



API Suppliers

US DMFs Filed
0

CEP/COS Certifications
0

JDMFs Filed
0
Other Certificates
0
Other Suppliers
0

USA (Orange Book)
0

Europe
0

Canada
0

Australia
0

South Africa
0
Uploaded Dossiers
0
U.S. Medicaid
0
Annual Reports
0
0
USFDA Orange Book Patents
0
USFDA Exclusivities
0
Blog #PharmaFlow
0
News
EDQM
0
USP
0
JP
0
Other Listed Suppliers
0
0
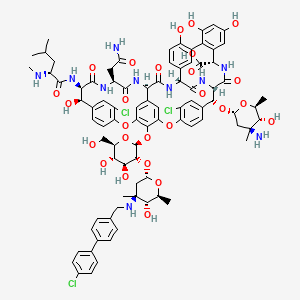

1. (4''r)-22-o-(3-amino-2,3,6-trideoxy-3-c-methyl-alpha-l-arabinohexopyranosyl)-n3''-(p-(p-chlorophenyl)benzyl)vancomycin
2. Ly 333328
3. Ly-333328
4. Ly333328
5. Orbactiv
1. 171099-57-3
2. Ly333328
3. Oritavancin [inn]
4. Ly-333328
5. Chlorobiphenyl-chloroeremomycin
6. Pug62frz2e
7. Chebi:82699
8. Kimyrsa
9. (4''r)-22-o-(3-amino-2,3,6-trideoxy-3-c-methyl-alpha-l-arabinohexopyranosyl)-n3''-(p-(p-chlorophenyl)benzyl)vancomycin
10. Unii-pug62frz2e
11. Chembl1688530
12. Orita-vancin
13. Ly 333328
14. C12034
15. Oritavancin [mi]
16. Oritavancin [vandf]
17. Oritavancin [mart.]
18. Oritavancin [who-dd]
19. Oritavancin; Ly-333328
20. Schembl9947049
21. Gtpl10877
22. Dtxsid20897570
23. Bdbm513037
24. Ex-a2372
25. Db04911
26. Ncgc00485478-01
27. Ncgc00485478-02
28. Q7102878
29. (4''r)-22-o-(3-amino-2,3,6-trideoxy-3-c-methyl-.alpha.-l-arabino-hexopyranosyl)-n(sup 3)''-(p-(p-chlorophenyl)benzyl)vancomycin
30. (4''r)-22-o-(3-amino-2,3,6-trideoxy-3-c-methyl-alpha-l-arabino-hexopyranosyl)-n(3'')-((4'-chloro(1,1'-biphenyl)-4-yl)methyl)vancomycin
31. (4'r)-22-o-(3-amino-2,3,6-trideoxy-3-c-methyl-alpha-l-arabino-hexopyranosyl)-n(sup 3)' -(p-(p-chlorophenyl)benzyl)vancomycin
32. Vancomycin, 22-o-(3-amino-2,3,6-trideoxy-3-c-methyl-.alpha.-l-arabino-hexopyranosyl)-n3''-((4'-chloro(1,1'-biphenyl)-4-yl)methyl)-, (4''r)-
| Molecular Weight | 1793.1 g/mol |
|---|---|
| Molecular Formula | C86H97Cl3N10O26 |
| XLogP3 | 1.5 |
| Hydrogen Bond Donor Count | 20 |
| Hydrogen Bond Acceptor Count | 29 |
| Rotatable Bond Count | 19 |
| Exact Mass | 1790.564106 g/mol |
| Monoisotopic Mass | 1790.564106 g/mol |
| Topological Polar Surface Area | 561 Ų |
| Heavy Atom Count | 125 |
| Formal Charge | 0 |
| Complexity | 3700 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 22 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Oritavancin is indicated for the treatment of adult patients with acute bacterial skin and skin structure (including subcutaneous) infection. It is used for confirmed/suspected infections with designated and susceptible gram-positive organisms. There are two preparations of oritavancin; the 400 mg dose that is administered over 3 hours, and the 1200 mg dose administered over 1 hour. Both are indicated for susceptible gram-positive skin and skin structure infections in adults. As antimicrobial susceptibility patterns are geographically distinct, local antibiograms should be consulted to ensure adequate coverage of relevant pathogens prior to use.
FDA Label
Tenkasi is indicated for the treatment of acute bacterial skin and skin structure infections (ABSSSI) in adults (see sections 4. 4 and 5. 1).
Consideration should be given to official guidance on the appropriate use of antibacterial agents.
Oritavancin interferes with bacterial cell wall synthesis and integrity, treating susceptible skin and subcutaneous tissue infections with gram-positive bacteria. This drug is known to artifically increase INR and aPTT, interfering with coagulation testing. Cases of infusion reactions have also been reported.
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
J01XA05
J - Antiinfectives for systemic use
J01 - Antibacterials for systemic use
J01X - Other antibacterials
J01XA - Glycopeptide antibacterials
J01XA05 - Oritavancin
Absorption
Pharmacokinetic analysis of oritavancin revealed a Cmax of 138 and g/mL and an AUC0- of 2800 gh/mL. The AUC0-t in a study of healthy volunteers after an 800 mg dose 1,1111 gh/mL. was also be Another pharmacokinetic study reported a Cmax of 4.7-7.6 micrograms/mL, generally achieved within 24 hours of administration.
Route of Elimination
Oritavancin is excreted as unchanged drug in both the urine and feces. Less than 5% has been recovered in the urine, and 1% has been recovered in the feces.
Volume of Distribution
The volume of distribution of oritavancin is estimated at 87.6 L, suggesting extensive tissue distribution.
Clearance
The clearance of oritavancin is approximately 0.445 L/h. One study revealed a renal clearance of 0.457 mL/min.
In vitro studies on human hepatocytes suggest that oritavancin is not metabolized, and is excreted unchanged.
The average terminal half-life of oritavancin is about 245 hours. A pharmacokinetic study revealed a terminal half-life ranging from 135.8-273.8 hours.
The cell wall is vital for the survival and replication of bacteria, making it a primary target for antibiotic therapy. Oritavancin works against susceptible gram-positive organisms via three separate mechanisms. Firstly, it binds to the stem peptide of peptidoglycan precursors, inhibiting transglycosylation (polymerization). This process normally occurs during cell wall synthesis. Secondly, oritavancin inhibits crosslinking during bacterial cell wall biosynthesis via binding to cell wall pentaglycyl peptide bridging segments. Finally, this drug also acts by disrupting the bacterial cell membrane, interfering with its integrity, which eventually leads to cell death by various mechanisms.


