
Synopsis
Synopsis
0
CEP/COS
0
NDC API
0
VMF
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
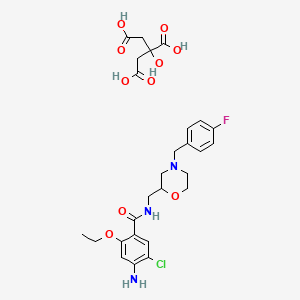

1. 4-amino-5-chloro-2-ethoxy-n-((4-(4-fluorobenzyl)-2-morpholinyl)methyl)benzamide
2. As 4370
3. As-4370
4. Mosapride
5. Mosapride Ui05msp015ct
1. 112885-42-4
2. Gasmotin
3. As-4370
4. Mosapride (citrate)
5. Tak-370
6. Rimopride Citrate
7. 4-amino-5-chloro-2-ethoxy-n-((4-(4-fluorobenzyl)morpholin-2-yl)methyl)benzamide 2-hydroxypropane-1,2,3-tricarboxylate
8. Gasmot
9. Mf497j489p
10. Dsstox_cid_26207
11. Dsstox_rid_81436
12. Dsstox_gsid_46207
13. 4-amino-5-chloro-2-ethoxy-n-[[4-[(4-fluorophenyl)methyl]morpholin-2-yl]methyl]benzamide Citrate
14. Cas-112885-42-4
15. C21h25clfn3o3.c6h8o7
16. Unii-mf497j489p
17. Gasmotin Citrate
18. Tak-370 Citrate
19. As-4370 Citrate
20. Mosapride Citrate- Bio-x
21. (+-)-4-amino-5-chloro-2-ethoxy-n-((4-(4-fluorobenzyl)-2-morpholinyl)methyl)benzamide Citrate
22. Mls006011997
23. Schembl258620
24. Mosapride Citrate [mi]
25. Tak370
26. Chembl1733174
27. Dtxsid7046207
28. Hy-b0189a
29. Amy8852
30. Tak 370
31. Hms2051p15
32. Hms2234k07
33. Hms3369k19
34. Hms3393p15
35. Hms3651o09
36. Hms3884e13
37. Mosapride Citrate [who-dd]
38. Bcp08880
39. Tox21_111874
40. Mfcd01666680
41. Mosapride Citrate - Adooq Bioscience
42. S1385
43. Akos015895400
44. Tox21_111874_1
45. Ccg-100966
46. Ccg-270241
47. Nc00216
48. Ncgc00160527-03
49. As-13388
50. Benzamide, 4-amino-5-chloro-2-ethoxy-n-((4-((4-fluorophenyl)methyl)-2-morpholinyl)methyl)-, 2-hydroxy-1,2,3-propanetricarboxylate (1:1)
51. Bm164611
52. Sw197596-2
53. 85m424
54. D70344
55. A921736
56. Q27283985
57. (+/-)-4-amino-5-chloro-2-ethoxy-n-((4-(p-fluorobenzyl)-2-morpholinyl)methyl)benzamide Citrate
58. 4-amino-5-chloro-2-ethoxy-n-[[4-[(4 -fluorophenyl)methyl]-2-morpholinyl]methyl]benzami De Citrate
59. 4-amino-5-chloro-2-ethoxy-n-[[4-[(4-fluorophenyl)methyl]-2-morpholinyl]methyl]benzamide Citrate
60. 4-amino-5-chloro-2-ethoxy-n-[[4-[(4-fluorophenyl)methyl]morpholin-2-yl]methyl]benzamide;2-hydroxypropane-1,2,3-tricarboxylic Acid.
61. Tak-370 Citrate; As-4370 Citrat;tak370 Citrate; As4370 Citrat;tak 370 Citrate; As 4370 Citrat
| Molecular Weight | 614.0 g/mol |
|---|---|
| Molecular Formula | C27H33ClFN3O10 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 12 |
| Exact Mass | 613.1838501 g/mol |
| Monoisotopic Mass | 613.1838501 g/mol |
| Topological Polar Surface Area | 209 Ų |
| Heavy Atom Count | 42 |
| Formal Charge | 0 |
| Complexity | 749 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Serotonin Receptor Agonists
Endogenous compounds and drugs that bind to and activate SEROTONIN RECEPTORS. Many serotonin receptor agonists are used as ANTIDEPRESSANTS; ANXIOLYTICS; and in the treatment of MIGRAINE DISORDERS. (See all compounds classified as Serotonin Receptor Agonists.)
Gastrointestinal Agents
Drugs used for their effects on the gastrointestinal system, as to control gastric acidity, regulate gastrointestinal motility and water flow, and improve digestion. (See all compounds classified as Gastrointestinal Agents.)

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 19425
Submission : 2006-04-20
Status : Active
Type : II

Registration Number : 304MF10119
Registrant's Address : No. 1, North Outer Ring Road, Feixian County, Shandong Province, China
Initial Date of Registration : 2022-08-10
Latest Date of Registration :



Registrant Name : Samhwa Biopharm Co., Ltd.
Registration Date : 2021-04-26
Registration Number : 20210426-209-J-813
Manufacturer Name : Samhwa Biopharmaceutical Co., Ltd. @ Fuan Pharmaceutical Group Chongqing Bosen Pharmaceutical Co., Ltd
Manufacturer Address : 150, Hyeopnyeop- ro, Siheung-si, Gyeonggi-do, No.1 Huanan Yi Road, Changshou, Chongqing China


Registrant Name : Gukjeon Co., Ltd.
Registration Date : 2025-09-17
Registration Number : 20250917-209-J-2025
Manufacturer Name : Fuan Pharmaceutical Group Chongqing Bosen Pharmaceutical Co., Ltd.
Manufacturer Address : No.1 Huanan Yi Road, Changshou District, Chongqing 401254, China


Registrant Name : Polaris AI Pharma Co., Ltd.
Registration Date : 2019-12-06
Registration Number : 20191206-209-J-384
Manufacturer Name : Polaris AI Pharma Co., Ltd. @ ['Mosapride' Manufacturer] Anhui Heryi Pharmaceutical Co., Ltd
Manufacturer Address : 25, Balan Gongdan-ro, Hyangnam-eup, Hwaseong-si, Gyeonggi-do @ ['Mosafrid' Manufacturing Plant] Industrial park, Yangcun town, Tianchang city, Anhui province, China


Date of Issue : 2026-03-24
Valid Till : 2028-07-02
Written Confirmation Number : WC-0153
Address of the Firm :




Registrant Name : Jeil Pharmaceutical Co., Ltd.
Registration Date : 2021-01-28
Registration Number : 20210128-209-J-827
Manufacturer Name : Jeil Pharmaceutical Co., Ltd.
Manufacturer Address : 7, Cheongganggachang-ro, Baegam-myeon, Cheoin-gu, Yongin-si, Gyeonggi-do (Building B and others)


Date of Issue : 2025-09-29
Valid Till : 2028-08-09
Written Confirmation Number : WC-0072
Address of the Firm :

Registrant Name : Pampia Co., Ltd.
Registration Date : 2026-01-28
Registration Number : 20181014-209-J-263(B)
Manufacturer Name : Symed Labs Limited
Manufacturer Address : Plot No. 25/B, Phase III, IDA Jeedimetla, Hyderabad, Jeedimetla Village, Quthbullapur Mandal, Medchal-Malkajgiri District Pincode 500 055, Telangana State India


 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
50
PharmaCompass offers a list of Mosapride Citrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Mosapride Citrate manufacturer or Mosapride Citrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Mosapride Citrate manufacturer or Mosapride Citrate supplier.
A Mosapride Citrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Mosapride Citrate, including repackagers and relabelers. The FDA regulates Mosapride Citrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Mosapride Citrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Mosapride Citrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Mosapride Citrate supplier is an individual or a company that provides Mosapride Citrate active pharmaceutical ingredient (API) or Mosapride Citrate finished formulations upon request. The Mosapride Citrate suppliers may include Mosapride Citrate API manufacturers, exporters, distributors and traders.
click here to find a list of Mosapride Citrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Mosapride Citrate DMF (Drug Master File) is a document detailing the whole manufacturing process of Mosapride Citrate active pharmaceutical ingredient (API) in detail. Different forms of Mosapride Citrate DMFs exist exist since differing nations have different regulations, such as Mosapride Citrate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Mosapride Citrate DMF submitted to regulatory agencies in the US is known as a USDMF. Mosapride Citrate USDMF includes data on Mosapride Citrate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Mosapride Citrate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Mosapride Citrate suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Mosapride Citrate Drug Master File in Japan (Mosapride Citrate JDMF) empowers Mosapride Citrate API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Mosapride Citrate JDMF during the approval evaluation for pharmaceutical products. At the time of Mosapride Citrate JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Mosapride Citrate suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Mosapride Citrate Drug Master File in Korea (Mosapride Citrate KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Mosapride Citrate. The MFDS reviews the Mosapride Citrate KDMF as part of the drug registration process and uses the information provided in the Mosapride Citrate KDMF to evaluate the safety and efficacy of the drug.
After submitting a Mosapride Citrate KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Mosapride Citrate API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Mosapride Citrate suppliers with KDMF on PharmaCompass.
A Mosapride Citrate written confirmation (Mosapride Citrate WC) is an official document issued by a regulatory agency to a Mosapride Citrate manufacturer, verifying that the manufacturing facility of a Mosapride Citrate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Mosapride Citrate APIs or Mosapride Citrate finished pharmaceutical products to another nation, regulatory agencies frequently require a Mosapride Citrate WC (written confirmation) as part of the regulatory process.
click here to find a list of Mosapride Citrate suppliers with Written Confirmation (WC) on PharmaCompass.
Mosapride Citrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Mosapride Citrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Mosapride Citrate GMP manufacturer or Mosapride Citrate GMP API supplier for your needs.
A Mosapride Citrate CoA (Certificate of Analysis) is a formal document that attests to Mosapride Citrate's compliance with Mosapride Citrate specifications and serves as a tool for batch-level quality control.
Mosapride Citrate CoA mostly includes findings from lab analyses of a specific batch. For each Mosapride Citrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Mosapride Citrate may be tested according to a variety of international standards, such as European Pharmacopoeia (Mosapride Citrate EP), Mosapride Citrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Mosapride Citrate USP).

