
Synopsis
Synopsis
0
KDMF
0
VMF
0
Australia
DRUG PRODUCT COMPOSITIONS
Annual Reports
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
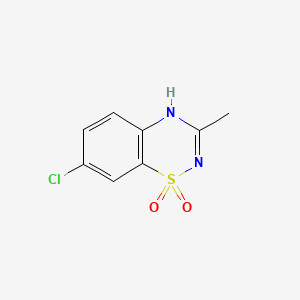

1. Hyperstat
2. Proglycem
1. 364-98-7
2. Proglycem
3. Hyperstat
4. Hypertonalum
5. Eudemine
6. Proglicem
7. Dizoxide
8. Mutabase
9. 7-chloro-3-methyl-2h-1,2,4-benzothiadiazine 1,1-dioxide
10. Sch 6783
11. Sch-6783
12. Diazossido
13. Diazoxidum
14. Srg 95213
15. Srg-95213
16. Diazoxido
17. 2h-1,2,4-benzothiadiazine, 7-chloro-3-methyl-, 1,1-dioxide
18. Eudemine Injection
19. Nsc-64198
20. Nsc 76130
21. Chebi:4495
22. Mfcd00078578
23. Nsc-76130
24. O5cb12l4fn
25. 3-methyl-7-chloro-1,2,4-benzothiadiazine 1,1-dioxide
26. Mls000028459
27. 7-cloro-3-metil-2h-1,2,4-benzotiodiazina-1,1-diossido
28. 364-98-7 (free)
29. 7-chloro-3-methyl-4h-1$l^{6},2,4-benzothiadiazine 1,1-dioxide
30. Diazossido [italian]
31. Nsc64198
32. Nsc76130
33. 7-chloro-3-methyl-2h-1,2,4-benzothiadiazine1,1-dioxide
34. C8h7cln2o2s
35. Diazossido [dcit]
36. Ncgc00015380-09
37. Cas-364-98-7
38. Smr000058392
39. 7-chloro-3-methyl-2h-benzo[e][1,2,4]thiadiazine 1,1-dioxide
40. Dsstox_cid_2914
41. Diazoxidum [inn-latin]
42. Dsstox_rid_76786
43. Diazoxido [inn-spanish]
44. Dsstox_gsid_22914
45. 7-chloro-3-methyl-4h-1,2,4-benzothiadiazine 1,1-dioxide
46. Aroglycem
47. 7-chloro-3-methyl-4h-benzo[e][1,2,4]thiadiazine 1,1-dioxide
48. Hyperstat (tn)
49. Sr-01000075314
50. Einecs 206-668-1
51. Nsc 64198
52. Unii-o5cb12l4fn
53. 7-chloro-3-methyl-2h-1,4-benzothiadiazine 1,1-dioxide
54. Diazoxide (jan/usp/inn)
55. Eudimine
56. 2h-1,4-benzothiadiazine, 7-chloro-3-methyl-, 1,1-dioxide
57. 7-chloro-3-methyl-2h-1?^{6},2,4-benzothiadiazine 1,1-dioxide
58. 7-cloro-3-metil-2h-1,2,4-benzotiodiazina-1,1-diossido [italian]
59. Proglycem (tn)
60. Prestwick_163
61. Diazoxide [usan:usp:inn:ban:jan]
62. Tocris-0964
63. Diazoxide [inn]
64. Diazoxide [jan]
65. Opera_id_608
66. Diazoxide [mi]
67. Diazoxide [usan]
68. Prestwick0_000087
69. Prestwick1_000087
70. Prestwick2_000087
71. Prestwick3_000087
72. Spectrum3_000735
73. Spectrum4_001248
74. Diazoxide [vandf]
75. Lopac-d-9035
76. Chembl181
77. Diazoxide [mart.]
78. D 9035
79. Diazoxide [usp-rs]
80. Diazoxide [who-dd]
81. Diazoxide [who-ip]
82. Cbiol_001750
83. Lopac0_000404
84. Schembl41254
85. Bspbio_000014
86. Bspbio_001307
87. Bspbio_002290
88. Kbiogr_000027
89. Kbiogr_001776
90. Kbioss_000027
91. Mls001076071
92. Mls001424164
93. Spectrum2300206
94. Spbio_001953
95. Bpbio1_000016
96. Gtpl2409
97. Diazoxide [orange Book]
98. Chembl1518123
99. Diazoxide [ep Monograph]
100. Dtxsid7022914
101. Bdbm86248
102. Kbio2_000027
103. Kbio2_002595
104. Kbio2_005163
105. Kbio3_000053
106. Kbio3_000054
107. Kbio3_001510
108. Diazoxide [usp Monograph]
109. Diazoxidum [who-ip Latin]
110. Bio1_000036
111. Bio1_000525
112. Bio1_001014
113. Bio2_000027
114. Bio2_000507
115. Hms1361b09
116. Hms1568a16
117. Hms1791b09
118. Hms1922l22
119. Hms1989b09
120. Hms2051p20
121. Hms2089l04
122. Hms2093n12
123. Hms2095a16
124. Hms2234b23
125. Hms3261a10
126. Hms3267i11
127. Hms3371l13
128. Hms3393p20
129. Hms3402b09
130. Hms3411l18
131. Hms3675l18
132. Hms3712a16
133. Hms3885h12
134. Pharmakon1600-02300206
135. 2h-1,2, 4-benzothiadiazine, 7-chloro-3-methyl-, 1,1-dioxide
136. 7-chloro-3-methyl-1lambda~4~,2,4-benzothiadiazin-1-ol 1-oxide
137. 7-chloro-3-methyl-4h-1lambda6,2,4-benzothiadiazine 1,1-dioxide
138. Bcp26107
139. Hy-b1140
140. Nsc_3019
141. Zinc3872277
142. Tox21_110132
143. Tox21_500404
144. Bdbm50237612
145. Kc-115
146. Nsc759574
147. S4630
148. Akos015896340
149. Akos024458715
150. Tox21_110132_1
151. Ccg-101062
152. Ccg-204497
153. Cs-4745
154. Db01119
155. Ks-1444
156. Lp00404
157. Nc00312
158. Nsc-759574
159. Sdccgsbi-0050390.p002
160. Idi1_033777
161. Diazoxide 100 Microg/ml In Acetonitrile
162. Ncgc00015380-01
163. Ncgc00015380-02
164. Ncgc00015380-03
165. Ncgc00015380-04
166. Ncgc00015380-05
167. Ncgc00015380-06
168. Ncgc00015380-07
169. Ncgc00015380-08
170. Ncgc00015380-10
171. Ncgc00015380-11
172. Ncgc00015380-12
173. Ncgc00015380-13
174. Ncgc00015380-20
175. Ncgc00022882-03
176. Ncgc00024907-01
177. Ncgc00024907-02
178. Ncgc00024907-03
179. Ncgc00024907-04
180. Ncgc00024907-05
181. Ncgc00024907-06
182. Ncgc00024907-07
183. Ncgc00024907-08
184. Ncgc00261089-01
185. Cas_364-98-7
186. Sy066792
187. Db-048966
188. B6526
189. D5402
190. Eu-0100404
191. Ft-0603087
192. Vu0239714-6
193. C06949
194. D00294
195. F12855
196. 364d987
197. A823275
198. Q420009
199. Sr-01000075314-1
200. Sr-01000075314-3
201. Sr-01000075314-4
202. Sr-01000075314-6
203. 3-methyl-7-chloro-1,2,4-benzothiadiazine1,1-dioxide
204. Brd-k73109821-001-05-2
205. Brd-k73109821-001-10-2
206. Diazoxide, European Pharmacopoeia (ep) Reference Standard
207. 4h-1,2,4-benzothiadiazine, 7-chloro-3-methyl-, 1,1-dioxide
208. 7-chloro-3-methyl-4h-1
209. E?,2,4-benzothiadiazine-1,1-dione
210. 7-chloro-3-methyl-4h-1
211. E6,2,4-benzothiadiazine 1,1-dioxide
212. Diazoxide, United States Pharmacopeia (usp) Reference Standard
213. 7-chloro-3-methyl-2h-1$l^{6},2,4-benzothiadiazine 1,1-dioxide
214. 7-chloro-3-methyl-4h-1$l^{6},2,4-benzothiadiazine-1,1-dione
| Molecular Weight | 230.67 g/mol |
|---|---|
| Molecular Formula | C8H7ClN2O2S |
| XLogP3 | 1.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 229.9916763 g/mol |
| Monoisotopic Mass | 229.9916763 g/mol |
| Topological Polar Surface Area | 66.9 Ų |
| Heavy Atom Count | 14 |
| Formal Charge | 0 |
| Complexity | 360 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Proglycem |
| PubMed Health | Diazoxide (By mouth) |
| Drug Classes | Gastrointestinal Agent, Glucose Regulation, Antihypoglycemic |
| Drug Label | PROGLYCEM (diazoxide) is a nondiuretic benzothiadiazine derivative taken orally for the management of symptomatic hypoglycemia. PROGLYCEM Capsules contain 50 mg diazoxide, USP. The Suspension contains 50 mg of diazoxide, USP in each milliliter an... |
| Active Ingredient | Diazoxide |
| Dosage Form | Suspension |
| Route | Oral |
| Strength | 50mg/ml |
| Market Status | Prescription |
| Company | Teva Branded Pharm |
| 2 of 2 | |
|---|---|
| Drug Name | Proglycem |
| PubMed Health | Diazoxide (By mouth) |
| Drug Classes | Gastrointestinal Agent, Glucose Regulation, Antihypoglycemic |
| Drug Label | PROGLYCEM (diazoxide) is a nondiuretic benzothiadiazine derivative taken orally for the management of symptomatic hypoglycemia. PROGLYCEM Capsules contain 50 mg diazoxide, USP. The Suspension contains 50 mg of diazoxide, USP in each milliliter an... |
| Active Ingredient | Diazoxide |
| Dosage Form | Suspension |
| Route | Oral |
| Strength | 50mg/ml |
| Market Status | Prescription |
| Company | Teva Branded Pharm |
Used parentally to treat hypertensive emergencies. Also used to treat hypoglycemia secondary to insulinoma.
Diazoxide is a potassium channel activator. Its mechanism of action revolves around enhancing cell membrane permeability to potassium ions. This action consequently elicits the relaxation of local smooth muscles. This switches off voltage-gated calcium ion channels which inhibits the generation of an action potential.
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
Vasodilator Agents
Drugs used to cause dilation of the blood vessels. (See all compounds classified as Vasodilator Agents.)
C - Cardiovascular system
C02 - Antihypertensives
C02D - Arteriolar smooth muscle, agents acting on
C02DA - Thiazide derivatives
C02DA01 - Diazoxide
V - Various
V03 - All other therapeutic products
V03A - All other therapeutic products
V03AH - Drugs for treatment of hypoglycemia
V03AH01 - Diazoxide
Absorption
Readily absorbed following oral administration.
Route of Elimination
Proglycem is extensively bound (more than 90%) to serum proteins, and is excreted in the kidneys.
Hepatic.
28 ±8.3 hours in normal adults.
Diazoxide inhibits insulin release from the pancreas, by opening potassium channels in the beta cell membrane. Diazoxide is chemically related to thiazide diuretics but does not inhibit carbonic anhydrase and does not have chloriuretic or natriuretic activity. It also exhibits hypotensive activity by reducing arteriolar smooth muscle and vascular resistance.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : DIAZOXIDE
Dosage Form : INJECTABLE;INJECTION
Dosage Strength : 15MG/ML
Approval Date : 1987-08-26
Application Number : 71519
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : HYPERSTAT
Dosage Form : INJECTABLE;INJECTION
Dosage Strength : 15MG/ML
Approval Date : 1982-01-01
Application Number : 16996
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : DIAZOXIDE
Dosage Form : SUSPENSION;ORAL
Dosage Strength : 50MG/ML
Approval Date : 2019-12-20
Application Number : 211050
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : DIAZOXIDE
Dosage Form : SUSPENSION;ORAL
Dosage Strength : 50MG/ML
Approval Date : 2020-07-08
Application Number : 210799
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD :
TE Code :
Brand Name : DIAZOXIDE
Dosage Form : Injectable; Injection
Dosage Strength : 15MG/ML
Approval Date :
Application Number : 71908
RX/OTC/DISCN :
RLD :
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code :
Brand Name : VYKAT XR
Dosage Form : TABLET, EXTENDED RELEASE;ORAL
Dosage Strength : 25MG
Approval Date : 2025-03-26
Application Number : 216665
RX/OTC/DISCN : RX
RLD : Yes
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code :
Brand Name : VYKAT XR
Dosage Form : TABLET, EXTENDED RELEASE;ORAL
Dosage Strength : 75MG
Approval Date : 2025-03-26
Application Number : 216665
RX/OTC/DISCN : RX
RLD : Yes
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code :
Brand Name : VYKAT XR
Dosage Form : TABLET, EXTENDED RELEASE;ORAL
Dosage Strength : 150MG
Approval Date : 2025-03-26
Application Number : 216665
RX/OTC/DISCN : RX
RLD : Yes
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : PROGLYCEM
Dosage Form : CAPSULE;ORAL
Dosage Strength : 50MG
Approval Date : 1982-01-01
Application Number : 17425
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : PROGLYCEM
Dosage Form : CAPSULE;ORAL
Dosage Strength : 100MG
Approval Date : 1982-01-01
Application Number : 17425
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Proglicem
Dosage Form : Diazoxide 100Mg 100 Unita' Oral Use
Dosage Strength : 100 cps 100 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Proglicem
Dosage Form : Diazoxide 100Mg 100 Unita' Oral Use
Dosage Strength : 100 cps 100 mg blister
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Proglicem
Dosage Form : Diazoxide 25Mg 100 Unita' Oral Use
Dosage Strength : 100 cps 25 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : Proglicem
Dosage Form : Capsule
Dosage Strength : 25mg
Packaging :
Approval Date : 01/12/1978
Application Number : 39081
Regulatory Info : Allowed
Registration Country : Switzerland

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
https://www.pharmacompass.com/radio-compass-blog/dmf-filings-surge-44-in-q1-2025-india-tops-list-with-51-rise-in-year-on-year-submissions
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
Patent Expiration Date : 2035-11-12
US Patent Number : 9757384
Drug Substance Claim :
Drug Product Claim :
Application Number : 216665
Patent Use Code : U-4167
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-11-12

Patent Expiration Date : 2029-03-05
US Patent Number : 7799777
Drug Substance Claim : Y
Drug Product Claim :
Application Number : 216665
Patent Use Code :
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2029-03-05

Patent Expiration Date : 2035-11-12
US Patent Number : 9757384
Drug Substance Claim :
Drug Product Claim :
Application Number : 216665
Patent Use Code : U-4167
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-11-12

Patent Expiration Date : 2035-11-12
US Patent Number : 9757384
Drug Substance Claim :
Drug Product Claim :
Application Number : 216665
Patent Use Code : U-4167
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-11-12

Patent Expiration Date : 2035-11-12
US Patent Number : 12419895
Drug Substance Claim :
Drug Product Claim :
Application Number : 216665
Patent Use Code : U-4275
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-11-12

Patent Expiration Date : 2035-11-12
US Patent Number : 12419895
Drug Substance Claim :
Drug Product Claim :
Application Number : 216665
Patent Use Code : U-4275
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-11-12

Patent Expiration Date : 2035-11-12
US Patent Number : 12343348
Drug Substance Claim :
Drug Product Claim :
Application Number : 216665
Patent Use Code : U-4211
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-11-12

Patent Expiration Date : 2035-11-12
US Patent Number : 12419895
Drug Substance Claim :
Drug Product Claim :
Application Number : 216665
Patent Use Code : U-4275
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-11-12

Patent Expiration Date : 2035-11-12
US Patent Number : 12343348
Drug Substance Claim :
Drug Product Claim :
Application Number : 216665
Patent Use Code : U-4211
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-11-12

Patent Expiration Date : 2029-03-05
US Patent Number : 7799777
Drug Substance Claim : Y
Drug Product Claim :
Application Number : 216665
Patent Use Code :
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2029-03-05

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : NP
Exclusivity Expiration Date : 2028-03-26
Application Number : 216665
Product Number : 1
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : NP
Exclusivity Expiration Date : 2028-03-26
Application Number : 216665
Product Number : 2
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : NP
Exclusivity Expiration Date : 2028-03-26
Application Number : 216665
Product Number : 3
Exclusivity Details :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
79
PharmaCompass offers a list of Diazoxide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Diazoxide manufacturer or Diazoxide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Diazoxide manufacturer or Diazoxide supplier.
PharmaCompass also assists you with knowing the Diazoxide API Price utilized in the formulation of products. Diazoxide API Price is not always fixed or binding as the Diazoxide Price is obtained through a variety of data sources. The Diazoxide Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A NCGC00024907-03 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of NCGC00024907-03, including repackagers and relabelers. The FDA regulates NCGC00024907-03 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. NCGC00024907-03 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of NCGC00024907-03 manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A NCGC00024907-03 supplier is an individual or a company that provides NCGC00024907-03 active pharmaceutical ingredient (API) or NCGC00024907-03 finished formulations upon request. The NCGC00024907-03 suppliers may include NCGC00024907-03 API manufacturers, exporters, distributors and traders.
click here to find a list of NCGC00024907-03 suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A NCGC00024907-03 DMF (Drug Master File) is a document detailing the whole manufacturing process of NCGC00024907-03 active pharmaceutical ingredient (API) in detail. Different forms of NCGC00024907-03 DMFs exist exist since differing nations have different regulations, such as NCGC00024907-03 USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A NCGC00024907-03 DMF submitted to regulatory agencies in the US is known as a USDMF. NCGC00024907-03 USDMF includes data on NCGC00024907-03's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The NCGC00024907-03 USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of NCGC00024907-03 suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The NCGC00024907-03 Drug Master File in Japan (NCGC00024907-03 JDMF) empowers NCGC00024907-03 API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the NCGC00024907-03 JDMF during the approval evaluation for pharmaceutical products. At the time of NCGC00024907-03 JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of NCGC00024907-03 suppliers with JDMF on PharmaCompass.
A NCGC00024907-03 CEP of the European Pharmacopoeia monograph is often referred to as a NCGC00024907-03 Certificate of Suitability (COS). The purpose of a NCGC00024907-03 CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of NCGC00024907-03 EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of NCGC00024907-03 to their clients by showing that a NCGC00024907-03 CEP has been issued for it. The manufacturer submits a NCGC00024907-03 CEP (COS) as part of the market authorization procedure, and it takes on the role of a NCGC00024907-03 CEP holder for the record. Additionally, the data presented in the NCGC00024907-03 CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the NCGC00024907-03 DMF.
A NCGC00024907-03 CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. NCGC00024907-03 CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of NCGC00024907-03 suppliers with CEP (COS) on PharmaCompass.
A NCGC00024907-03 written confirmation (NCGC00024907-03 WC) is an official document issued by a regulatory agency to a NCGC00024907-03 manufacturer, verifying that the manufacturing facility of a NCGC00024907-03 active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting NCGC00024907-03 APIs or NCGC00024907-03 finished pharmaceutical products to another nation, regulatory agencies frequently require a NCGC00024907-03 WC (written confirmation) as part of the regulatory process.
click here to find a list of NCGC00024907-03 suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing NCGC00024907-03 as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for NCGC00024907-03 API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture NCGC00024907-03 as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain NCGC00024907-03 and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a NCGC00024907-03 NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of NCGC00024907-03 suppliers with NDC on PharmaCompass.
NCGC00024907-03 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of NCGC00024907-03 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right NCGC00024907-03 GMP manufacturer or NCGC00024907-03 GMP API supplier for your needs.
A NCGC00024907-03 CoA (Certificate of Analysis) is a formal document that attests to NCGC00024907-03's compliance with NCGC00024907-03 specifications and serves as a tool for batch-level quality control.
NCGC00024907-03 CoA mostly includes findings from lab analyses of a specific batch. For each NCGC00024907-03 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
NCGC00024907-03 may be tested according to a variety of international standards, such as European Pharmacopoeia (NCGC00024907-03 EP), NCGC00024907-03 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (NCGC00024907-03 USP).

