
Synopsis
Synopsis
0
JDMF
0
KDMF
0
VMF
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
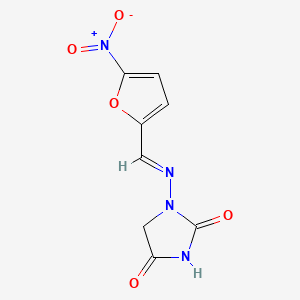

1. Furadantin
2. Furadantine
3. Furadoine
4. Furadonine
5. Furantoin
6. Macrodantin
7. Nitrofurantoin Sodium Salt
8. Nitrofurantoin, Monohydrate
1. 67-20-9
2. Macrodantin
3. Furadantin
4. 5-nitrofurantoin
5. Furadonine
6. Furadantine
7. Furadantoin
8. Furadoine
9. Furadontin
10. Furantoin
11. Nifurantin
12. Furalan
13. Nitrofuradantin
14. Nitrofurantoine
15. Berkfurin
16. Chemiofuran
17. Furachel
18. Furadonin
19. Furatoin
20. Furobactina
21. Macrobid
22. Novofuran
23. Orafuran
24. Parfuran
25. Trantoin
26. Urantoin
27. Urofurin
28. Welfurin
29. Zoofurin
30. Cyantin
31. Cystit
32. Furina
33. Ituran
34. Nitoin
35. Urizept
36. Urodin
37. Urolong
38. Furadantine Mc
39. Uro-tablinen
40. Fur-ren
41. Macrofuran
42. Nitrofurantoina
43. Benkfuran
44. Dantafur
45. Furaloid
46. Nierofu
47. Macpac
48. Nitrex
49. Nitrofurantoinum
50. Fua Med
51. N-toin
52. Usaf Ea-2
53. 1-(5-nitro-2-furfurylideneamino)hydantoin
54. 1-((5-nitrofurfurylidene)amino)hydantoin
55. N-(5-nitrofurfurylidene)-1-aminohydantoin
56. Nitrofurantoin, Macrocrystalline
57. 1-[(5-nitrofurfurylidene)amino]hydantoin
58. 1-(5-nitro-2-furfurylidenamino)hydantoin
59. N-(5-nitro-2-furfurylidene)-1-aminohydantoin
60. Nitrofurantoin Anhydrous
61. Nci-c55196
62. Nsc 2107
63. Nitrofurantoin Macrocrystalline
64. Nsc 44150
65. N-(5-nitro-2-furfurylideno)-1-aminohydantoina
66. Nsc-2107
67. Furadoninum
68. Nitrofurantoin Macrocrystal
69. 1-(((5-nitrofuran-2-yl)methylene)amino)imidazolidine-2,4-dione
70. 2,4-imidazolidinedione, 1-[[(5-nitro-2-furanyl)methylene]amino]-
71. 1-[(e)-(5-nitrofuran-2-yl)methylideneamino]imidazolidine-2,4-dione
72. Nitrofurantoin, Macrocrystals
73. Mls000028500
74. Cistofuran
75. Macrodantina
76. Macrofurin
77. Nifuretten
78. Alfuran
79. Berkfuran
80. Ceduran
81. Furabid
82. Furedan
83. Gerofuran
84. Phenurin
85. Siraliden
86. Uerineks
87. Urofuran
88. Urolisa
89. Chebi:71415
90. Hydantoin, 1-((5-nitrofurfurylidene)amino)-
91. Piyeloseptyl
92. 1-[[(5-nitro-2-furanyl)methylene]amino]-2,4-imidazolidinedione
93. Furadantina Mc
94. Furadantine-mc
95. 2,4-imidazolidinedione, 1-(((5-nitro-2-furanyl)methylene)amino)-
96. Furophen T
97. Nitrofur-c
98. Ro-antoin
99. Furadantin Retard
100. Uro-selz
101. Ivadantin
102. 927ah8112l
103. Smr000058271
104. 1-(((5-nitrofuran-2-yl)methylene)-amino)imidazolidine-2,4-dione
105. 1-{[(1e)-(5-nitrofuran-2-yl)methylidene]amino}imidazolidine-2,4-dione
106. Nitrofurantoina [dcit]
107. Nitrofurantoine [inn-french]
108. Nitrofurantoinum [inn-latin]
109. Furodantin
110. 1-((5-nitro-2-furanyl)methylene)amino-2,4-imidazolidenedione
111. Fuamed
112. 1-{[(e)-(5-nitrofuran-2-yl)methylidene]amino}imidazolidine-2,4-dione
113. Nsc-44150
114. Furadantin (tn)
115. Macrobid (tn)
116. 178170-37-1
117. Ccris 1192
118. Hydantoin, 1-[(5-nitrofurfurylidene)amino]-
119. Hsdb 3135
120. Sr-05000001681
121. Einecs 200-646-5
122. Nsc2107
123. Furantoina
124. Furadoin
125. Uvamin
126. N-(5-nitro-2-furfurylideno)-1-aminohydantoina [polish]
127. Nsc44150
128. Ai3-26388
129. (e)-1-(((5-nitrofuran-2-yl)methylene)amino)imidazolidine-2,4-dione
130. Nitrofurantoin (jan/usp/inn)
131. Unii-927ah8112l
132. Furophen T-caps
133. Ncgc00091505-01
134. Ncgc00091505-07
135. Prestwick_358
136. Nitrofurantoin Macro
137. 5-nitrofurantoindorn
138. J01xe01
139. Nitrofurantoin [usp:inn:ban:jan]
140. Mfcd00003224
141. Nd-3320
142. Nd-7248
143. Nitrofurantoin, 97%
144. Nitrofurantoinum Anhydrous
145. Prestwick2_000168
146. Prestwick3_000168
147. Spectrum5_001367
148. Nitrofurantoin, Crystalline
149. Chembl572
150. Nitrofurantoin [mi]
151. Nitrofurantoin Macrocrystals
152. Cid_4509
153. Nitrofurantoin [inn]
154. Nitrofurantoin [jan]
155. Schembl29470
156. Schembl29472
157. Bspbio_000035
158. Bspbio_002073
159. Nitrofurantoin [hsdb]
160. Nitrofurantoin [iarc]
161. Bidd:gt0181
162. Nitrofurantoin [vandf]
163. Spectrum1500433
164. Nitrofurantoin [mart.]
165. Bpbio1_000039
166. Nitrofurantoin [usp-rs]
167. Nitrofurantoin [who-dd]
168. Bdbm57045
169. Chebi:95222
170. Hms500l06
171. Hms1568b17
172. Hms1920p21
173. Hms2091h16
174. Hms2095b17
175. Hms3712b17
176. Pharmakon1600-01500433
177. Nitrofurantoin [green Book]
178. 1-[(e)-(5-nitro-2-furyl)methyleneamino]imidazolidine-2,4-dione
179. Hy-a0090
180. Zinc7997568
181. Nitrofurantoin [orange Book]
182. 2,4-imidazolidenedione, 1-(((5-nitro-2-furanyl)methylene)amino)-
183. Ccg-40108
184. Nitrofurantoin [ep Monograph]
185. Nsc757243
186. S4536
187. Stk009471
188. Stl454163
189. Nitrofurantoin [usp Monograph]
190. Akos001678301
191. Db00698
192. Nsc-757243
193. Idi1_000224
194. Macrobid Component Nitrofurantoin
195. Ncgc00091505-03
196. Ncgc00091505-04
197. Ncgc00091505-05
198. Ncgc00091505-06
199. Ncgc00091505-08
200. Ncgc00091505-09
201. Ncgc00091505-10
202. Nitrofurantoin Anhydrous [who-ip]
203. Ls-13402
204. 1-((5-nitrofurfurylidene)amino)-hydantoin
205. Nitrofurantoin Component Of Macrobid
206. Sbi-0051457.p003
207. Ab00513815
208. Bb 0310231
209. N0883
210. Nitrofurantoin Macrocrystalline [vandf]
211. A16008
212. C07268
213. D00439
214. Ab00052052_03
215. Nitrofurantoinum Anhydrous [who-ip Latin]
216. A835659
217. Hydantoin, N-(5-nitro-2-furfurylidene)-1-amino-
218. Nitrofurantoin, Vetranal(tm), Analytical Standard
219. Q-201479
220. Sr-05000001681-1
221. Sr-05000001681-2
222. Sr-05000001681-3
223. Sr-05000001681-4
224. Brd-k76927775-001-05-0
225. Nitrofurantoin, Antibiotic For Culture Media Use Only
226. Hydantoin, 1-[(5-nitrofurfurylidene)amino]- (7ci,8ci)
227. Macrobid Component Nitrofurantoin, Macrocrystalline
228. 1-{[(5-nitro-2-furyl)methylene]amino}imidazolidine-2,4-dione
229. Nitrofurantoin, Macrocrystalline Component Of Macrobid
230. (e)-1-[(5-nitro-2-furyl)methylideneamino]imidazolidine-2,4-dione
231. 1-([(5-nitro-2-furyl)methylidene]amino)-2,4-imidazolidinedione #
232. 1-[(e)-(5-nitro-2-furanyl)methylideneamino]imidazolidine-2,4-dione
233. 1-{[(5-nitrofuran-2-yl)methylidene]amino}imidazolidine-2,4-dione
234. Nitrofurantoin, United States Pharmacopeia (usp) Reference Standard
235. 1-{[(e)-1-(5-nitro-2-furyl)methylidene]amino}-1h-imidazole-2,4(3h,5h)-dione
236. 4-hydroxy-1-{[(e)-(5-nitrofuran-2-yl)methylidene]amino}-1,5-dihydro-2h-imidazol-2-one
237. N-(5-nitro-2-furfurylidine)-1-aminohydantoin; N-(5-nitrofurfurylidene)-1aminohydantoin
238. Nitrofurantoin, Pharmaceutical Secondary Standard; Certified Reference Material
1. Nitrofurantoin [monohydrate]
2. 1-((5-nitrofurfurylidene)amino)hydantoin Monohydrate
3. Anw-71970
4. Nifurantin Monohydrate
| Molecular Weight | 238.16 g/mol |
|---|---|
| Molecular Formula | C8H6N4O5 |
| XLogP3 | -0.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Exact Mass | 238.03381930 g/mol |
| Monoisotopic Mass | 238.03381930 g/mol |
| Topological Polar Surface Area | 121 Ų |
| Heavy Atom Count | 17 |
| Formal Charge | 0 |
| Complexity | 390 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 8 | |
|---|---|
| Drug Name | Furadantin |
| PubMed Health | Nitrofurantoin |
| Drug Classes | Antibiotic |
| Drug Label | Furadantin (nitrofurantoin), a synthetic chemical, is a stable, yellow, crystalline compound. Furadantin is an antibacterial agent for specific urinary tract infections. Furadantin is available in 25mg/5mL liquid suspension for oral administration.1-... |
| Active Ingredient | Nitrofurantoin |
| Dosage Form | Suspension |
| Route | Oral |
| Strength | 25mg/5ml |
| Market Status | Prescription |
| Company | Shionogi |
| 2 of 8 | |
|---|---|
| Drug Name | Macrobid |
| PubMed Health | Nitrofurantoin Combination (By mouth) |
| Drug Classes | Antibiotic |
| Active Ingredient | nitrofurantoin, macrocrystalline; Nitrofurantoin |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 25mg; 75mg |
| Market Status | Prescription |
| Company | Alvogen |
| 3 of 8 | |
|---|---|
| Drug Name | Macrodantin |
| PubMed Health | Nitrofurantoin |
| Drug Classes | Antibiotic |
| Drug Label | Macrodantin (nitrofurantoin macrocrystals) is a synthetic chemical of controlled crystal size. It is a stable, yellow, crystalline compound. Macrodantin is an antibacterial agent for specific urinary tract infections. It is available in 25 mg, 50 mg,... |
| Active Ingredient | Nitrofurantoin, macrocrystalline |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 25mg; 100mg; 50mg |
| Market Status | Prescription |
| Company | Alvogen |
| 4 of 8 | |
|---|---|
| Drug Name | Nitrofurantoin |
| PubMed Health | Nitrofurantoin |
| Drug Classes | Antibiotic |
| Drug Label | Nitrofurantoin oral suspension, USP, a synthetic chemical, is a stable, yellow, crystalline compound. Nitrofurantoin oral suspension, USP is an antibacterial agent for specific urinary tract infections. Nitrofurantoinoral suspension, USP is available... |
| Active Ingredient | Nitrofurantoin, macrocrystalline; Nitrofurantoin |
| Dosage Form | Capsule; Suspension |
| Route | Oral |
| Strength | 25mg/5ml; 100mg; 50mg |
| Market Status | Prescription |
| Company | Amneal Pharms; Novel Labs; Ivax Sub Teva Pharms; Caraco; Mylan |
| 5 of 8 | |
|---|---|
| Drug Name | Furadantin |
| PubMed Health | Nitrofurantoin |
| Drug Classes | Antibiotic |
| Drug Label | Furadantin (nitrofurantoin), a synthetic chemical, is a stable, yellow, crystalline compound. Furadantin is an antibacterial agent for specific urinary tract infections. Furadantin is available in 25mg/5mL liquid suspension for oral administration.1-... |
| Active Ingredient | Nitrofurantoin |
| Dosage Form | Suspension |
| Route | Oral |
| Strength | 25mg/5ml |
| Market Status | Prescription |
| Company | Shionogi |
| 6 of 8 | |
|---|---|
| Drug Name | Macrobid |
| PubMed Health | Nitrofurantoin Combination (By mouth) |
| Drug Classes | Antibiotic |
| Active Ingredient | nitrofurantoin, macrocrystalline; Nitrofurantoin |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 25mg; 75mg |
| Market Status | Prescription |
| Company | Alvogen |
| 7 of 8 | |
|---|---|
| Drug Name | Macrodantin |
| PubMed Health | Nitrofurantoin |
| Drug Classes | Antibiotic |
| Drug Label | Macrodantin (nitrofurantoin macrocrystals) is a synthetic chemical of controlled crystal size. It is a stable, yellow, crystalline compound. Macrodantin is an antibacterial agent for specific urinary tract infections. It is available in 25 mg, 50 mg,... |
| Active Ingredient | Nitrofurantoin, macrocrystalline |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 25mg; 100mg; 50mg |
| Market Status | Prescription |
| Company | Alvogen |
| 8 of 8 | |
|---|---|
| Drug Name | Nitrofurantoin |
| PubMed Health | Nitrofurantoin |
| Drug Classes | Antibiotic |
| Drug Label | Nitrofurantoin oral suspension, USP, a synthetic chemical, is a stable, yellow, crystalline compound. Nitrofurantoin oral suspension, USP is an antibacterial agent for specific urinary tract infections. Nitrofurantoinoral suspension, USP is available... |
| Active Ingredient | Nitrofurantoin, macrocrystalline; Nitrofurantoin |
| Dosage Form | Capsule; Suspension |
| Route | Oral |
| Strength | 25mg/5ml; 100mg; 50mg |
| Market Status | Prescription |
| Company | Amneal Pharms; Novel Labs; Ivax Sub Teva Pharms; Caraco; Mylan |
Anti-Infective Agents, Urinary /SRP: Antibacterial/
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
NITROFURANTOIN IS BACTERIOSTATIC AT CONCN OF 5-10 UG/ML & BACTERICIDAL AT 100 UG/ML, BUT IT IS NOT KNOWN WHETHER BACTERICIDAL ACTION OCCURS IN VIVO. ANTIBACTERIAL ACTIVITY IS HIGHER IN ACIDIC URINE. ...SUPERSATURATED SOLN OF NITROFURANTOIN DO NOT CAUSE CRYSTALLURIA.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 1008
NITROFURANTOIN IS ACTIVE AGAINST MANY STRAINS OF COMMON URINARY TRACT PATHOGENS E COLI, PROTEUS SPECIES, PSEUDOMONAS...ENTEROBACTER, AND STAPHYLOCOCCI, AS WELL AS ENTEROCOCCI, STREPTOCOCCI, CLOSTRIDIA, & BACILLUS SUBTILIS.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 1008
...APPROVED ONLY FOR TREATMENT OF URINARY TRACT INFECTIONS CAUSED BY MICROORGANISMS THAT ARE KNOWN TO BE SENSITIVE TO DRUG. ... IT HAS BEEN USED EFFECTIVELY TO PREVENT RECURRENT INFECTIONS & FOR PREVENTION OF BACTERIURIA AFTER PROSTATECTOMY.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1070
For more Therapeutic Uses (Complete) data for NITROFURANTOIN (8 total), please visit the HSDB record page.
A COURSE OF THERAPY SHOULD NOT EXCEED 14 DAYS, & REPEATED COURSES SHOULD BE SEPARATED BY REST PERIODS. ... PREGNANT WOMEN AT TERM, INDIVIDUALS WITH IMPAIRED RENAL FUNCTION (CREATININE CLEARANCE LESS THAN 40 ML/MIN), & CHILDREN BELOW 1 MONTH OF AGE SHOULD NOT RECEIVE NITROFURANTOIN.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1070
Maternal Medication Usually Compatible with Breast-Feeding: Nitrofurantoin: Hemolysis in infant with glucose-6-phosphate dehydrogenase (G-6-PD) deficiency. /from Table 6/
Report of the American Academy of Pediatrics Committee on Drugs in Pediatrics 93 (1): 141 (1994)
...MOST SPECIES OF PROTEUS & PSEUDOMONAS & MANY OF ENTEROBACTER & KLEBSIELLA ARE RESISTANT. ANTIBACTERIAL CONCN ARE NOT ACHIEVED IN PLASMA FOLLOWING INGESTION OF RECOMMENDED DOSES, BECAUSE DRUG IS RAPIDLY ELIMINATED. ...IN PT WITH IMPAIRED GLOMERULAR FUNCTION EFFICACY OF DRUG MAY BE DECR & SYSTEMIC TOXICITY INCR.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1069
INJECTION OF NITROFURANTOIN SODIUM IS INDICATED ONLY FOR USE IN ACUTELY ILL PT WHO CANNOT TOLERATE ORAL NITROFURANTOIN. /SODIUM NITROFURANTOIN/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 1009
For more Drug Warnings (Complete) data for NITROFURANTOIN (31 total), please visit the HSDB record page.
Nitrofurantoin is indicated to treat acute uncomplicated urinary tract infections.
FDA Label
Nitrofurantoin interferes with vital processes in bacteria, which leads to their death. Nitrofurantoin rapidly reaches therapeutic concentrations in the urine and is also cleared rapidly.
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
Anti-Infective Agents, Urinary
Substances capable of killing agents causing urinary tract infections or of preventing them from spreading. (See all compounds classified as Anti-Infective Agents, Urinary.)
J - Antiinfectives for systemic use
J01 - Antibacterials for systemic use
J01X - Other antibacterials
J01XE - Nitrofuran derivatives
J01XE01 - Nitrofurantoin
Absorption
Nitrofurantoin reaches a Cmax of 0.875-0.963mg/L with an AUC of 2.21-2.42mg\*h/L. It is 38.8-44.3% bioavailable. Taking nitrofurantoin with food increases the absorption and duration of therapeutic concentrations in the urine.
Route of Elimination
27-50% of an oral dose is excreted in the urine as unchanged nitrofurantoin. 90% of the total dose is eliminated in the urine.
Volume of Distribution
Data regarding the volume of distribution in humans is scarce but it has been reported as 0.46L/kg in dogs.
Clearance
The clearance of nitrofurantoin is 16.7-19.4L/h.
.../IT/ IS RAPIDLY & COMPLETELY ABSORBED FROM GI TRACT. ... PLASMA HALF-LIFE IS 0.3 TO 1 HR; ABOUT 40% IS EXCRETED UNCHANGED INTO URINE. AVG DOSE OF NITROFURANTOIN YIELDS URINE CONCN OF APPROX 200 UG/ML. ... RATE OF EXCRETION IS LINEARLY RELATED TO CREATININE CLEARANCE...
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1069
CLINICAL STUDIES...INDICATE THAT IN NORMAL FASTING INDIVIDUALS, LESS NITROFURANTOIN IS ABSORBED & AT SLOWER RATE FROM MACROCRYSTALLINE THAN MICROCRYSTALLINE FORM. PRESENCE OF FOOD IN INTESTINE DELAYS ABSORPTION OF BOTH FORMS APPRECIABLY, INCR PEAK LEVELS OF MACROCRYSTALLINE COMPD, BUT NOT MICROCRYSTALLINE COMPD, ENHANCES BIOAVAILABILITY OF BOTH FORMS, & PROLONGS DURATION OF THERAPEUTIC URINARY CONCN.
American Medical Association, AMA Department of Drugs. AMA Drug Evaluations. 4th ed. Chicago: American Medical Association, 1980., p. 1323
ENHANCEMENT OF...ABSORPTION BY FOOD RANGED FROM 20 TO 400%, WITH GREATEST EFFECT OCCURRING WITH LEAST SOLUBLE DOSAGE FORMS. .../IT/ IS INEFFICIENTLY ABSORBED FROM RECTAL SUPPOSITORIES...
The Chemical Society. Foreign Compound Metabolism in Mammals. Volume 5: A Review of the Literature Published during 1976 and 1977. London: The Chemical Society, 1979., p. 76
NITROFURANTOIN ABSORPTION IS SIGNIFICANTLY INCR IN MAN FROM A DRUG-DEOXYCHOLIC ACID CO-PRECIPITATE COMPARED WITH PHYS MIXT, & FASTER ABSORPTION...FROM CO-PRECIPITATE WAS ASSOCIATED WITH FASTER IN VITRO DISSOLUTION RATE. ... EXCRETED IN BILE OF DOGS & ABOUT 1/3 OF THAT EXCRETED IS REABSORBED FROM INTESTINE WITHIN 3 HR.
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 187
For more Absorption, Distribution and Excretion (Complete) data for NITROFURANTOIN (13 total), please visit the HSDB record page.
0.8-1.8% of a dose is metabolized to aminofurantoin, and 0.9% of a dose is metabolized to other metabolites.
AFTER DOSE OF 0.200 MG/KG, 22% IS EXCRETED IN URINE AS N-(5-NITROFURFURYLIDENEAMINO)-2-IMIDAZOLINE-ONE. /FROM TABLE/
Sunshine, I. (ed.). CRC Handbook of Analytical Toxicology. Cleveland: The Chemical Rubber Co., 1969., p. 356
READILY DEGRADED BY ALL /BODY/ TISSUES (EXCEPT BLOOD) INTO INACTIVE METABOLITES-HYDROXYLAMINO COMPD & AMINOFURALDEHYDENITROFURIC ACID. /HUMAN, ORAL/
American Society of Hospital Pharmacists. Data supplied on contract from American Hospital Formulary Service and other current ASHP sources., p. 1971
AFTER NITROFURANTOIN (50 MG) IV INFUSION, 47% OF THE DOSE WAS EXCRETED UNCHANGED IN THE URINE AND 1.2% WAS RECOVERED AS THE REDUCED METABOLITE AMINOFURANTOIN.
HOENER BA, PATTERSON SE; NITROFURANTOIN DISPOSITION; CLIN PHARMACOL THER 29(6) 808 (1981)
Nitrofurantoin is partially metabolized, mainly in the liver. A small fraction of the drug is reduced to form aminofurantoin.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 632
The half life of nitrofurantoin is 0.72-0.78h.
PLASMA HALF-LIFE IS 0.3 TO 1 HR...
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1069
NITROFURANTOIN HALF-LIFE WAS 0.41 HOURS IN ADULTS AND 0.95 HOURS IN 2-WEEK-OLD RATS.
WIERZBA K ET AL; PAEDIATR PAEDOL 17 (2): 293 (1982)
Nitrofurantoin is converted by bacterial nitroreductases to electrophilic intermediates which inhibit the citric acid cycle as well as synthesis of DNA, RNA, and protein.
MICROSOMAL AND SOLUBLE FRACTIONS FROM BOTH RAT LIVER AND LUNG MEDIATED THE COVALENT BINDING OF (14)C-LABELED NITROFURANTOIN (I) TO TISSUE MACROMOLECULES IN VITRO. OXYGEN STRONGLY INHIBITED THE BINDING IN BOTH FRACTIONS, AND CARBON MONOXIDE FAILED TO INHIBIT THE BINDING IN MICROSOMAL PREPARATIONS, INDICATING ACTIVATION OF I IN BOTH SYSTEMS BY NITROREDUCTION RATHER THAN OXIDATION OF THE FURAN RING. MICROSOMAL NITROREDUCTION AND COVALENT BINDING OF I WERE INHIBITED BY AN ANTIBODY AGAINST NADPH-CYTOCHROME C REDUCTASE AND COVALENT BINDING WAS ENHANCED BY THE ADDITION OF FAD. IN SOLUBLE FRACTIONS, MAXIMUM RATES OF COVALENT BINDING WERE OBTAINED IN THE PRESENCE OF NADH AND HYPOXANTHINE, AND IT WAS INHIBITED BY ALLOPURINOL, A XANTHINE OXIDASE INHIBITOR. REDUCED GLUTATHIONE DECREASED COVALENT BINDING OF I IN BOTH MICROSOMAL AND SOLUBLE FRACTIONS OF LIVER AND LUNG, BUT THE RATE OF NITROREDUCTION WAS UNAFFECTED.
PMID:36083 BOYD MR ET AL; BIOCHEM PHARMACOL 28 (5): 601 (1979)
THE HYPOTHESIS IS PRESENTED THAT THE TOXICITY OF NITROFURANS SUCH AS NITROFURANTOIN (I), WHICH ARE USED IN COMMERCIAL POULTRY PRODUCTION, IS DUE TO OXIDATIVE METABOLIC STRESS CAUSED BY THE O2- FREE RADICAL FORMED DURING METABOLISM OF THE COMPOUNDS.
COMBS GF JR; PROC-CORNELL NUTR CONF FEED MANUF: 9 (1979)
 Click Us!
Click Us! 
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38079
Submission : 2023-05-10
Status : Active
Type : II

Certificate Number : CEP 2023-346 - Rev 00
Issue Date : 2025-06-19
Type : Chemical
Substance Number : 101
Status : Valid

Date of Issue : 2024-01-23
Valid Till : 2026-06-17
Written Confirmation Number : WC-0476
Address of the Firm :

NDC Package Code : 84668-015
Start Marketing Date : 2024-08-28
End Marketing Date : 2027-12-31
Dosage Form (Strength) : POWDER (15kg/15kg)
Marketing Category : BULK INGREDIENT FOR HUMAN PRESCRIPTION COMPOUNDING
 Delivering quality and niche Active Pharmaceutical Ingredients across the global from our USFDA-approved facility.
Delivering quality and niche Active Pharmaceutical Ingredients across the global from our USFDA-approved facility.

GDUFA
DMF Review : Reviewed
Rev. Date : 2019-04-26
Pay. Date : 2019-03-05
DMF Number : 33351
Submission : 2018-12-28
Status : Active
Type : II

| Available Reg Filing : ASMF |
 Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.
Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.
 Olpha, a JSC AB City subsidiary, is a leading Baltic firm with 50 years of experience in medicines & chemical pharmaceuticals.
Olpha, a JSC AB City subsidiary, is a leading Baltic firm with 50 years of experience in medicines & chemical pharmaceuticals.
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23500
Submission : 2010-02-16
Status : Active
Type : II

Date of Issue : 2024-04-23
Valid Till : 2027-04-22
Written Confirmation Number : WC-0170
Address of the Firm :

GDUFA
DMF Review : Reviewed
Rev. Date : 2022-09-08
Pay. Date : 2022-07-27
DMF Number : 33877
Submission : 2019-05-31
Status : Active
Type : II

Certificate Number : R0-CEP 2022-074 - Rev 00
Issue Date : 2022-12-12
Type : Chemical
Substance Number : 101
Status : Valid

Date of Issue : 2024-02-12
Valid Till : 2027-02-11
Written Confirmation Number : WC-0407
Address of the Firm :

NDC Package Code : 70600-012
Start Marketing Date : 2021-12-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

| Available Reg Filing : ASMF, CA, BR |

 Panvo Organics: Advancing quality APIs and formulations with Halal, Kosher, US FDA, and WHO-approved manufacturing excellence.
Panvo Organics: Advancing quality APIs and formulations with Halal, Kosher, US FDA, and WHO-approved manufacturing excellence.
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
30
PharmaCompass offers a list of Nitrofurantoin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Nitrofurantoin manufacturer or Nitrofurantoin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Nitrofurantoin manufacturer or Nitrofurantoin supplier.
A NITROFURANTOIN MACROCRYSTALLINE manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of NITROFURANTOIN MACROCRYSTALLINE, including repackagers and relabelers. The FDA regulates NITROFURANTOIN MACROCRYSTALLINE manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. NITROFURANTOIN MACROCRYSTALLINE API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of NITROFURANTOIN MACROCRYSTALLINE manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A NITROFURANTOIN MACROCRYSTALLINE supplier is an individual or a company that provides NITROFURANTOIN MACROCRYSTALLINE active pharmaceutical ingredient (API) or NITROFURANTOIN MACROCRYSTALLINE finished formulations upon request. The NITROFURANTOIN MACROCRYSTALLINE suppliers may include NITROFURANTOIN MACROCRYSTALLINE API manufacturers, exporters, distributors and traders.
click here to find a list of NITROFURANTOIN MACROCRYSTALLINE suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A NITROFURANTOIN MACROCRYSTALLINE DMF (Drug Master File) is a document detailing the whole manufacturing process of NITROFURANTOIN MACROCRYSTALLINE active pharmaceutical ingredient (API) in detail. Different forms of NITROFURANTOIN MACROCRYSTALLINE DMFs exist exist since differing nations have different regulations, such as NITROFURANTOIN MACROCRYSTALLINE USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A NITROFURANTOIN MACROCRYSTALLINE DMF submitted to regulatory agencies in the US is known as a USDMF. NITROFURANTOIN MACROCRYSTALLINE USDMF includes data on NITROFURANTOIN MACROCRYSTALLINE's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The NITROFURANTOIN MACROCRYSTALLINE USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of NITROFURANTOIN MACROCRYSTALLINE suppliers with USDMF on PharmaCompass.
A NITROFURANTOIN MACROCRYSTALLINE CEP of the European Pharmacopoeia monograph is often referred to as a NITROFURANTOIN MACROCRYSTALLINE Certificate of Suitability (COS). The purpose of a NITROFURANTOIN MACROCRYSTALLINE CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of NITROFURANTOIN MACROCRYSTALLINE EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of NITROFURANTOIN MACROCRYSTALLINE to their clients by showing that a NITROFURANTOIN MACROCRYSTALLINE CEP has been issued for it. The manufacturer submits a NITROFURANTOIN MACROCRYSTALLINE CEP (COS) as part of the market authorization procedure, and it takes on the role of a NITROFURANTOIN MACROCRYSTALLINE CEP holder for the record. Additionally, the data presented in the NITROFURANTOIN MACROCRYSTALLINE CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the NITROFURANTOIN MACROCRYSTALLINE DMF.
A NITROFURANTOIN MACROCRYSTALLINE CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. NITROFURANTOIN MACROCRYSTALLINE CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of NITROFURANTOIN MACROCRYSTALLINE suppliers with CEP (COS) on PharmaCompass.
A NITROFURANTOIN MACROCRYSTALLINE written confirmation (NITROFURANTOIN MACROCRYSTALLINE WC) is an official document issued by a regulatory agency to a NITROFURANTOIN MACROCRYSTALLINE manufacturer, verifying that the manufacturing facility of a NITROFURANTOIN MACROCRYSTALLINE active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting NITROFURANTOIN MACROCRYSTALLINE APIs or NITROFURANTOIN MACROCRYSTALLINE finished pharmaceutical products to another nation, regulatory agencies frequently require a NITROFURANTOIN MACROCRYSTALLINE WC (written confirmation) as part of the regulatory process.
click here to find a list of NITROFURANTOIN MACROCRYSTALLINE suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing NITROFURANTOIN MACROCRYSTALLINE as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for NITROFURANTOIN MACROCRYSTALLINE API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture NITROFURANTOIN MACROCRYSTALLINE as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain NITROFURANTOIN MACROCRYSTALLINE and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a NITROFURANTOIN MACROCRYSTALLINE NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of NITROFURANTOIN MACROCRYSTALLINE suppliers with NDC on PharmaCompass.
NITROFURANTOIN MACROCRYSTALLINE Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of NITROFURANTOIN MACROCRYSTALLINE GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right NITROFURANTOIN MACROCRYSTALLINE GMP manufacturer or NITROFURANTOIN MACROCRYSTALLINE GMP API supplier for your needs.
A NITROFURANTOIN MACROCRYSTALLINE CoA (Certificate of Analysis) is a formal document that attests to NITROFURANTOIN MACROCRYSTALLINE's compliance with NITROFURANTOIN MACROCRYSTALLINE specifications and serves as a tool for batch-level quality control.
NITROFURANTOIN MACROCRYSTALLINE CoA mostly includes findings from lab analyses of a specific batch. For each NITROFURANTOIN MACROCRYSTALLINE CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
NITROFURANTOIN MACROCRYSTALLINE may be tested according to a variety of international standards, such as European Pharmacopoeia (NITROFURANTOIN MACROCRYSTALLINE EP), NITROFURANTOIN MACROCRYSTALLINE JP (Japanese Pharmacopeia) and the US Pharmacopoeia (NITROFURANTOIN MACROCRYSTALLINE USP).

