
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
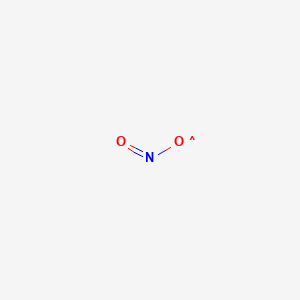

1. 10102-44-0
2. Nitrogen Dioxide, >=99.5%
3. Dtxsid7020974
4. Mfcd00085341
| Molecular Weight | 46.006 g/mol |
|---|---|
| Molecular Formula | NO2 |
| XLogP3 | -0.3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 45.992903243 g/mol |
| Monoisotopic Mass | 45.992903243 g/mol |
| Topological Polar Surface Area | 30.4 Ų |
| Heavy Atom Count | 3 |
| Formal Charge | 0 |
| Complexity | 7.5 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Several reports suggest that nitrogen dioxide is absorbed in lungs and reacts with blood ... Nitrous fumes ... act chiefly on alveolar walls.
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. III-321
50 to 60% of inspired nitrogen dioxide in rhesus monkeys was retained during quiet resp, the gas was distributed throughout lungs. Once absorbed, nitrogen dioxide or chem intermediates remained within lungs for prolonged periods after cessation of exposure, & dissemination to extrapulmonary sites occurred via blood stream.
PMID:402875 GOLDSTEIN E ET AL; AM REV RESPIR DIS 115 (3): 403-12 (1977)
... Found that about 50% of nitrogen dioxide was absorbed in passage through the nose and throat of a rabbit. ... 50 to 60% of nitrogen dioxide was absorbed by monkeys exposed to 0.3 to 0.9 ppm of nitrogen dioxide. The radioactive tracer ... used remained in the lung for prolonged periods, with some spread to other tissues.
Clayton, G. D. and F. E. Clayton (eds.). Patty's Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 4099
Nitrogen dioxide is partly dissolved in the mucus of the upper airways, but it is also fairly evenly distributed throughout the respiratory tract; it is furthur spread in the body by the circulatory system.
Linvall T; J Work Environ Health 11 (3): 10-28 (1985)
For more Absorption, Distribution and Excretion (Complete) data for Nitrogen dioxide (7 total), please visit the HSDB record page.
Inhalation of nitrogen dioxide (NO2) by mice administered orally amidopyrine (AP) and sodium nitrite resulted in increased biosynthesis of N-nitrosodimethylamine (NDMA), as determined by analysis using gas chromatography with thermal energy analyzer detector. These results were also confirmed indirectly in chronic experiments on rats using the system of biomarkers of NDMA formation (single-stranded DNA liver damages, alanine-aminotransferase, glutathione-S-transferase, and liver S9 fraction activity). The inhibition of NDMA metabolism by 4-methylpyrazol (4-MP) administration increases the sensitivity of NDMA biosynthesis assay in frozen whole-mouse powder. The results confirm that NO2 can serve as the precursor of nitrosamines.
PMID:9372840 Rubenchik BL et al; J Environ Pathol Toxicol Oncol 14 (2): 111-5 (1995)
Sprague-Dawley rats exposed to atmospheres containing low levels of nitrogen dioxide for 24 hr had increased levels of nitrate in their urine on the day of exposure & on the 3 subsequent days. Authors findings support the hypothesis that the major interaction of nitrogen dioxide in the lung is with readily oxidizable tissue components to form nitrite, & they estimate that 9.6 umol of nitrite is formed in the respiratory tract per ppm nitrogen dioxide 24 hr exposure.
PMID:6836583 SAUL RL, ARCHER MC; TOXICOL APPL PHARMACOL 67 (2): 284-91 (1983)
Nitrates and nitrites have been detected in the urine of animals after the inhalation of nitrogen dioxide.
Clayton, G. D. and F. E. Clayton (eds.). Patty's Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 4099
The lung can be exposed to a variety of reactive nitrogen intermediates through the inhalation of environmental oxidants and those produced during inflammation. Reactive nitrogen species (RNS) include, nitrogen dioxide (.NO2) and peroxynitrite (ONOO-). Classically known as a major component of both indoor and outdoor air pollution, .NO2 is a toxic free radical gas. .NO2 can also be formed during inflammation by the decomposition of ONOO- or through peroxidase-catalyzed reactions. Due to their reactive nature, RNS may play an important role in disease pathology. Depending on the dose and the duration of administration, .NO, has been documented to cause pulmonary injury in both animal and human studies. Injury to the lung epithelial cells following exposure to .NO2 is characterized by airway denudation followed by compensatory proliferation. The persistent injury and repair process may contribute to airway remodeling, including the development of fibrosis. To better understand the signaling pathways involved in epithelial cell death by .NO2 or otherRNS, ... cells /are exposed/ in culture to continuous gas-phase .NO2. Studies using the .NO2 exposure system revealed that lung epithelial cell death occurs in a density dependent manner. In wound healing experiments, .NO2 induced cell death is limited to cells localized in the leading edge of the wound. Importantly, .NO2-induced death does not appear to be dependent on oxidative stress per se. Potential cell signaling mechanisms ... include the mitogen activated protein kinase, c-Jun N-terminal Kinase and the Fas/Fas ligand pathways. During periods of epithelial loss and regeneration that occur in diseases such as asthma or during lung development, epithelial cells in the lung may be uniquely susceptible to death...
PMID:12162462 Persinger RL et al; Mol Cellular Biochem 234-235 (1-2): 71-80 (2002)
The biochemical mechanisms of nitrogen dioxide cellular injury ... One emphasizes lipid peroxidation, the other the oxidation of low molecular wt reducing substances & proteins. These need not be mutually exclusive. ... Nitrogen dioxide ... Initiate oxidation through free radicals ... Induction period ... Can be prolonged by vitamin E or other free radical scavenging agents. Polar nitrogen containing cmpd & peroxides are produced.
Clayton, G. D. and F. E. Clayton (eds.). Patty's Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 4102
ABOUT THIS PAGE
12
PharmaCompass offers a list of Nitrogen Dioxide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Nitrogen Dioxide manufacturer or Nitrogen Dioxide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Nitrogen Dioxide manufacturer or Nitrogen Dioxide supplier.
A Nitrogen Dioxide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Nitrogen Dioxide, including repackagers and relabelers. The FDA regulates Nitrogen Dioxide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Nitrogen Dioxide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Nitrogen Dioxide supplier is an individual or a company that provides Nitrogen Dioxide active pharmaceutical ingredient (API) or Nitrogen Dioxide finished formulations upon request. The Nitrogen Dioxide suppliers may include Nitrogen Dioxide API manufacturers, exporters, distributors and traders.
click here to find a list of Nitrogen Dioxide suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Nitrogen Dioxide DMF (Drug Master File) is a document detailing the whole manufacturing process of Nitrogen Dioxide active pharmaceutical ingredient (API) in detail. Different forms of Nitrogen Dioxide DMFs exist exist since differing nations have different regulations, such as Nitrogen Dioxide USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Nitrogen Dioxide DMF submitted to regulatory agencies in the US is known as a USDMF. Nitrogen Dioxide USDMF includes data on Nitrogen Dioxide's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Nitrogen Dioxide USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Nitrogen Dioxide suppliers with USDMF on PharmaCompass.
Nitrogen Dioxide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Nitrogen Dioxide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Nitrogen Dioxide GMP manufacturer or Nitrogen Dioxide GMP API supplier for your needs.
A Nitrogen Dioxide CoA (Certificate of Analysis) is a formal document that attests to Nitrogen Dioxide's compliance with Nitrogen Dioxide specifications and serves as a tool for batch-level quality control.
Nitrogen Dioxide CoA mostly includes findings from lab analyses of a specific batch. For each Nitrogen Dioxide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Nitrogen Dioxide may be tested according to a variety of international standards, such as European Pharmacopoeia (Nitrogen Dioxide EP), Nitrogen Dioxide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Nitrogen Dioxide USP).

