


API Suppliers
0

US DMFs Filed
0

CEP/COS Certifications
0

JDMFs Filed
0
Other Certificates
0
Other Suppliers
0
0
0

USA (Orange Book)
0

Europe
0

Canada
0

Australia
0

South Africa
0
Uploaded Dossiers
0
U.S. Medicaid
0
Annual Reports
0
0
USFDA Orange Book Patents
0
USFDA Exclusivities
0
Blog #PharmaFlow
0
News
EDQM
0
USP
0
JP
0
Other Listed Suppliers
0
0
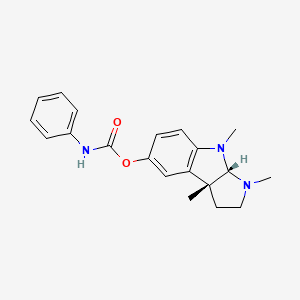

1. N-phenylcarbamoyl Eseroline
2. N-phenylcarbamoyleseroline
3. Phenserine
1. (+)-phenserine
2. R-phenserine
3. 116839-68-0
4. (+)-posiphen
5. Phenserine, (+)-
6. Buntanetap
7. Anvs401
8. Buntanetap [usan]
9. (+)-phenylcarbamoyleseroline
10. Anvs-401
11. Z0o4tj588o
12. (+)-o-(phenylcarbamoyl)eseroline
13. (3ar,8as)-1,3a,8-trimethyl-1,2,3,3a,8,8a-hexahydropyrrolo[2,3-b]indol-5-yl Phenylcarbamate
14. Pyrrolo(2,3-b)indol-5-ol, 1,2,3,3a,8,8a-hexahydro-1,3a,8-trimethyl-, 5-(n-phenylcarbamate), (3ar,8as)-
15. Unii-z0o4tj588o
16. Chembl4297417
17. Dtxsid001110145
18. Who 12342
19. Db15317
20. Hy-16009
21. Cs-0006062
22. [(3as,8br)-3,4,8b-trimethyl-2,3a-dihydro-1h-pyrrolo[2,3-b]indol-7-yl] N-phenylcarbamate
| Molecular Weight | 337.4 g/mol |
|---|---|
| Molecular Formula | C20H23N3O2 |
| XLogP3 | 2.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Exact Mass | 337.17902698 g/mol |
| Monoisotopic Mass | 337.17902698 g/mol |
| Topological Polar Surface Area | 44.8 Ų |
| Heavy Atom Count | 25 |
| Formal Charge | 0 |
| Complexity | 507 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Cholinesterase Inhibitors
Drugs that inhibit cholinesterases. The neurotransmitter ACETYLCHOLINE is rapidly hydrolyzed, and thereby inactivated, by cholinesterases. When cholinesterases are inhibited, the action of endogenously released acetylcholine at cholinergic synapses is potentiated. Cholinesterase inhibitors are widely used clinically for their potentiation of cholinergic inputs to the gastrointestinal tract and urinary bladder, the eye, and skeletal muscles; they are also used for their effects on the heart and the central nervous system. (See all compounds classified as Cholinesterase Inhibitors.)


