



API Suppliers

US DMFs Filed

CEP/COS Certifications
0

JDMFs Filed
0
Other Certificates
Other Suppliers
0
0

USA (Orange Book)

Europe
0

Canada
0

Australia
0

South Africa
0
Uploaded Dossiers
0
U.S. Medicaid
Annual Reports
0
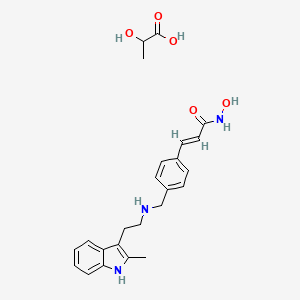

1. 960055-56-5
2. Panobinostat Lactate [jan]
3. Panobinostat (lactate)
4. Hn0t99oo4v
5. Panobinostat Lactate Anhydrous
6. Chebi:85991
7. 960055-56-5 (lactate)
8. (e)-n-hydroxy-3-(4-(((2-(2-methyl-1h-indol-3-yl)ethyl)amino)methyl)phenyl)acrylamide 2-hydroxypropanoate
9. Farydak (tn)
10. Panobinostat Lactate (jan)
11. (2e)-n-hydroxy-3-(4-(((2-(2-methyl-1h-indol-3-yl)ethyl)amino(methyl)phenyl)prop-2-enamide Mono((2rs)-2-hydroxypropanoate)
12. Propanoic Acid, 2-hydroxy-, Compd. With (2e)-n-hydroxy-3-(4-(((2-(2-methyl-1h-indol-3-yl)ethyl)amino)methyl)phenyl)-2-propenamide (1:1)
13. (2e)-n-hydroxy-3-[4-({[2-(2-methyl-1h-indol-3-yl)ethyl]amino}methyl)phenyl]acrylamide Lactic Acid
14. Unii-hn0t99oo4v
15. Propanoic Acid, 2-hydroxy-, Compd. With (2e)-n-hydroxy-3-[4-[[[2-(2-methyl-1h-indol-3-yl)ethyl]amino]methyl]phenyl]-2-propenamide (1:1)
16. Panobinostat Dl-lactate
17. Schembl2315056
18. Schembl2315067
19. Chembl3545368
20. Dtxsid401026247
21. Bcp25197
22. Ex-a2659
23. Hy-10224a
24. Panobinostat Lactate [who-dd]
25. Panobinostat Lactate [orange Book]
26. Cs-0030991
27. D10019
28. A917191
29. Q27158837
30. (2e)-n-hydroxy-3-[4-({[2-(2-methyl-1h-indol-3-yl)ethyl]amino}methyl)phenyl]prop-2-enamide; 2-hydroxypropanoic Acid
31. (e)-n-hydroxy-3-(4-(((2-(2-methyl-1h-indol-3-yl)ethyl)amino)methyl)phenyl)acrylamide2-hydroxypropanoate
32. (e)-n-hydroxy-3-[4-[[2-(2-methyl-1h-indol-3-yl)ethylamino]methyl]phenyl]prop-2-enamide;2-hydroxypropanoic Acid
33. 2-hydroxypropanoic Acid--(2e)-n-hydroxy-3-[4-({[2-(2-methyl-1h-indol-3-yl)ethyl]amino}methyl)phenyl]prop-2-enamide (1/1)
34. N-({4-[(1e)-3-(hydroxyamino)-3-oxoprop-1-en-1-yl]phenyl}methyl)-2-(2-methyl-1h-indol-3-yl)ethan-1-aminium 2-hydroxypropanoate
35. N-hydroxy-3-[4-[[[2-(2-methyl-1h-indol-3-yl) Ethyl]amino]methyl]phenyl]-2e-2-propenamide Dl-lactate Salt
36. N-hydroxy-3-[4-[[[2-(2-methyl-1h-indol-3-yl)ethyl]amino]methyl]phenyl]-2e-2-propenamide Dl-lactate Salt
37. N-hydroxy-3-[4-[[[2-(2-methyl-1h-indol-3-yl)ethyl]amino]methyl]phenyl]-2e-2-propenamide Lactate Salt
| Molecular Weight | 439.5 g/mol |
|---|---|
| Molecular Formula | C24H29N3O5 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 8 |
| Exact Mass | 439.21072103 g/mol |
| Monoisotopic Mass | 439.21072103 g/mol |
| Topological Polar Surface Area | 135 Ų |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 533 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Farydak, in combination with bortezomib and dexamethasone, is indicated for the treatment of adult patients with relapsed and/or refractory multiple myeloma who have received at least two prior regimens including bortezomib and an immunomodulatory agent.
Farydak, in combination with bortezomib and dexamethasone, is indicated for the treatment of adult patients with relapsed and/or refractory multiple myeloma who have received at least two prior regimens including bortezomib and an immunomodulatory agent.
L01XH03








