



API Suppliers
0

US DMFs Filed
0

CEP/COS Certifications
0

JDMFs Filed
0
Other Certificates
0
Other Suppliers
0
0

USA (Orange Book)
0

Europe
0

Canada
0

Australia
0

South Africa
0
Uploaded Dossiers
0
U.S. Medicaid
0
Annual Reports
0
0
USFDA Orange Book Patents
0
USFDA Exclusivities
0
Blog #PharmaFlow
0
News
EDQM
0
USP
0
JP
0
Other Listed Suppliers
0
0
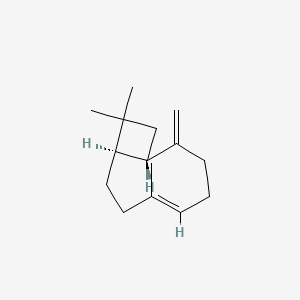

1. (e)-beta-caryophyllene
2. Beta-caryophyllene
3. Caryophyllene, (r*,4z,9s*)-(+-)-isomer
4. Caryophyllene, (r-(r*,4e,9s))-isomer
5. Caryophyllene, (r-(r*,4z,9s*))-isomer
6. Caryophyllene, (s-(r*,4e,9s*))-isomer
7. Caryophyllene, (s-(r*,4z,9s*))-isomer
8. Isocaryophyllene
9. Trans-caryophyllene
1. Beta-caryophyllene
2. Trans-caryophyllene
3. (-)-trans-caryophyllene
4. 87-44-5
5. L-caryophyllene
6. (-)-beta-caryophyllene
7. (-)-(e)-caryophyllene
8. .beta.-caryophyllene
9. B-caryophyllene
10. Trans-beta-caryophyllene
11. (e)-caryophyllene
12. (e)-beta-caryophyllene
13. Beta-caryophylene
14. Trans-.beta.-caryophyllene
15. (-)-caryophyllene
16. .beta.-caryophyllen
17. Fema No. 2252
18. Chebi:10357
19. (e)-beta-caryophylene
20. .beta.-(e)-caryophyllene
21. Beta-caryophyllen
22. Nsc-11906
23. (1r,4e,9s)-4,11,11-trimethyl-8-methylidenebicyclo[7.2.0]undec-4-ene
24. G-caryophyllene
25. 8-methylene-4,11,11-(trimethyl)bicyclo(7.2.0)undec-4-ene
26. Nsc 11906
27. (1r,4e,9s)-4,11,11-trimethyl-8-methylenebicyclo[7.2.0]undec-4-ene
28. Bicyclo(7.2.0)undec-4-ene, 8-methylene-4,11,11-trimethyl-, (e)-(1r,9s)-(-)-
29. E-.beta.-caryophyllene
30. Bhw853au9h
31. .beta.-caryophyllene, (-)
32. Tincturoid
33. 8-methylene-4,11,11-(trimethyl)bicyclo[7.2.0]undec-4-ene
34. Nsc11906
35. Caryophyllene B
36. Beta-cariofillene
37. (1r,4e,9s)-4,11,11-trimethyl-8-methylene-bicyclo[7.2.0]undec-4-ene
38. Beta-caryophillene
39. (1r,4e,9s)-4,11,11-trimethyl-8-methylenebicyclo(7.2.0)undec-4-ene
40. Bicyclo[7.2.0]undec-4-ene, 8-methylene-4,11,11-trimethyl-, (e)-(1r,9s)-(-)-
41. Bicyclo[7.2.0]undec-4-ene, 4,11,11-trimethyl-8-methylene-, [1r-(1r*,4e,9s*)]-
42. E-beta-caryophyllene
43. Mfcd00075925
44. Caryophyllene, (e)
45. (?)-b-caryophyllene
46. Beta-(e)-caryophyllene
47. Beta-trans-caryophyllene
48. (?)-trans-caryophyllene
49. Dsstox_cid_4739
50. Caryophyllene [mi]
51. .beta.-trans-caryophyllene
52. (-)-i(2)-caryophyllene
53. Dsstox_rid_77517
54. Dsstox_gsid_24739
55. Chembl445740
56. Beta-caryophyllene [fcc]
57. Dtxsid8024739
58. Beta-caryophyllene [inci]
59. Bicyclo(7.2.0)undec-4-ene, 4,11,11-trimethyl-8-methylene-, (1r,4e,9s)-
60. Hy-n1415
61. Zinc8234282
62. .beta.-caryophyllene [fhfi]
63. Tox21_301497
64. (1s,9r)-6,10,10-trimethyl-2-methylenebicyclo[7.2.0]undec-5-ene
65. Bdbm50529607
66. S6058
67. Akos024283988
68. Lmpr0103120001
69. Beta-caryophyllene, >=80%, Fcc, Fg
70. Cas-87-44-5
71. Ncgc00142620-01
72. Ncgc00255159-01
73. (-)-trans-caryophyllene, Analytical Standard
74. Cs-0016839
75. C09629
76. Beta-caryophyllene 2000 Microg/ml In Acetonitrile
77. Q421614
78. W-109317
79. (-)-trans-caryophyllene, >=98.5% (sum Of Enantiomers, Gc)
80. Trans-(1r,9s)-4,11,11-trimethyl-8-methylenebicyclo[7.2.0]undec-4-ene
81. 8-methylene-4,11,11-(trimethyl)bicyclo(7.2.0)undec-4-ene, (1r,4e,9s)-
82. Bicyclo[7.2.0]undec-4-ene, 4,11,11-trimethyl-8-methylene-, (e)-(1r,9s)-(-)-
83. Bicyclo[7.2.0]undec-4-ene, 4,11,11-trimethyl-8-methylene-, [1r- (1r*,4e,9s*)]-
| Molecular Weight | 204.35 g/mol |
|---|---|
| Molecular Formula | C15H24 |
| XLogP3 | 4.4 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 0 |
| Exact Mass | 204.187800766 g/mol |
| Monoisotopic Mass | 204.187800766 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 15 |
| Formal Charge | 0 |
| Complexity | 293 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Inflammatory Agents, Non-Steroidal
Anti-inflammatory agents that are non-steroidal in nature. In addition to anti-inflammatory actions, they have analgesic, antipyretic, and platelet-inhibitory actions. They act by blocking the synthesis of prostaglandins by inhibiting cyclooxygenase, which converts arachidonic acid to cyclic endoperoxides, precursors of prostaglandins. Inhibition of prostaglandin synthesis accounts for their analgesic, antipyretic, and platelet-inhibitory actions; other mechanisms may contribute to their anti-inflammatory effects. (See all compounds classified as Anti-Inflammatory Agents, Non-Steroidal.)


