
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Australia
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
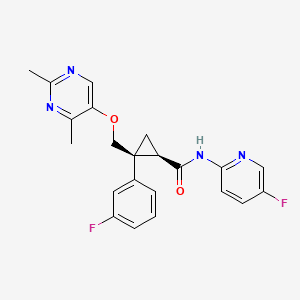

1. (1r,2s)-2-(((2,4-dimethyl-5-pyrimidinyl)oxy)methyl)-2-(3-fluorophenyl)-n-(5-fluoro-2-pyridinyl)cyclopropanecarboxamide
2. Cyclopropanecarboxamide, 2-(((2,4-dimethyl-5-pyrimidinyl)oxy)methyl)-2-(3-fluorophenyl)-n-(5-fluoro-2-pyridinyl)-, (1r,2s)-
3. Dayvigo
1. 1369764-02-2
2. Dayvigo
3. E-2006
4. E2006
5. 0k5743g68x
6. (1r,2s)-2-[(2,4-dimethylpyrimidin-5-yl)oxymethyl]-2-(3-fluorophenyl)-n-(5-fluoropyridin-2-yl)cyclopropane-1-carboxamide
7. (1r,2s)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluorophenyl)-n-(5-fluoropyridin-2-yl)cyclopropanecarboxamide
8. Cyclopropanecarboxamide, 2-(((2,4-dimethyl-5-pyrimidinyl)oxy)methyl)-2-(3-fluorophenyl)-n-(5-fluoro-2-pyridinyl)-, (1r,2s)-
9. (1~{r},2~{s})-2-[(2,4-dimethylpyrimidin-5-yl)oxymethyl]-~{n}-(5-fluoranylpyridin-2-yl)-2-(3-fluorophenyl)cyclopropane-1-carboxamide
10. E 2006
11. Lemborexant [usan:inn]
12. Unii-0k5743g68x
13. (1r,2s)-2-(((2,4-dimethyl-5-pyrimidinyl)oxy)methyl)-2-(3-fluorophenyl)-n-(5-fluoro-2-pyridinyl)cyclopropanecarboxamide
14. (1r,2s)-2-((2,4-dimethylpyrimidin-5-yl)oxymethyl)-2-(3-fluorophenyl)-n-(5-fluoropyridin-2-yl)cyclopropane-1-carboxamide
15. (1r,2s)-2-[[(2,4-dimethyl-5-pyrimidinyl)oxy]methyl]-2-(3-fluorophenyl)-n-(5-fluoro-2-pyridinyl)cyclopropanecarboxamide
16. Cyclopropanecarboxamide, 2-[[(2,4-dimethyl-5-pyrimidinyl)oxy]methyl]-2-(3-fluorophenyl)-n-(5-fluoro-2-pyridinyl)-, (1r,2s)-
17. Dayvigo (tn)
18. Lemborexant [inn]
19. Lemborexant(e2006)
20. Lemborexant [mi]
21. Lemborexant; E-2006
22. Lemborexant [jan]
23. Lemborexant [usan]
24. Lemborexant [who-dd]
25. Lemborexant (jan/usan/inn)
26. Gtpl9302
27. Schembl2116558
28. Chembl3545367
29. Lemborexant [orange Book]
30. Dtxsid401027940
31. Amy27888
32. Ex-a2337
33. Bdbm50093793
34. Zinc118073503
35. Db11951
36. Compound 34 [pmid: 25953512]
37. Ncgc00488783-01
38. Ac-30918
39. Hy-16725
40. J3.610.985h
41. D11022
42. E-2006e-2006
43. A886588
44. Q20707990
45. (1r,2s)-2-{[(2,4-dimethylpyrimidin-5-yl)oxy]methyl}-2-(3-fluorophenyl)-n-(5-fluoropyridin-2-yl)cyclopropane-1-carboxamide
| Molecular Weight | 410.4 g/mol |
|---|---|
| Molecular Formula | C22H20F2N4O2 |
| XLogP3 | 3.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Exact Mass | 410.15543222 g/mol |
| Monoisotopic Mass | 410.15543222 g/mol |
| Topological Polar Surface Area | 77 Ų |
| Heavy Atom Count | 30 |
| Formal Charge | 0 |
| Complexity | 612 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Lemborexant is indicated for the treatment of adult patients with insomnia characterized by difficulties with sleep onset and/or sleep maintenance.
FDA Label
Lemborexant promotes sleep by antagonizing the actions of wake-promoting chemicals in the brain. Episodes of complex sleep behaviors (e.g. eating food, having sex, making phone calls) have been reported in patients using lemborexant - these events may occur in hypnotic-naive and hyponotic-experienced patients, and patients are unlikely to remember these events. Patients exhibiting complex sleep behaviors should discontinue lemborexant immediately. Lemborexant may carry some risk of abuse, and should be used with caution in patients with a history of alcohol or drug addiction. Its controlled substance schedule is currently under review by the Drug Enforcement Administration.
Orexin Receptor Antagonists
Substances that bind to and inhibit the action of OREXIN RECEPTORS. Drugs in this class have been used as SLEEP AIDS, PHARMACEUTICAL. (See all compounds classified as Orexin Receptor Antagonists.)
N - Nervous system
N05 - Psycholeptics
N05C - Hypnotics and sedatives
N05CM - Other hypnotics and sedatives
N05CM21 - Lemborexant
Absorption
Animal models of lemborexant disposition have demonstrated rapid absorption following oral administration. The Tmax of lemborexant is approximately 1-3 hours, or 3-5 hours following administration of a high-fat, high-calorie meal. Cmax and AUC0-24h increase at a rate slightly less than proportionate to the given dose. Following administration of a high-fat, high-calorie meal, Cmax is decreased by 23% and AUC0-inf is increased by 18%. AUC, Cmax, and terminal half-life are increased in the presence of moderate hepatic impairment, and AUC (but not half-life) is increased in the presence of mild hepatic impairment.
Route of Elimination
Following oral administration, 57.4% of the dose is found in the feces and 29.1% in the urine. Less than 1% of the dose recovered in the urine exists as unchanged parent drug, suggesting extensive metabolism.
Volume of Distribution
The volume of distribution of lemborexant is 1970 L, indicating extensive tissue distribution.
Given that less than 1% of an administered dose is recovered unchanged in the urine, it is likely that lemborexant is extensively metabolized - this has been confirmed in rat and monkey models, but its metabolism in humans has not been fully characterized. Prescribing information states that it is predominantly metabolized by CYP3A4, with a smaller contribution by CYP3A5. The major circulating metabolite is lemborexant's M10 metabolite, which is pharmacologically active and binds to orexin receptors with a similar affinity to the parent drug. The M10 metabolite has the potential to induce CYP3A and CYP2B6 enzymes, weakly inhibit CYP3A enzymes, and is a substrate of P-gp transporters.
The half-life for lemborexant at doses of 5mg and 10mg is 17 and 19 hours, respectively.
The orexin neuropeptide signaling system is involved in many physiologic functions, including sleep/wake control. Orexin-A and orexin-B activate post-synaptic G-protein coupled orexin-1 receptors (OX1R) and orexin-2 receptors (OX2R), which are found on neurons in the hypothalamus that project to numerous wake-controlling nuclei. Each receptor carries slightly different activity - activation of OX1R appears to suppress the onset of rapid eye movement (REM) sleep, whereas activation of OX2R appears to suppress non-REM sleep. Lemborexant is an competitive antagonist of OX1R and OX2R. By blocking the binding of wake-promoting orexin-A and -B at these receptors, lemborexant suppresses the wake-drive, thereby promoting sleep.
 Click Us!
Click Us! 
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 43962
Submission : 2026-03-30
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38219
Submission : 2023-03-27
Status : Active
Type : II

 Zhejiang Hengkang Pharm Group is a dynamic pharmaceutical entity, spanning drug research, large-scale production, and global marketing.
Zhejiang Hengkang Pharm Group is a dynamic pharmaceutical entity, spanning drug research, large-scale production, and global marketing.
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.



 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 43962
Submission : 2026-03-30
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38219
Submission : 2023-03-27
Status : Active
Type : II
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

Reply
10 Jan 2023
Reply
14 Feb 2022
Reply
11 Jan 2021
Reply
26 Sep 2020
Reply
01 Sep 2020
Reply
24 Jul 2020
Reply
21 May 2020
Reply
06 Jan 2020
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
Patent Expiration Date : 2031-09-20
Date Granted : 2015-12-08
Brand Name : DAYVIGO
Patent Number : 2811895
Filing Date : 2011-09-20
Strength per Unit : 5 mg
Dosage Form : TABLET
Human Or VET : Human
Route of Administration : ORAL
Patent Expiration Date : 2031-09-20
Date Granted : 2015-12-08

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Patent Expiration Date : 2031-09-20
Date Granted : 2015-12-08
Brand Name : DAYVIGO
Patent Number : 2811895
Filing Date : 2011-09-20
Strength per Unit : 10 mg
Dosage Form : TABLET
Human Or VET : Human
Route of Administration : ORAL
Patent Expiration Date : 2031-09-20
Date Granted : 2015-12-08

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Patent Expiration Date : 2035-10-21
Date Granted : 2022-08-23
Brand Name : DAYVIGO
Patent Number : 2964504
Filing Date : 2015-10-21
Strength per Unit : 5 mg
Dosage Form : TABLET
Human Or VET : Human
Route of Administration : ORAL
Patent Expiration Date : 2035-10-21
Date Granted : 2022-08-23

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Patent Expiration Date : 2035-10-21
Date Granted : 2022-08-23
Brand Name : DAYVIGO
Patent Number : 2964504
Filing Date : 2015-10-21
Strength per Unit : 10 mg
Dosage Form : TABLET
Human Or VET : Human
Route of Administration : ORAL
Patent Expiration Date : 2035-10-21
Date Granted : 2022-08-23

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE
41
PharmaCompass offers a list of Lemborexant API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Lemborexant manufacturer or Lemborexant supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Lemborexant manufacturer or Lemborexant supplier.
A Lemborexant manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Lemborexant, including repackagers and relabelers. The FDA regulates Lemborexant manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Lemborexant API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Lemborexant manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Lemborexant supplier is an individual or a company that provides Lemborexant active pharmaceutical ingredient (API) or Lemborexant finished formulations upon request. The Lemborexant suppliers may include Lemborexant API manufacturers, exporters, distributors and traders.
click here to find a list of Lemborexant suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Lemborexant DMF (Drug Master File) is a document detailing the whole manufacturing process of Lemborexant active pharmaceutical ingredient (API) in detail. Different forms of Lemborexant DMFs exist exist since differing nations have different regulations, such as Lemborexant USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Lemborexant DMF submitted to regulatory agencies in the US is known as a USDMF. Lemborexant USDMF includes data on Lemborexant's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Lemborexant USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Lemborexant suppliers with USDMF on PharmaCompass.
Lemborexant Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Lemborexant GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Lemborexant GMP manufacturer or Lemborexant GMP API supplier for your needs.
A Lemborexant CoA (Certificate of Analysis) is a formal document that attests to Lemborexant's compliance with Lemborexant specifications and serves as a tool for batch-level quality control.
Lemborexant CoA mostly includes findings from lab analyses of a specific batch. For each Lemborexant CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Lemborexant may be tested according to a variety of international standards, such as European Pharmacopoeia (Lemborexant EP), Lemborexant JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Lemborexant USP).

