
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Europe
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Bloat Guard
2. Oxyethylene Oxypropylene Polymer
3. Oxypropylene Polymer, Oxyethylene
4. Poloxalene 2930
5. Polymer, Oxyethylene Oxypropylene
6. Polyoxyethylene Polyoxypropylene Polyoxyethylene Polymer
7. Proxanol
8. Sk And F 18667
9. Sk And F-18667
10. Sk And F18667
11. Tergitol
12. Therabloat
1. Poloxamer 188
2. 9003-11-6
3. Poloxamer 407
4. Pluronic F-68
5. 106392-12-5
6. 2-methyloxirane;oxirane
7. Tergitol(tm)xh(nonionic)
8. Pluronic L
9. Pluronic L 122
10. Polyethylene-polypropylene Glycol
11. Pluronic
12. Polyethylene Oxide-polypropylene Oxide
13. 691397-13-4
14. Poloxalkol
15. F-108
16. Poloxamer 331
17. Pluronic L 61
18. Pluronic L-81
19. Therabloat
20. Proxanol
21. Epan 485
22. Epan 710
23. Epan 785
24. Bloat Guard
25. Tergitol Xh
26. Oxirane, Methyl-, Polymer With Oxirane, Ether With 1,2-propanediol (2:1)
27. Pluronic L44
28. Pluronic F 38
29. Pluronic F 68
30. Pluronic F 108
31. Pluronic F 127
32. Pluronic L 101
33. Pluronic L 121
34. Pluronic L-101
35. 2-methyloxirane; Oxirane
36. Hydrowet
37. Polykol
38. Proksanol
39. Regulaid
40. Slovanik
41. Magcyl
42. Ethylene Oxide-propylene Oxide Block Polymer
43. Pluracol V
44. Pluronic F
45. Pluronic P
46. Monolan Pb
47. Pluriol Pe
48. Polyethylene Glycol, Propoxylated
49. Poloxalene L64
50. Glycols, Polyethylene-polypropylene
51. Poloxamer (nf)
52. Pluronic-68
53. Pluronic F86
54. Bsp 5000
55. Poloxamer 108
56. Poloxamer 182lf
57. Rokopol 16p
58. Rokopol 30p
59. Component Of Casakol
60. Pluronic 10r8
61. Pluronic 31r2
62. Pluronic F 68lf
63. Pluronic F 87
64. Pluronic F 88
65. Pluronic F 98
66. Pluronic L 24
67. Pluronic L 31
68. Pluronic L 35
69. Pluronic L 44
70. Pluronic L 62
71. Pluronic L 64
72. Pluronic L 68
73. Pluronic L 92
74. Pluronic L122
75. Pluronic P 75
76. Pluronic P 85
77. Pluronic P-65
78. Pluronic P-75
79. Propylen M 12
80. Proxanol 158
81. Proxanol 228
82. Slovanik 630
83. Slovanik 660
84. Supronic B 75
85. Rc 102
86. Wyandotte 7135
87. Emkalyx Ep 64
88. Emkalyx L101
89. Genapol Pf 10
90. Nixolen Sl 19
91. Rokopol 30p9
92. Tergitol Monionic Xh
93. Vepoloxamer (usan)
94. Pluronic C 121
95. Pluronic F 125
96. Pluronic P 104
97. Supronic E 400
98. Teric Pe40
99. Teric Pe60
100. Teric Pe70
101. Velvetol Oe 2nt1
102. Lutrol F (tn)
103. Newpol Pe-88
104. Nissan Pronon 201
105. Emkalyx L 101
106. Oxirane, Methyl-, Polymer With Oxirane
107. Plonon 201
108. Plonon 204
109. Pronon 102
110. Pronon 104
111. Pronon 201
112. Pronon 204
113. Pronon 208
114. Unilube 50mb26x
115. Polylon 13-5
116. Oxirane-propylene Oxide
117. Sk&f 18,667
118. Teric Pe 61
119. Teric Pe 62
120. Laprol 1502
121. Pluriol Pe 6810
122. Voranol P 2001
123. Berol Tvm 370
124. Peg-ppg-peg
125. Unilube 50mb168x
126. Monolan 8000e80
127. Niax Lg 56
128. Tergitol Xh (nonionic)
129. Thanol E 4003
130. Eban 710
131. Epan 750
132. Epon 420
133. Ppg Diol 3000eo
134. Synperonic Pe 30/40
135. Pluronic F87-a7850
136. Methyloxirane-oxirane Polymer
137. Niax 16-46
138. Oxirane-methyloxirane Polymer
139. Schembl11737
140. Pluronic L62(mw 2500)
141. Pluronic L64(mw 2900)
142. Ethylene Oxide Propylene Oxide
143. Oxirane, Polymer With Oxirane
144. Methyloxirane-oxirane Copolymer
145. Polyethylene-pluronic L-62lf
146. Propylene Oxide Ethylene Oxide
147. Tsl 431
148. Adeka Pluronic F-108
149. Polyoxyethylene Polyoxypropylene
150. Polyoxyethylene-polyoxypropylene
151. Oligoether L-1502-2-30
152. Chebi:32026
153. Polypropylene Glycol, Ethoxylated
154. Glycols, Polyethylenepolypropylene
155. Tvm 370
156. Lg 56
157. Oxirane, Polymer With Methyloxirane
158. Peg/ppg-24/34 Triblock Copolymer
159. Nsc63908
160. Poly(propylene Oxide-ethylene Oxide)
161. Polyoxyethylenated Poly(oxypropylene)
162. Nsc-63908
163. Ws 661
164. Poly(mixed Ethylene, Propylene)glycol
165. Propylene Oxide-ethylene Oxide Polymer
166. Akos015912614
167. Db11451
168. Polyoxyethylene-polyoxypropylene Polymer
169. Ethylene Glycol-propylene Glycol Polymer
170. Ethylene Oxide-propylene Oxide Copolymer
171. Propylene Oxide-ethylene Oxide Copolymer
172. Sk & F 18,667
173. Poly(oxyethylene)-poly(oxypropylene) Glycol
174. N 480
175. Poly(oxyethylene)-poly(oxypropylene) Polymer
176. Polypropylene Glycol-ethylene Oxide Copolymer
177. Propane-1,2-diol, Ethoxylated, Propoxylated
178. D01941
179. D10680
180. Polyethylene Oxide-polypropylene Oxide Copolymer
181. Polypropoxylated, Polyethoxylated Propylene Glycol
182. 1,2-propyleneglycol, Ethoxylated And Propoxylated
183. M 90/20
184. 75h90000
185. Propylene Glycol, Propylene Oxide, Ethylene Oxide Polymer
186. Oxirane, 2-methyl-, Polymer With Oxirane, Ether With 1,2-propanediol (2:1)
187. .alpha.-hydro-.omega.-hydroxypoly(oxyethylene)poly(oxypropylene)poly(oxyethylene) Block Copolymer
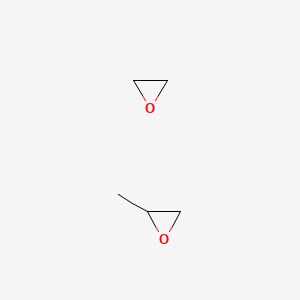
| Molecular Weight | 102.13 g/mol |
|---|---|
| Molecular Formula | C5H10O2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 102.068079557 g/mol |
| Monoisotopic Mass | 102.068079557 g/mol |
| Topological Polar Surface Area | 25.1 Ų |
| Heavy Atom Count | 7 |
| Formal Charge | 0 |
| Complexity | 36.7 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Mechanical cleansing of a wound with a sponge soaked in a surfactant has prevented the development of experimental wound infection. The surfactant utilized for wound cleansing is Pluronic F-68 (poloxalene; I), a member of a family of block copolymers called Pluronic polyls. Long term toxicity studies and clinical trials suggest that this surfactant is safe for human use. I is a nonionic detergent that does not have any intrinsic antibacterial activity. Although mechanical cleansing with saline-soaked sponges effectively removes bacteria, it damages the wound and impairs its resistance to infection. The severity of the damage to the skin exerted by the sponge can be correlated with its porosity. Sponges with a low porosity are abrasive and exert more damage to skin than do sponges with a higher porosity. The addition of I to even the most abrasive sponges ensures that the bacterial removal efficiency of the sponge scrub is maintained, while tissue trauma is minimized. This dual effect of the surfactant results in a dramatic reduction in the infection rate of contaminated wounds.
PMID:1119685 Rodeheaver GT et al; Am J Surg 129 (3): 241-5 (1975)
(VET): Prevention of bloat in cattle.
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. 13th Edition, Whitehouse Station, NJ: Merck and Co., Inc., 2001., p. 1357
A randomized, double blind, placebo controlled, dose escalation study of the toxicity and pharmacokinetics of poloxamer 188 (RheothRx) was conducted in 36 healthy male subjects (ages 19-35 yr) who received iv injections of 10-90 mg/kg/hr poloxamer or placebo. ... The most common adverse effects were pain, injection site abnormality, and nausea. Eight subjects discontinued treatment with poloxamer due to adverse events.
PMID:9232521 Jewell RC et al; J Pharm Sci 86 (7): 808-812 (1997)
Indicated to reduce viscosity in the blood before transfusions.
Poloxamer 188 (P188) exerts a protective action against oxidative stress and inflammation in tissue injury in various experimental models. In the rat model of excitotoxic injury, immediate intrathecal administration of P188 reduced neuronal loss, indicated by smaller spherical excitotoxic lesions. In a murine hind-limb model, P188 mediated a protective action against ischemia-reperfusion injury as indicated by decreased myocyte injury, preserved tissue adenosine 5'-triphosphate levels, and improved survival rates, suggesting that P188 can seal defects in cell membranes and attenuate damage induced by reactive oxygen species. P188 was shown to elicit protective effects against excitotoxic injury, and trauma-induced necrotic and apoptotic cell death in cultured neurons. In the mouse stroke models, P188 exerted a neuroprotective effect in brain ischemia-reperfusion induced acute injury by significantly reducing infarct volume and water content in brain edema and ameliorating the neurological symptoms 24 h after ischemia or reperfusion injury. P188 also significantly inhibited inflammatory, coagulation, and apoptotic responses resulting from superior mesenteric artery occlusion. In the experimental model of striatum injury in rats, P188 was shown to reduce excitotoxicity-induced tissue loss and macrophage infiltrate.
Disinfectants
Substances used on inanimate objects that destroy harmful microorganisms or inhibit their activity. Disinfectants are classed as complete, destroying SPORES as well as vegetative forms of microorganisms, or incomplete, destroying only vegetative forms of the organisms. They are distinguished from ANTISEPTICS, which are local anti-infective agents used on humans and other animals. (From Hawley's Condensed Chemical Dictionary, 11th ed) (See all compounds classified as Disinfectants.)
Surface-Active Agents
Agents that modify interfacial tension of water; usually substances that have one lipophilic and one hydrophilic group in the molecule; includes soaps, detergents, emulsifiers, dispersing and wetting agents, and several groups of antiseptics. (See all compounds classified as Surface-Active Agents.)
Cathartics
Agents that are used to stimulate evacuation of the bowels. (See all compounds classified as Cathartics.)
Absorption
Following a 48-hour continuous intravenous infusion of purified P188 in healthy volunteers, the mean concentration of P188 at steady state concentration (Css) was 522 118 mg/L and the maximum concentration occurring at the end of the loading dose was approximately 909 165 mg/L. The plasma concentrations were dose-proportional.
Route of Elimination
Renal clearance accounted for 90% of total plasma clearance in healthy male subjects.
Volume of Distribution
The volume of distribution at steady state (Vss) after a continuous intravenous infusion of 500 mg/kg of P188 on day 7 was approximately 2.13 mL/kg in pregnant female rats. Vss was 876 mL/kg in dogs receiving a dose of 720 mg/kg/day.
Clearance
Following a 48-hour continuous intravenous infusion of purified P188 in healthy volunteers, the mean total body clearance was estimated to be 4.40 0.77 L/h when using the plasma concentration data only. Estimated mean renal clearance from the amount of P-188 excreted in urine was 5.21 1.28 L/h. The clearance of a single metabolite HW1 was slower than the parent compound.
Absorption and excretion of 14C-Poloxalene 2930 (PX), a nonionic hydrophobic surfactant of large molecular weight, were studied using bile fistula rats. Approximately half of the dose infused intraduodenally was absorbed and some of the absorbed surfactant was excreted in bile. The remainder was excreted in urine. Only trace quantities of the 14C-PX were recovered in liver and carcass at termination of the study. Two studies were also performed with 14C-PX incorporated into the diet. In the first feeding study of 7 days duration, most of the agent was excreted via the gastrointestinal tract within 72 hr of discontinuing treatment. In the second study, rats were fed dietary 14C-PX for 7, 14, or 23 days to determine whether the surfactant continued to accumulate in the body as the test period was extended. Further accumulation did occur between the 7th and 14th days but not when feeding was continued for a total of 23 days. Of the amount of 14C-PX ingested after 23 days of feeding, essentially all was excreted by the end of 7 days after discontinuing treatment. These studies indicate that despite its large molecular weight of about 3,000 some 14C-PX is absorbed. Furthermore, absorbed material is promptly excreted in bile and urine with little retained in body tissues.
PMID:6149916 Rodgers JB et al; Drug Metab Dispos 12 (5): 631-4 (1984)
To investigate the distribution of poloxamer 407 (Pluronic F-127), rats received 300 mg of intraperitoneal poloxamer 407 solution; blood and urine samples were taken frequently and liver and kidney homogenates were prepared and analyzed using a colorimetry assay. Clearance was 0.014 mL/min. The mean amount excreted in the urine was 76.3 mg in 24 hr. Amounts of poloxamer 407 in supernatants of liver and kidney homogenates were 15.9 and 3.1 mg, respectively.
PMID:8738197 Li C et al; J Pharm Biomed Anal 14 (5): 659-65 (1996)
...The disposition and pharmacokinetics of Poloxamer 108 was studied in rats as an initial step towards understanding its behavior in man. After iv administration in rats, about 94% of 7 or 100 mg/kg doses of ethylene-14-C-labeled polymer was excreted in the urine in 3 days. About 6% of the label appeared in feces. Erythrocyte membranes were not permeable to the polymer, and only the parent compound was demonstrable in urine. Twenty hours after dosing, small residues were detectable only in the kidney, liver, small intestine and carcass. The 3rd phase of the plasma disappearance pattern was evident only at the larger dose, but plasma disappearance kinetics were independent of the dose in the used range. Most of Poloxamer 108 was eliminated rapidly in rats by renal excretion; a smaller portion probably was removed by biliary excretion.
PMID:1231 Jo Wang Z-Y et al; Drug Metab Dispos 3 (6): 536-42 (1975)
A randomized, double blind, placebo controlled, dose escalation study of the toxicity and pharmacokinetics of poloxamer 188 (RheothRx) was conducted in 36 healthy male subjects (ages 19-35 yr) who received iv injections of 10-90 mg/kg/hr poloxamer or placebo. Poloxamer was eliminated primarily by renal excretion. Estimates of clearance, elimination rate constant, and apparent volume of distribution at steady state were independent of infusion rate. Steady state plasma levels increased linearly with increasing infusion rate values up to 90 mg/kg/hr. Mean plasma clearance was 1.06 mL/min/kg. ...
PMID:9232521 Jewell RC et al; J Pharm Sci 86 (7): 808-812 (1997)
A single metabolite, HW1, with a molecular weight of approximately 16000 Daltons was detected in a pharmacokinetic study in dogs and man. HW1 was present in 10-20 and 40% of the parent compound at steady state in dogs and humans, respectively. However, it is suggested that block polymers are not metabolized and are excreted unchanged in the urine and feces, and HW1 may be a component of the higher molecular weight distribution of P188 that concentrates in the plasma due to its lower clearance rate.
In humans, P188 has half-life of 18 hours. The terminal plasma elimination half-life was approximately 7.65 1.10 hours in healthy volunteers receiving a 48-hour continuous intravenous infusion of purified P188.
P188 seals stable defects in cell membranes induced by skeletal muscle cell membranes rupture induced by ischemia-reperfusion injury, electroporation, irradiation, and heat damage. The full mechanism of action of P188 in inducing cytoprotective effects is not clear; however, based on _in vitro_ experiments and the structural similarity to plasmalemma, P188 may be directly incorporated into the phospholipid bilayer to attenuate the extent of tissue injury. Its high surface activity facilitates P188 to be inserted into lipid monolayers. P188 is proposed to exert localized actions by only interacting with damaged and compromised bilayers where the local lipid packing density is reduced. In addition to the direct interaction with the membrane, P188 was shown to inhibit MMP-9 protein levels and activity, as well as the NF-B signal pathway, in the model of acute cerebral ischemia, which is associated with increased BBB permeability leading to cerebral edema and increased penetration. MMP-9 is a key factor in extracellular matrix (ECM) degradation and BBB disruption.
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
47
PharmaCompass offers a list of Detalan API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Detalan manufacturer or Detalan supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Detalan manufacturer or Detalan supplier.
A POLOXAMER API manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of POLOXAMER API, including repackagers and relabelers. The FDA regulates POLOXAMER API manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. POLOXAMER API API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of POLOXAMER API manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A POLOXAMER API supplier is an individual or a company that provides POLOXAMER API active pharmaceutical ingredient (API) or POLOXAMER API finished formulations upon request. The POLOXAMER API suppliers may include POLOXAMER API API manufacturers, exporters, distributors and traders.
click here to find a list of POLOXAMER API suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A POLOXAMER API DMF (Drug Master File) is a document detailing the whole manufacturing process of POLOXAMER API active pharmaceutical ingredient (API) in detail. Different forms of POLOXAMER API DMFs exist exist since differing nations have different regulations, such as POLOXAMER API USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A POLOXAMER API DMF submitted to regulatory agencies in the US is known as a USDMF. POLOXAMER API USDMF includes data on POLOXAMER API's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The POLOXAMER API USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of POLOXAMER API suppliers with USDMF on PharmaCompass.
POLOXAMER API Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of POLOXAMER API GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right POLOXAMER API GMP manufacturer or POLOXAMER API GMP API supplier for your needs.
A POLOXAMER API CoA (Certificate of Analysis) is a formal document that attests to POLOXAMER API's compliance with POLOXAMER API specifications and serves as a tool for batch-level quality control.
POLOXAMER API CoA mostly includes findings from lab analyses of a specific batch. For each POLOXAMER API CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
POLOXAMER API may be tested according to a variety of international standards, such as European Pharmacopoeia (POLOXAMER API EP), POLOXAMER API JP (Japanese Pharmacopeia) and the US Pharmacopoeia (POLOXAMER API USP).

