



API Suppliers

US DMFs Filed

CEP/COS Certifications
0

JDMFs Filed
0
Other Certificates
0
Other Suppliers
0
0

USA (Orange Book)
0

Europe
0

Canada
0

Australia
0

South Africa
0
Uploaded Dossiers
0
U.S. Medicaid
0
Annual Reports
0
0
USFDA Orange Book Patents
0
USFDA Exclusivities
0
Blog #PharmaFlow
0
News
0
EDQM
0
USP
0
JP
0
Other Listed Suppliers
0
0
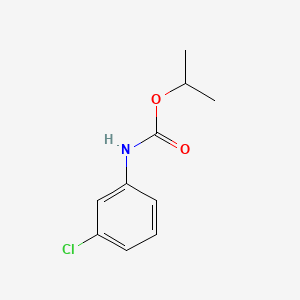

1. Cipc
2. Isopropyl N-(3-chlorophenol)carbamate
1. 101-21-3
2. Chloropropham
3. Cipc
4. Chloro-ipc
5. Isopropyl N-(3-chlorophenyl)carbamate
6. Chlorprophame
7. Elbanil
8. Metoxon
9. Nexoval
10. Preweed
11. Taterpex
12. Furloe
13. Isopropyl (3-chlorophenyl)carbamate
14. Isopropyl M-chlorocarbanilate
15. Stopgerme-s
16. Sprout Nip
17. Chloripc
18. Spud-nic
19. Spud-nie
20. Chloro-ifk
21. Triherbicide Cipc
22. Preventol 56
23. Prevenol 56
24. Liro Cipc
25. Isopropyl 3-chlorocarbanilate
26. Mirvale
27. Fasco Wy-hoe
28. Furloe 3 Ec
29. Keim-stop
30. Isopropyl 3-chlorophenylcarbamate
31. Isopropyl N-(m-chlorophenyl)carbamate
32. Ci-ipc
33. Chlor Ifc
34. Chlor Ifk
35. Carbamic Acid, (3-chlorophenyl)-, 1-methylethyl Ester
36. Cl-ifk
37. Jack Wilson Chloro 51(oil)
38. N-(3-chlorophenyl)isopropyl Carbamate
39. N-3-chlorophenylisopropylcarbamate
40. 3-chlorocarbanilic Acid, Isopropyl Ester
41. Isopropyl-m-chlorocarbanilate
42. Isopropyl-n-(3-chlorophenyl)carbamate
43. Isopropyl Meta-chlorocarbanilate
44. Ent 18,060
45. Isopropyl N-chlorophenylcarbamate
46. O-isopropyl N-(3-chlorophenyl)carbamate
47. N-(3-chlorophenyl)carbamic Acid, Isopropyl Ester
48. (3-chlorophenyl)carbamic Acid, 1-methylethyl Ester
49. Carbanilic Acid, M-chloro-, Isopropyl Ester
50. Isopropyl-n-m-chlorophenyl-carbamate
51. Isopropyl-n-3-chlorophenyl Carbamate
52. M-chlorocarbanilic Acid Isopropyl Ester
53. N-(3-chlor-phenyl)-isopropyl-carbamat
54. 1-methylethyl (3-chlorophenyl)carbamate
55. N-(3-chloor-fenyl)-isopropyl Carbamaat
56. N-(3-cloro-fenil)-isopropil-carbammato
57. M-chlorocarbanilic Acid, Isopropyl Ester
58. Isopropyl-n-(3-chlorphenyl)-carbamat
59. Propan-2-yl N-(3-chlorophenyl)carbamate
60. 0hbu04r8b0
61. Y 3
62. Chebi:34630
63. Nsc-29466
64. Chlorpropham 100 Microg/ml In Methanol
65. Chlorpropham 10 Microg/ml In Acetonitrile
66. N-(3-chloro Phenyl)carbamate D'isopropyle
67. Chlorpropham 100 Microg/ml In Acetonitrile
68. Dsstox_cid_764
69. Sprout-nip Ec
70. Unicrop Cipc
71. Bud Nip
72. Bud-nip
73. Chloro Ipc
74. Chlor Ipc
75. Chlor-ifc
76. Dsstox_rid_75778
77. Dsstox_gsid_20764
78. Furloe 3ec
79. Furloe 4ec
80. Caswell No. 510a
81. Chlorprophame [french]
82. Ci-ifk
83. Chlorpropham [bsi:iso]
84. Chlorprophame [iso-french]
85. Cas-101-21-3
86. Chlorpropham [iso]
87. Hsdb 981
88. Einecs 202-925-7
89. Nsc 29466
90. 1-methylethyl(3-chlorophenyl)carbamate
91. Epa Pesticide Chemical Code 018301
92. Brn 2211397
93. Unii-0hbu04r8b0
94. Ai3-18060
95. Ccris 9259
96. Isopropyl-n-(3-chlorphenyl)-carbamat [german]
97. N-(3-chloor-fenyl)-isopropyl Carbamaat [dutch]
98. N-(3-chlor-phenyl)-isopropyl-carbamat [german]
99. N-(3-cloro-fenil)-isopropil-carbammato [italian]
100. N-(3-chloro Phenyl) Carbamate D'isopropyle [french]
101. Propan-2-yl (3-chlorophenyl)carbamate
102. Isopropyl-n-(3-chlorophenyl) Carbamate
103. Prevenol (salt/mix)
104. Spectrum_001883
105. Gro Stop (salt/mix)
106. Specplus_000504
107. N-(3-chloro Phenyl) Carbamate D'isopropyle
108. Spectrum2_000044
109. Spectrum3_000842
110. Spectrum4_000682
111. Spectrum5_002000
112. Chlorpropham [mi]
113. Isopropyl Chlorocarbanilate
114. Chlorpropham [hsdb]
115. Schembl54464
116. Bspbio_002423
117. Kbiogr_001143
118. Kbioss_002413
119. Spectrum330051
120. Bidd:er0253
121. Divk1c_006600
122. Isopropyl M-chloro Carbanilate
123. Spbio_000167
124. Chembl104560
125. Dtxsid7020764
126. Schembl22437601
127. Kbio1_001544
128. Kbio2_002407
129. Kbio2_004975
130. Kbio2_007543
131. Kbio3_001923
132. Wln: Gr Cmvoy1 & 1
133. Zinc57368
134. Chlorpropham, ~95%, Crystalline
135. Nsc29466
136. Tox21_201866
137. Tox21_301139
138. Ccg-39995
139. Mfcd00037108
140. Akos003369049
141. Cs-w014956
142. Hy-w014240
143. Ks-5118
144. Ncgc00094538-01
145. Ncgc00094538-02
146. Ncgc00094538-03
147. Ncgc00094538-04
148. Ncgc00094538-05
149. Ncgc00094538-06
150. Ncgc00094538-07
151. Ncgc00255037-01
152. Ncgc00259415-01
153. Ac-11051
154. Chlorpropham 1000 Microg/ml In N-hexane
155. Nci60_002445
156. Ft-0631534
157. 01c213
158. D97609
159. Chlorpropham, Pestanal(r), Analytical Standard
160. N-(3-chlorophenyl)carbamic Acid Isopropyl Ester
161. 1-methylethyl N-(3-chlorophenyl)carbamate
162. A851706
163. Q904901
164. J-000342
165. Brd-k59590127-001-02-9
166. Carbamic Acid, N-(3-chlorophenyl)-, 1-methylethyl Ester
167. Chlorpropham Solution, 100 Mug/ml In Acetonitrile, Pestanal(r), Analytical Standard
| Molecular Weight | 213.66 g/mol |
|---|---|
| Molecular Formula | C10H12ClNO2 |
| XLogP3 | 3.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 3 |
| Exact Mass | 213.0556563 g/mol |
| Monoisotopic Mass | 213.0556563 g/mol |
| Topological Polar Surface Area | 38.3 Ų |
| Heavy Atom Count | 14 |
| Formal Charge | 0 |
| Complexity | 197 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Herbicides
Pesticides used to destroy unwanted vegetation, especially various types of weeds, grasses (POACEAE), and woody plants. Some plants develop HERBICIDE RESISTANCE. (See all compounds classified as Herbicides.)
Plant Growth Regulators
Any of the hormones produced naturally in plants and active in controlling growth and other functions. There are three primary classes: auxins, cytokinins, and gibberellins. (See all compounds classified as Plant Growth Regulators.)
After oral or ip administration of (14)C isopropyl and (14)C ring-labelled chloropropham to rats, 4 day urinary excretions were 50 and 85% of dose, respectively for two sites of labelling; in case of isopropyl-labelled compound, an additional 17-20% of dose was excreted as CO2 via lungs. ...When pregnant rats were given (14)C chloropropham, radioactivity was readily transferred to fetuses, and its level did not decline in fetal tissues as rapidly as it did in maternal organs. Pups of lactating rats that were given labelled chloropropham also contained radioactivity.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V12 61 (1976)
In Wistar rats given single oral dose of labeled chlorpropham average urinary excretions of radioactivity were 55.9% and 82.6% of chain (14)C CIPC and ring (14)C CIPC. With chain CIPC 35.4 + or - 7.5% of administered dose was excreted in respired air.
Fang SC et al; Pestic Biochem Physiol 4 (1): 1-11 (1974)
Dermal absorption is not significant.
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-327
Chlorpropham or its metabolites are readily transported acropetally following root absorption and may be transported basipetally from foliar application in some plants. ...Polar metabolites are not translocated once they are formed in either roots or shoots.
Weed Science Society of America. Herbicide Handbook. 5th ed. Champaign, Illinois: Weed Science Society of America, 1983., p. 104
For more Absorption, Distribution and Excretion (Complete) data for CHLORPROPHAM (7 total), please visit the HSDB record page.
In rats, most important metabolic transformation of chloropropham is hydroxylation in the para-position and conjugation of the resulting 4-hydroxychloropropham with sulfate. Hydroxylation of the isopropyl residue accounts for about 1/3 of the metabolism of this herbicide. ...Approximately 4 times more monohydroxy compound than dihydroxy compound were detected. Compound undergoes further oxidation... hydrolytic fission... yields meta-chloroaniline, carbon dioxide and isopropanol, which is further oxidized to acetone and carbon dioxide. Hydroxylation of... meta-chloroaniline also takes place to give N-acetyl-4-amino-2-chlorophenol... plus N-acetyl-2-amino-4-chlorophenol... .
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V12 62 (1976)
...It was... suggested that chlorpropham... metabolized differently between sensitive and resistant plants. ...Intact carbamates /found/ in sensitive plants... were not observed in resistant plant.
Kearney, P.C., and D. D. Kaufman (eds.) Herbicides: Chemistry, Degredation and Mode of Action. Volumes 1 and 2. 2nd ed. New York: Marcel Dekker, Inc., 1975., p. 654
Hydrolysis of carbamate linkage occurred in neomycin treated rats, and in vitro investigation suggested that liver is site of hydrolysis.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V12 61 (1976)
Following oral administration... to rats, renal excretion was followed. ...Although the expected hydrolysis products were not found free, n-acetyl hydroxyl analogs were excreted and identified. The 1-carbethoxy compound and a metabolite believed to be 1-hydroxypropyl-2-n-(3-chloro-4-hydroxyphenyl) carbamate was found... after root treatment of soybean plants with CIPC, polar metabolites of CIPC from root and shoot tissues were isolated and purified. Data showed major root metabolite was o-glucoside of 2-hydroxy CIPC. This was also found in shoots... .
Menzie, C. M. Metabolism of Pesticides, An Update. U.S. Department of the Interior, Fish, Wild-life Service, Special Scientific Report - Wildlife No. 184, Washington, DC: U.S. Government Printing Office, l974., p. 106
For more Metabolism/Metabolites (Complete) data for CHLORPROPHAM (15 total), please visit the HSDB record page.
...The average biological half-life of 14C from both compounds in most organs /of rats administered an oral dose of 14C-labelled propham or chloropropham/ was short, ranging from 3-8 hr. However, in brain, fat, and muscle, the half-life was about twice this value.
WHO; Environ Health Criteria 64: Carbamate Pesticides: A General Introduction (1986). Available from, as of February 17, 2005: https://www.inchem.org/documents/ehc/ehc/ehc64.htm


