
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
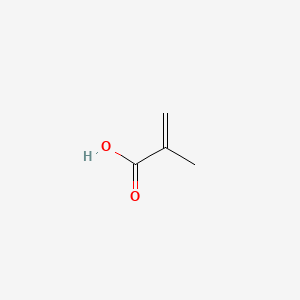

1. 2-methylacrylic Acid
2. Methacrylic Acid, Calcium Salt
3. Methacrylic Acid, Sodium Salt
4. Sodium Methacrylate
1. 79-41-4
2. 2-propenoic Acid, 2-methyl-
3. 2-methylacrylic Acid
4. Methylacrylic Acid
5. 2-methylpropenoic Acid
6. 2-methylprop-2-enoic Acid
7. 2-methyl-2-propenoic Acid
8. Alpha-methylacrylic Acid
9. 2-methylenepropionic Acid
10. Acrylic Acid, 2-methyl-
11. Alpha-methacrylic Acid
12. Propionic Acid, 2-methylene-
13. Polymethacrylic Acid
14. Kyselina Methakrylova
15. Methacrylsaeure
16. Methakrylsaeure
17. Nsc 7393
18. 25087-26-7
19. 2-methylpropensaeure
20. 2-methyl-acrylic Acid
21. .alpha.-methacrylic Acid
22. .alpha.-methylacrylic Acid
23. Chebi:25219
24. Nsc-7393
25. 1cs02g8656
26. 2-methacrylic Acid
27. Methacrylicacid
28. Propenoic Acid, 2-methyl
29. Acido Metacrilico
30. Acide Methacrylique
31. Acido Metacrilico [spanish]
32. Acide Methacrylique [french]
33. Kyselina Methakrylova [czech]
34. Ccris 5925
35. 2-propenoic Acid, 2-methyl-, Homopolymer
36. Hsdb 2649
37. Einecs 201-204-4
38. Un2531
39. Methacrylic Acid Homopolymer
40. Brn 1719937
41. Ai3-15724
42. Unii-1cs02g8656
43. Methyl Acrylic Acid
44. Gmaa Methacrylic Acid
45. Methacrylic Acid Glacial
46. Methacrylic Acid Monomer
47. Glacial Methacrylic Acid
48. Methacrylic Ac Id
49. Dsstox_cid_5542
50. Ch2=c(ch3)cooh
51. Ec 201-204-4
52. Methacrylic Acid, Inhibited
53. Methacrylic Acid, Stabilized
54. Dsstox_rid_77824
55. Dsstox_gsid_25542
56. Wln: Qvy1&u1
57. 4-02-00-01518 (beilstein Handbook Reference)
58. Un 2531 (salt/mix)
59. Methacrylic Acid [mi]
60. Methacrylic Acid [hsdb]
61. Methacrylic Acid [inci]
62. Chembl1213531
63. Dtxsid3025542
64. Nsc7393
65. Methacrylic Acid [who-dd]
66. Str02118
67. Zinc1683524
68. Tox21_200826
69. Bbl011616
70. Mfcd00002651
71. Stl163339
72. Akos000121138
73. Cas-79-41-4
74. Methacrylic Acid, Inhibited (salt/mix)
75. Ncgc00248843-01
76. Ncgc00258380-01
77. Bp-30227
78. Ft-0613003
79. M0079
80. Methacrylic Acid, Saj First Grade, >=98.0%
81. Q165949
82. J-521686
83. Methacrylic Acid, Inhibited [un2531] [corrosive]
84. Methacrylic Acid, Contains 250 Ppm Mehq As Inhibitor, 99%
85. 25068-55-7
| Molecular Weight | 86.09 g/mol |
|---|---|
| Molecular Formula | C4H6O2 |
| XLogP3 | 0.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Exact Mass | 86.036779430 g/mol |
| Monoisotopic Mass | 86.036779430 g/mol |
| Topological Polar Surface Area | 37.3 Ų |
| Heavy Atom Count | 6 |
| Formal Charge | 0 |
| Complexity | 83.5 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Methacrylic acid is readily absorbed through mucous membranes of the lungs and gastrointestinal tract and the skin; and is rapidly distributed to all major tissues.
PMID:16596768 Cosmetic Ingredient Review; International Journal of Toxicology 24(suppl 5):33-51(2005)
Deposition of methacrylic acid vapors in the surgically isolated upper respiratory tract (URT) of anesthetised rats was studied after inhalation of 450 ug/L (133 ppm) using a unidirectional respiratory flow technique (cyclic flow studies were not possible due to vapor absorption on the cyclic flow pump) for 60 min. Deposition of methacrylic acid was measured throughout exposure determining the difference in vapor concentration of methacrylic acid in the inspired and the URT expiring air. Deposition rates (from 30 to 60 min of exposure) of about 95% were observed under 200 mL/min unidirectional flow conditions. However, the degree of penetration to underlying cells could not be derived from this experiment. These results indicate that after inhalation in rats most of the methacrylic acid does not reach the lung.
European Chemicals Bureau, Institute for Health and Consumer Protection, European Union Risk Assessment Report, Methacrylic acid (CAS # 79-41-4) p. 48 (2002). Available from, as of July 9, 2008: https://ecb.jrc.it
After a single oral administration of the sodium salt of methacrylic acid to Wistar rats (540 mg/kg bw) methacrylic acid was detected in the blood serum by means of HPCL. The maximum concentration was found after 10 min, whereas after 60 min no more methacrylic acid was detectable. /Methacrylic acid, sodium salt/
European Chemicals Bureau, Institute for Health and Consumer Protection, European Union Risk Assessment Report, Methacrylic acid (CAS # 79-41-4) p. 48 (2002). Available from, as of July 9, 2008: https://ecb.jrc.it
Methacrylates are metabolized via two basic pathways, hydrolysis and conjugation. Methacrylic acid is a physiological substrate of the valine pathway and is metabolized toCO2 by two substrates of the citric acid cycle, methylmalonyl and succinyl- CoA.
PMID:16596768 Cosmetic Ingredient Review; International Journal of Toxicology 24(suppl 5):33-51(2005)
Male Wistar rats dosed orally with radiolabelled methyl methacrylate in corn oil revealed that endogenously generated methacrylic acid (0.08% of the dose) was metabolized using the pathway present in mammalian cells for the metabolism of valine, with CO2 and water as the ultimate metabolites.
PMID:16596768 Cosmetic Ingredient Review; International Journal of Toxicology 24(suppl 5):33-51(2005)
Methacrylic acid is a physiological metabolite of the Valine pathway. After activation with Acetyl-CoA it is converted into methyl-malonyl CoA and Succinyl-CoA which enters the citric acid cycle.
European Chemicals Bureau; IUCLID Dataset, Methacrylic Acid (CAS # 79-41-4) p.137 (2000 CD-ROM edition). Available from, as of July 11, 2008: https://esis.jrc.ec.europa.eu/
Methacrylic acid is metabolized primarily through B12-dependent pathway of propionate metabolism. It is first converted to the coenzyme-A ester. This catabolic pathway leads to the tricarboxylic acid (TCA) cycle and ultimately to CO2.
European Chemicals Bureau; IUCLID Dataset, Methacrylic Acid (CAS # 79-41-4) p.130 (2000 CD-ROM edition). Available from, as of July 11, 2008: https://esis.jrc.ec.europa.eu/
METHYL METHACRYLATE, THE MONOMER COMPONENT OF POLY(METHYL METHACRYLATE) CEMENT USED IN ORTHOPEDIC SURGERY, UNDERWENT HYDROLYSIS, TO METHACRYLIC ACID DURING HIP REPLACEMENT OPERATIONS. CIRCULATING LEVELS OF METHACRYLIC ACID WERE COMPARABLE TO THOSE OF METHYL METHACRYLATE, THE CONCENTRATIONS BEING 0-15 UG/CC.
PMID:477128 CROUT DH G; CLIN ORTHOP RELAT RES 141: 90-5 (1979)
Market Place

ABOUT THIS PAGE
57
PharmaCompass offers a list of Methacrylic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Methacrylic Acid manufacturer or Methacrylic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Methacrylic Acid manufacturer or Methacrylic Acid supplier.
A Methacrylic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Methacrylic Acid, including repackagers and relabelers. The FDA regulates Methacrylic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Methacrylic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Methacrylic Acid supplier is an individual or a company that provides Methacrylic Acid active pharmaceutical ingredient (API) or Methacrylic Acid finished formulations upon request. The Methacrylic Acid suppliers may include Methacrylic Acid API manufacturers, exporters, distributors and traders.
Methacrylic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Methacrylic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Methacrylic Acid GMP manufacturer or Methacrylic Acid GMP API supplier for your needs.
A Methacrylic Acid CoA (Certificate of Analysis) is a formal document that attests to Methacrylic Acid's compliance with Methacrylic Acid specifications and serves as a tool for batch-level quality control.
Methacrylic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Methacrylic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Methacrylic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Methacrylic Acid EP), Methacrylic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Methacrylic Acid USP).

