
Synopsis
Synopsis
0
USDMF
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Hydrochloride, Lymecycline
2. Lymecycline Hydrochloride
3. Lysine, Tetracycline-l-methylene
4. Lysine, Tetracyclinemethylene
5. N Lysinomethyltetracycline
6. N-lysinomethyltetracycline
7. Tetracycline L Methylene Lysine
8. Tetracycline-l-methylene Lysine
9. Tetracyclinemethylene Lysine
1. Tetralysal
2. N-lysinomethyltetracycline
3. Limeciclina
4. Lymecyclinum
5. Tetracycline-l-methylenelysine
6. Ciclolysal
7. Mucomycin
8. 992-21-2
9. Tetracycline-l-methylene Lysine
10. Ciclolysine
11. Tetracyclinemethylenelysine
12. Lymecycline (inn)
13. (+)-n-(5-amino-5-carboxypentylaminomethyl)-4-dimethylamino-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxonaphthacene-2-carboxamide
14. Tetralisal
15. 7d6em3s13p
16. N6-((4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamido)methyl)lysine
17. Chebi:59040
18. N(2)-(((+)-5-amino-5-carboxypentylamino)methyl)tetracycline
19. Lymecycline [inn]
20. L-lysine,n6-[[[[(4s,4as,5as,6s,12as)-4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenyl]carbonyl]amino]methyl]-
21. N(6)-[({[(4s,4as,5as,6s,12as)-4-(dimethylamino)-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracen-2-yl]carbonyl}amino)methyl]-l-lysine
22. Infaciclina
23. Lisinbiotic
24. Vebicyclysal
25. Ciclisin
26. Armyl
27. Tetraciclina-l-metilenlisina
28. (2s)-6-[[[(4s,4as,5as,6s,12ar)-4-(dimethylamino)-1,6,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4,4a,5,5a-tetrahydrotetracene-2-carbonyl]amino]methylamino]-2-aminohexanoic Acid
29. Lymecycline [inn:ban]
30. Lymecyclinum [inn-latin]
31. Limeciclina [inn-spanish]
32. Tetracycline, Lysinomethyl-
33. Unii-7d6em3s13p
34. Einecs 213-592-2
35. Tetraciclina-l-metilenlisina [italian]
36. Lymecycline (85%)
37. Lymecycline [mi]
38. Lymecycline [mart.]
39. Lymecycline [who-dd]
40. Schembl149162
41. Tetracycline-l Methylene-lysine
42. Chembl2103929
43. Schembl23248015
44. Gtpl10912
45. Lymecycline [ep Monograph]
46. N(sup 2)-(((+)-5-amino-5-carboxypentylamino)methyl)tetracycline
47. Zinc53682936
48. Akos016340328
49. Db00256
50. Ks-1366
51. 2-naphthacenecarboxamide, 4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-, Lysinemethylene Deriv.
52. D06884
53. Q897051
54. N2-(((+)-5-amino-5-carboxypentylamino)methyl)tetracycline
55. L-lysine, N6-(((((4s,4as,5as,6s,12as)-4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenyl)carbonyl)amino)methyl)-
56. L-lysine, N6-((((4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenyl)carbonyl)amino)methyl)-, (4s-(4alpha,4aalpha,6beta,12aalpha))-
57. Lysine, N(sup 6)-((4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamido)methyl)-, (+)-
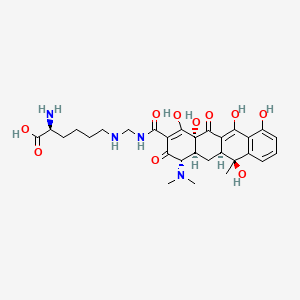
| Molecular Weight | 602.6 g/mol |
|---|---|
| Molecular Formula | C29H38N4O10 |
| XLogP3 | -4.4 |
| Hydrogen Bond Donor Count | 9 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 10 |
| Exact Mass | 602.25879342 g/mol |
| Monoisotopic Mass | 602.25879342 g/mol |
| Topological Polar Surface Area | 243 Ų |
| Heavy Atom Count | 43 |
| Formal Charge | 0 |
| Complexity | 1230 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Lymecycline is used for the treatment of acne in addition to other susceptible infections; propionibacterium is often the cause of acne. Some of the infections that can be treated with lymecycline include upper respiratory tract infections, urinary tract infections, bronchitis, chlamydial infections, and rickettsial infections.
Lymecycline, like other tetracyclines, exerts bacteriostatic actions on intracellular and extracellular bacteria, treating susceptible bacterial infections. It has been shown to be safe and effective in the treatment of moderate to severe acne. It is important to note that like other tetracyclines, lymecycline may cause esophageal irritation and ulceration, which can be prevented by drinking adequate fluids during administration. It also has the potential to cause photosensitivity. Lymecycline can lead to renal tubular acidosis or hepatic toxicity. It is not recommended to administer this drug in patients with renal disease or severe hepatic disease.
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
J - Antiinfectives for systemic use
J01 - Antibacterials for systemic use
J01A - Tetracyclines
J01AA - Tetracyclines
J01AA04 - Lymecycline
Absorption
Lymecycline is 77-88% absorbed after oral administration with a relative bioavailability of 70%. The Cmax of lymecycline is 2.1 mg/L and is achieved about 3 hours after administration. The AUC is 21.9 4.3 mgh/L.
Route of Elimination
Lymecycline is 25% eliminated in the urine. Based on being a member of the tetracycline drug class, fecal elimination is likely another route of elimination.
Volume of Distribution
Lymecycline is lipophilic and easily crosses the cell membrane and passively diffuses through bacterial porin channels. As a second-generation tetracycline, the concentration in the bile ranges from 10 to 25 times higher than plasma concentration. In general, the volume of distribution of tetracyclines ranges from 1.31.7 L/kg or 100130 L.
Clearance
Lymecycline is partially cleared by the kidneys, like other tetracyclines.
The half-life of lymecycline is approximately 8 hours.
Normally, the ribosome synthesizes proteins through the binding of aminoacyl-tRNA to the mRNA-ribosome complex. Lymecycline binds to the 30S ribosomal subunit, preventing amino-acyl tRNA from binding to the A site of the ribosome, which prevents the elongation of polypeptide chains. This results in bacteriostatic actions, treating various infections.

Certificate Number : R1-CEP 2016-186 - Rev 01
Issue Date : 2023-04-26
Type : Chemical
Substance Number : 1654
Status : Valid



Certificate Number : R0-CEP 2023-172 - Rev 00
Issue Date : 2023-08-09
Type : Chemical
Substance Number : 1654
Status : Valid






USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others

Certificate Number : CEP 2024-386 - Rev 00
Issue Date : 2026-02-05
Type : Chemical
Substance Number : 1654
Status : Valid

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
86
PharmaCompass offers a list of Lymecycline API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Lymecycline manufacturer or Lymecycline supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Lymecycline manufacturer or Lymecycline supplier.
A Lymecycline manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Lymecycline, including repackagers and relabelers. The FDA regulates Lymecycline manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Lymecycline API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Lymecycline manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Lymecycline supplier is an individual or a company that provides Lymecycline active pharmaceutical ingredient (API) or Lymecycline finished formulations upon request. The Lymecycline suppliers may include Lymecycline API manufacturers, exporters, distributors and traders.
click here to find a list of Lymecycline suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Lymecycline CEP of the European Pharmacopoeia monograph is often referred to as a Lymecycline Certificate of Suitability (COS). The purpose of a Lymecycline CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Lymecycline EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Lymecycline to their clients by showing that a Lymecycline CEP has been issued for it. The manufacturer submits a Lymecycline CEP (COS) as part of the market authorization procedure, and it takes on the role of a Lymecycline CEP holder for the record. Additionally, the data presented in the Lymecycline CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Lymecycline DMF.
A Lymecycline CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Lymecycline CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Lymecycline suppliers with CEP (COS) on PharmaCompass.
A Lymecycline written confirmation (Lymecycline WC) is an official document issued by a regulatory agency to a Lymecycline manufacturer, verifying that the manufacturing facility of a Lymecycline active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Lymecycline APIs or Lymecycline finished pharmaceutical products to another nation, regulatory agencies frequently require a Lymecycline WC (written confirmation) as part of the regulatory process.
click here to find a list of Lymecycline suppliers with Written Confirmation (WC) on PharmaCompass.
Lymecycline Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Lymecycline GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Lymecycline GMP manufacturer or Lymecycline GMP API supplier for your needs.
A Lymecycline CoA (Certificate of Analysis) is a formal document that attests to Lymecycline's compliance with Lymecycline specifications and serves as a tool for batch-level quality control.
Lymecycline CoA mostly includes findings from lab analyses of a specific batch. For each Lymecycline CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Lymecycline may be tested according to a variety of international standards, such as European Pharmacopoeia (Lymecycline EP), Lymecycline JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Lymecycline USP).

