoxy-3beta,20alpha-yohimban-16beta-carboxylate 3,4,5-trimethoxybenzoate (ester)
294. Methyl Dimethoxy-(3,4,5-trimethoxybenzoyl)oxy-[?]carboxylate
295. Mls002154046
296. Mls006011754
297. Divk1c_000012
298. Spectrum1500526
299. Spbio_002870
300. Reserpine (jp17/usp/inn)
301. Amy591
302. Bpbio1_001045
303. Gtpl4823
304. Megxp0_001904
305. Reserpine [orange Book]
306. Dtxsid7021237
307. Reserpine [ep Monograph]
308. Acon1_000086
309. Hms500a14
310. Kbio1_000012
311. Kbio2_000549
312. Kbio2_003117
313. Kbio2_005685
314. Kbio3_001808
315. Reserpine [usp Monograph]
316. Ninds_000012
317. Reserpinum [who-ip Latin]
318. Unipres Component Reserpine
319. Hms1570p11
320. Hms1920p04
321. Hms2092g05
322. Hms2097p11
323. Hms2234e24
324. Hms3260o07
325. Hms3413d20
326. Hms3677d20
327. Hms3714p11
328. Hms3884i21
329. Pharmakon1600-01500526
330. Dralserp Component Reserpine
331. Naquival Component Reserpine
332. Regroton Component Reserpine
333. Hy-n0480
334. Hydropres Component Reserpine
335. Renese-r Component Reserpine
336. Reserpine, Vetec(tm) Reagent Grade
337. Rkl10049
338. Zinc3938746
339. Hydrap-es Component Reserpine
340. Tox21_111107
341. Tox21_202395
342. Tox21_300537
343. Tox21_500073
344. Bbl028800
345. Bdbm50017712
346. Cam-ap-es Component Reserpine
347. H.r.-50 Component Reserpine
348. Metatensin Component Reserpine
349. Nsc237659
350. Nsc757309
351. Salutensin Component Reserpine
352. Ser-a-gen Component Reserpine
353. Ser-ap-es Component Reserpine
354. Stk801975
355. Hydromox R Component Reserpine
356. Reserpine 100 Microg/ml In Methanol
357. Reserpine Component Of Unipres
358. Akos000277559
359. Diutensen-r Component Reserpine
360. Reserpine Component Of Dralserp
361. Reserpine Component Of Naquival
362. Reserpine Component Of Regroton
363. Tox21_111107_1
364. Ccg-204168
365. Cs-1913
366. Db00206
367. Hydro-reserp Component Reserpine
368. Ks-5106
369. Lp00073
370. Nsc-757309
371. Reserpine Component Of Hydropres
372. Reserpine Component Of Renese-r
373. Sdccgsbi-0050061.p005
374. Demi-regroton Component Reserpine
375. Idi1_000012
376. Reserpine Component Of Cam-ap-es
377. Reserpine Component Of Hydrap-es
378. Reserpine Component Of Metatensin
379. Reserpine Component Of Salutensin
380. Reserpine Component Of Ser-a-gen
381. Reserpine Component Of Ser-ap-es
382. Ncgc00091250-01
383. Ncgc00091250-02
384. Ncgc00091250-03
385. Ncgc00091250-05
386. Ncgc00091250-06
387. Ncgc00091250-07
388. Ncgc00091250-08
389. Ncgc00091250-09
390. Ncgc00091250-10
391. Ncgc00091250-12
392. Ncgc00091250-14
393. Ncgc00091250-25
394. Ncgc00254489-01
395. Ncgc00259944-01
396. Ncgc00260758-01
397. Reserpine Component Of H.r.-50
398. Reserpine Component Of Hydromox R
399. 3-beta,20-alpha-yohimban-16-beta-carboxylic Acid, 18-beta-hydroxy-11,17-alpha-dimethoxy-, Methyl Ester, 3,4,5-trimethoxybenzoate (ester)
400. 3beta,20alpha-yohimban-16beta-carboxylic Acid, 18beta-hydroxy-11,17alpha-dimethoxy- Methyl Ester 3,4,5-trimethoxybenzoate (ester)
401. Ac-13142
402. Ac-34405
403. Br164333
404. Methyl (1r,15s,17r,18r,19s,20s)-6,18-dimethoxy-17-(3,4,5-trimethoxybenzoyloxy)-3,13-diazapentacyclo[11.8.0.0^{2,10}.0^{4,9}.0^{15,20}]henicosa-2(10),4,6,8-tetraene-19-carboxylate
405. Nci60_004446
406. Reserpine Component Of Diutensen-r
407. Reserpine Component Of Hydro-reserp
408. Sbi-0050061.p004
409. Serpasil-esidrix Component Reserpine
410. Reserpine Component Of Demi-regroton
411. Eu-0100073
412. Reserpine, Crystallized, >=99.0% (hplc)
413. Sw196458-3
414. Unm000011053801
415. Reserpine Component Of Serpasil-esidrix
416. Serpasil-apresoline Component Reserpine
417. C06539
418. D00197
419. Ab01562943_01
420. Reserpine Component Of Serpasil-apresoline
421. 005r091
422. A913268
423. Q407841
424. Q-100566
425. Brd-k95921201-001-07-0
426. Hydroserpine Plus (r-h-h) Component Reserpine
427. Methyl Reserpate; 3,4,5-trimethoxybenzoic Acid Ester
428. Reserpine, Certified Reference Material, Tracecert(r)
429. Reserpine Component Of Hydroserpine Plus (r-h-h)
430. Reserpine Solution, 1 Pg/mul In Methanol: Water (1:1)
431. Reserpine, European Pharmacopoeia (ep) Reference Standard
432. Reserpine Standard For Lc-ms, Analytical Standard, For Lc-ms
433. Reserpine, United States Pharmacopeia (usp) Reference Standard
434. Reserpine, Pharmaceutical Secondary Standard; Certified Reference Material
435. Hydralazine Hydrochloride-hydrochlorothiazide-reserpine Component Reserpine
436. Reserpine Component Of Hydralazine Hydrochloride-hydrochlorothiazide-reserpine
437. (1s,2r,3r,4as,13br,14as)-methyl2,11-dimethoxy-3-((3,4,5-trimethoxybenzoyl)oxy)-1,2,3,4,4a,5,7,8,13,13b,14,14a-dodecahydroindolo[2',3':3,4]pyrido[1,2-b]isoquinoline-1-carboxylate
438. (3?,16?,17?,18?,20?)-11,17-dimethoxy-18-[(3,4,5-trimethoxybenzoyl)oxy]yohimban-16-carboxylic Acid Methyl Ester
439. (3beta,16beta,17alpha,18beta,20alpha)-11,17-dimethoxy-18- [(3,4,5-trimethoxybenzoyl)oxy]yohimban-16-carboxyl Ic Acid Methyl Ester
440. Methyl (1r,15s,17r,18r,19s,20s)-6,18-dimethoxy-17-[(3,4,5-trimethoxyphenyl)carbonyloxy]-3,13-diazapentacyclo[11.8.0.0^{2,10}.0^{4,9}.0^{15,20}]henicosa-2(10),4,6,8-tetraene-19-carboxylate
441. Methyl (3.beta.,16.beta.,17.alpha.,18.beta.,20.alpha.)-11,17-dimethoxy-18-((3,4,5-trimethoxybenzoyl)oxy)yohimban-16-carboxylate
442. Methyl 18.beta.-hydroxy-11,17.alpha.-dimethoxy-3.beta.,20.alpha.-yohimban-16.beta.-carboxylate 3,4,5-trimethoxybenzoate (ester).
443. Yohimban-16-carboxylic Acid, 11,17-dimethoxy-18-(3,4,5-trimethoxybenzoyl)oxy-, Methyl Ester, (3.beta.,16.beta.,17.alpha.,18.beta.,20.alpha.)-
| Molecular Weight | 608.7 g/mol |
|---|---|
| Molecular Formula | C33H40N2O9 |
| XLogP3 | 4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 10 |
| Exact Mass | 608.27338086 g/mol |
| Monoisotopic Mass | 608.27338086 g/mol |
| Topological Polar Surface Area | 118 Ų |
| Heavy Atom Count | 44 |
| Formal Charge | 0 |
| Complexity | 1000 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Reserpine |
| PubMed Health | Reserpine (By mouth) |
| Drug Classes | Antihypertensive |
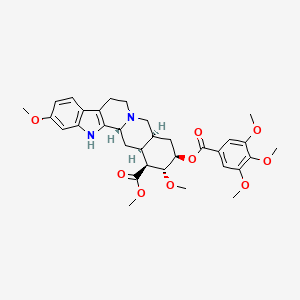
| Drug Label | Reserpine, USP is an antihypertensive, available as 0.1 mg and 0.25 mg tablets for oral administration. Its chemical name is methyl 18-hydroxy-11,17 -dimethoxy-3, 20-yohimban-16-carboxylate 3,4,5-trimethoxybenzoate (ester) and its structura... |
| Active Ingredient | Reserpine |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 0.25mg; 0.1mg |
| Market Status | Prescription |
| Company | Sandoz |
| 2 of 2 | |
|---|---|
| Drug Name | Reserpine |
| PubMed Health | Reserpine (By mouth) |
| Drug Classes | Antihypertensive |
| Drug Label | Reserpine, USP is an antihypertensive, available as 0.1 mg and 0.25 mg tablets for oral administration. Its chemical name is methyl 18-hydroxy-11,17 -dimethoxy-3, 20-yohimban-16-carboxylate 3,4,5-trimethoxybenzoate (ester) and its structura... |
| Active Ingredient | Reserpine |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 0.25mg; 0.1mg |
| Market Status | Prescription |
| Company | Sandoz |
Adrenergic Uptake Inhibitors; Antihypertensive Agents; Antipsychotic Agents; Sympatholytics
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
RESERPINE ... FOUND TO REDUCE HEART RATE, TREMOR, & STARE IN HYPERTHYROIDISM & RELIEVE PALPITATION, ANXIETY, & TENSION.
Gilman, A.G., L.S.Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 7th ed. New York: Macmillan Publishing Co., Inc., 1985., p. 209
ONLY IMPORTANT APPLICATION OF CARDIOVASCULAR EFFECTS OF RESERPINE IS IN TREATMENT OF HYPERTENSION ... IT IS OCCASIONALLY USED IN MANAGEMENT OF RAYNAUD'S SYNDROME.
Gilman, A.G., L.S.Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 7th ed. New York: Macmillan Publishing Co., Inc., 1985., p. 209
IV ... USEFUL IN ... SEVERE HYPERTENSION & HYPERTENSIVE EMERGENCIES.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 848
For more Therapeutic Uses (Complete) data for RESERPINE (18 total), please visit the HSDB record page.
... Should not be given to patients with a history of depression and, if depressive symptoms appear, the drug should be discontinued. Reserpine may incr gastric acid secretion and should be used cautiously in patients with history of peptic ulcers. If symptoms suggest recurrence of the ulcer, the drug should be discontinued.
Gilman, A.G., L.S.Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 7th ed. New York: Macmillan Publishing Co., Inc., 1985., p. 209
WHEN GIVEN PARENTERALLY FOR TREATMENT OF ECLAMPSIA, RESERPINE PASSES THROUGH PLACENTAL CIRCULATION & MAY CAUSE DROWSINESS, NASAL CONGESTION, CYANOSIS, & ANOREXIA IN NEWBORN INFANT. SODIUM & WATER RETENTION MAY OCCUR IF DIURETIC IS NOT GIVEN CONCOMITANTLY.
American Medical Association, AMA Department of Drugs. AMA Drug Evaluations. 5th ed. Chicago: American Medical Association, 1983., p. 714
BECAUSE OF SERIOUSNESS OF SIDE EFFECTS, RESERPINE IS NO LONGER MUCH USED AS TRANQUILIZER.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 848
... CONTRAINDICATED IN ULCERATIVE COLITIS BECAUSE OF INCR IN BOWEL MOBILITY.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 848
For more Drug Warnings (Complete) data for RESERPINE (23 total), please visit the HSDB record page.
For the treatment of hypertension
Reserpine is an adrenergic blocking agent used to treat mild to moderate hypertension via the disruption of norepinephrine vesicular storage. The antihypertensive actions of Reserpine are a result of its ability to deplete catecholamines from peripheral sympathetic nerve endings. These substances are normally involved in controlling heart rate, force of cardiac contraction and peripheral resistance.
Adrenergic Uptake Inhibitors
Drugs that block the transport of adrenergic transmitters into axon terminals or into storage vesicles within terminals. The tricyclic antidepressants (ANTIDEPRESSIVE AGENTS, TRICYCLIC) and amphetamines are among the therapeutically important drugs that may act via inhibition of adrenergic transport. Many of these drugs also block transport of serotonin. (See all compounds classified as Adrenergic Uptake Inhibitors.)
Antipsychotic Agents
Agents that control agitated psychotic behavior, alleviate acute psychotic states, reduce psychotic symptoms, and exert a quieting effect. They are used in SCHIZOPHRENIA; senile dementia; transient psychosis following surgery; or MYOCARDIAL INFARCTION; etc. These drugs are often referred to as neuroleptics alluding to the tendency to produce neurological side effects, but not all antipsychotics are likely to produce such effects. Many of these drugs may also be effective against nausea, emesis, and pruritus. (See all compounds classified as Antipsychotic Agents.)
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
C - Cardiovascular system
C02 - Antihypertensives
C02A - Antiadrenergic agents, centrally acting
C02AA - Rauwolfia alkaloids
C02AA02 - Reserpine
Route of Elimination
Reserpine is extensively metabolized to inactive compounds. It is slowly excreted via the urine and feces.
In man, after oral admin of 0.25 mg (3)H-reserpine, tritium was rapidly absorbed into the blood, reaching a peak within 1-2 hr. Radioactivity was tightly bound to red blood cells and remained constant over a 96 hr period. ... Six percent of the dose was excreted in the urine by 24 hr, mainly as trimethoxybenzoic acid; but radioactivity was still detectable in plasma, urine, and feces 11-12 days after drug admin.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V10 222 (1976)
PARENTERAL ADMIN OF RESERPINE PRODUCES GREATER CONCN IN NEONATAL RAT BRAIN THAN IN ADULT ANIMALS ... PARALLELED BY GREATER DEPLETION OF NOREPINEPHRINE IN INFANT THAN IN ADULT BRAIN. THIS MAY BE DUE TO LESSER CAPACITY OF RAT NEONATES TO METABOLIZE RESERPINE ... MIGHT ALSO EXPLAIN HIGHER PLASMA & TISSUE LEVELS IN HUMAN ADULTS.
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 680
RESERPINE LEAVES BLOOD WITHIN FEW MIN AFTER IV INJECTION & ACCUMULATES IN FATTY TISSUES ... MAX CONCN IN 4-6 HR. LIVER ALSO ACCUMULATES RESERPINE. MOST OF SINGLE DOSE HAS LEFT FAT & LIVER IN 48 HR. BRAIN RETAINS ... RESERPINE & ... METABOLITES ... 5 DAYS AFTER SINGLE DOSE.
Thienes, C., and T.J. Haley. Clinical Toxicology. 5th ed. Philadelphia: Lea and Febiger, 1972., p. 71
... CLAIMED TO BE ADEQUATELY ABSORBED FROM GI TRACT, BUT DIFFERENCE IN EFFICACY OF ORAL & IV DOSES RAISES DOUBTS ABOUT ADEQUACY OF ABSORPTION. ... HAS LONG LATENCY OF ONSET & PROLONGED DURATION OF ACTION.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 848
For more Absorption, Distribution and Excretion (Complete) data for RESERPINE (8 total), please visit the HSDB record page.
YIELDS 3,4,5-TRIMETHOXYBENZOIC ACID IN RAT, CAT, MOUSE;
Goodwin, B.L. Handbook of Intermediary Metabolism of Aromatic Compounds. New York: Wiley, 1976., p. R-1
YIELDS METHYL RESERPATE IN RAT, CAT;
Goodwin, B.L. Handbook of Intermediary Metabolism of Aromatic Compounds. New York: Wiley, 1976., p. R-1
IDENTIFIED METABOLIC PRODUCTS ARE RESERPIC ACID, SYRINGIC ACID, & SYRINGOYL METHYL RESERPATE.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 168
In rats, orally administered reserpine is rapidly hydrolysed to methyl reserpate; and in mice, orally or intravenously administered reserpine is metabolized to trimethoxybenzoic acid. In rats, methyl reserpate appears to be formed in the intestinal mucosa. Trimethoxybenzoic acid is rapidly eliminated in the urine of mice.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V24 220 (1980)
In man, after oral /dose/ of 0.25 mg (3)H-reserpine ... disappearance of radioactivity in plasma was biphasic: first component had half-life of 4.5 hr, and the second, 271 hr.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V10 222 (1976)
Reserpine's mechanism of action is through inhibition of the ATP/Mg2+ pump responsible for the sequestering of neurotransmitters into storage vesicles located in the presynaptic neuron. The neurotransmitters that are not sequestered in the storage vesicle are readily metabolized by monoamine oxidase (MAO) causing a reduction in catecholamines.
... DEPLETES OR RELEASES SEROTONIN (5-HYDROXYTRYPTAMINE), DOPAMINE ... NOREPINEPHRINE FROM ... BRAIN & ... BODY ... DEFICIENCY OF DOPAMINE IN BRAIN ... IN STRIATUM, SUBSTANTIA NIGRA ... PALLIDUM. ... HYPOTHERMIC EFFECT ... RELATED TO SEROTONIN DEPLETION OF HYPOTHALAMIC REGION. HYPOTHALMUS CONTAINS ... HIGHEST CONCN OF SEROTONIN. ...
Jones, L.M., et al. Veterinary Pharmacology & Therapeutics. 4th ed. Ames: Iowa State University Press, 1977., p. 395
ANTIHYPERTENSIVE ACTION ... DERIVES FROM ADRENERGIC NEURONAL BLOCKADE CONSEQUENT TO DEPLETION OF CATECHOLAMINE-CONTAINING GRANULES OF POSTGANGLIONIC SYMPATHETIC NEURON. MECHANISM OF CENTRAL EFFECTS IS UNKNOWN. IT DEPLETES BOTH BRAIN SEROTONIN & CATECHOLAMINES.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 848
... REPORTED TO INHIBIT GROWTH OF LEUKEMIA IN L1210 CELLS IN MALE MICE ... AND TO SUPPRESS GROWTH OF SARCOMA 37 IN MICE. ... RESERPINE DID NOT AFFECT GROWTH OF TRANSPLANTED MAMMARY ADENOCARCINOMAS IN C3H MICE. IT BLOCKS RELEASE OF PROLACTIN-INHIBITING FACTOR & THUS RAISES SERUM PROLACTIN LEVELS.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V10 222
Peripheral-Acting Adrenergic Antagonist /from table/
Ellenhorn, M.J., S. Schonwald, G. Ordog, J. Wasserberger. Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore, MD: Williams and Wilkins, 1997., p. 556
For more Mechanism of Action (Complete) data for RESERPINE (8 total), please visit the HSDB record page.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
31
PharmaCompass offers a list of Reserpine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Reserpine manufacturer or Reserpine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Reserpine manufacturer or Reserpine supplier.
A Reserpine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Reserpine, including repackagers and relabelers. The FDA regulates Reserpine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Reserpine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Reserpine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Reserpine supplier is an individual or a company that provides Reserpine active pharmaceutical ingredient (API) or Reserpine finished formulations upon request. The Reserpine suppliers may include Reserpine API manufacturers, exporters, distributors and traders.
click here to find a list of Reserpine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Reserpine DMF (Drug Master File) is a document detailing the whole manufacturing process of Reserpine active pharmaceutical ingredient (API) in detail. Different forms of Reserpine DMFs exist exist since differing nations have different regulations, such as Reserpine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Reserpine DMF submitted to regulatory agencies in the US is known as a USDMF. Reserpine USDMF includes data on Reserpine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Reserpine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Reserpine suppliers with USDMF on PharmaCompass.
A Reserpine written confirmation (Reserpine WC) is an official document issued by a regulatory agency to a Reserpine manufacturer, verifying that the manufacturing facility of a Reserpine active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Reserpine APIs or Reserpine finished pharmaceutical products to another nation, regulatory agencies frequently require a Reserpine WC (written confirmation) as part of the regulatory process.
click here to find a list of Reserpine suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Reserpine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Reserpine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Reserpine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Reserpine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Reserpine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Reserpine suppliers with NDC on PharmaCompass.
Reserpine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Reserpine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Reserpine GMP manufacturer or Reserpine GMP API supplier for your needs.
A Reserpine CoA (Certificate of Analysis) is a formal document that attests to Reserpine's compliance with Reserpine specifications and serves as a tool for batch-level quality control.
Reserpine CoA mostly includes findings from lab analyses of a specific batch. For each Reserpine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Reserpine may be tested according to a variety of international standards, such as European Pharmacopoeia (Reserpine EP), Reserpine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Reserpine USP).

