



API Suppliers

US DMFs Filed

CEP/COS Certifications
0

JDMFs Filed
Other Certificates
Other Suppliers
0

USA (Orange Book)

Europe

Canada
0

Australia
0

South Africa
Uploaded Dossiers
U.S. Medicaid
Annual Reports
0
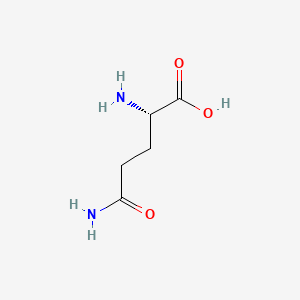

1. D Glutamine
2. D-glutamine
3. L Glutamine
4. L-glutamine
1. L-glutamine
2. 56-85-9
3. Levoglutamide
4. L-(+)-glutamine
5. Glutamic Acid Amide
6. H-gln-oh
7. Stimulina
8. Cebrogen
9. (s)-2,5-diamino-5-oxopentanoic Acid
10. Glumin
11. Levoglutamid
12. Glavamin
13. 2-aminoglutaramic Acid
14. Glutamic Acid 5-amide
15. L-glutamide
16. Miglu-p
17. Nutrestore
18. Polyglutamine
19. Saforis
20. L-2-aminoglutaramidic Acid
21. L-glutamic Acid Gamma-amide
22. L-glutamin
23. Glumin (amino Acid)
24. L-glutamic Acid 5-amide
25. (2s)-2-amino-4-carbamoylbutanoic Acid
26. Glutamine (van)
27. L-2-aminoglutaramic Acid
28. (2s)-2,5-diamino-5-oxopentanoic Acid
29. Fema No. 3684
30. Levoglutamida
31. Levoglutamidum
32. 2-aminoglutaramic Acid, L-
33. Glutamine, L-
34. Glutamine [usan]
35. Levoglutamidum [inn-latin]
36. Levoglutamida [inn-spanish]
37. Levoglutamina
38. Pentanoic Acid, 2,5-diamino-5-oxo-, (s)-
39. L-glutamid
40. 2,5-diamino-5-oxopentanoic Acid, (s)-
41. Ai3-24392
42. 26700-71-0
43. Nsc 27421
44. Brn 1723797
45. L-glutaminsaeure-5-amid
46. Mfcd00008044
47. Chembl930
48. Chebi:18050
49. D(-)-glutamine
50. 0rh81l854j
51. Nsc-27421
52. Gln
53. L-gln
54. L-glutamine-13c5
55. Glutamine (d)
56. Glutamine (l-glutamine)
57. Poly(glutamine)
58. Levoglutamide [dcf:inn]
59. Gamma-glutamine
60. 184161-19-1
61. (2s)-2,5-diamino-5-oxopentanoate
62. L-glutamine [jan]
63. Glutamine (l)
64. Einecs 200-292-1
65. L-glutamic Acid .gamma.-amide
66. Glutamine [usan:usp:inn]
67. Laevo-glutamine
68. Unii-0rh81l854j
69. Ccris 9428
70. 3h-l-glutamine
71. Hsdb 8165
72. 1wdn
73. (s)-glutamine
74. [3h]glutamine
75. Nutrestore (tn)
76. S(+)glutamine
77. Glutamine (usp)
78. [14c]glutamine
79. [3h]-glutamine
80. L-glutamine Powder
81. Cebrogen, Stimulina
82. L-glutamine,(s)
83. S(+)-glutamine
84. [14c]-glutamine
85. H-gln
86. Endari (tn)
87. Endari
88. L-alanyl-l-glutamide
89. Spectrum_000131
90. L-glutamine [jan]
91. Starbld0006818
92. Glutamine [inn]
93. Specplus_000380
94. Glutamine [mi]
95. L-glutamine (jp17)
96. L-glutamine, Homopolymer
97. Glutamine [inci]
98. L-glutamine-[13c5]
99. Spectrum2_001377
100. Spectrum3_001416
101. Spectrum4_001709
102. Spectrum5_000418
103. Glutamine [vandf]
104. L-glutamine, 98.5%
105. Bmse000038
106. Bmse001014
107. Glutamine [mart.]
108. L-glutamine [fcc]
109. Glutamine [usp-rs]
110. L-glutamine [fhfi]
111. Schembl7453
112. L-glutamine [vandf]
113. Lopac0_000549
114. Bspbio_003092
115. Gtpl723
116. Kbiogr_002038
117. Kbioss_000591
118. 4-04-00-03038 (beilstein Handbook Reference)
119. Divk1c_006476
120. Spectrum1500987
121. N-(2)-l-alanyl-l-glutamine
122. S(+)-glutamic Acid 5-amide
123. Spbio_001334
124. L-[3,4-3h(n)]glutamine
125. Levoglutamide [who-dd]
126. Gtpl4633
127. Gtpl4634
128. Glutamine [orange Book]
129. Dtxsid1023100
130. Schembl19240116
131. Schembl23124227
132. Bdbm18121
133. Kbio1_001420
134. Kbio2_000591
135. Kbio2_003159
136. Kbio2_005727
137. Kbio3_002312
138. L-glutamine, Cell Culture Grade
139. Glutamine [usp Monograph]
140. L-glutamine [orange Book]
141. Hms3261n19
142. Hms3264c03
143. Pharmakon1600-01300018
144. Pharmakon1600-01500987
145. (s)-2,5-diamino-5-oxopentanoate
146. Hy-n0390
147. Zinc1532526
148. (2s)-2-amino-4-carbamoylbutanoate
149. Tox21_500549
150. Ccg-38853
151. Nsc759628
152. Nsc760081
153. S1749
154. Akos015854078
155. (s)-2-amino-4-carbamoyl-butyric Acid
156. Am81759
157. Cs-1947
158. Db00130
159. Lp00549
160. Nsc-759628
161. Nsc-760081
162. Sdccgmls-0066691.p001
163. Sdccgsbi-0050532.p005
164. Ncgc00093936-01
165. Ncgc00093936-02
166. Ncgc00093936-03
167. Ncgc00093936-04
168. Ncgc00093936-05
169. Ncgc00093936-15
170. Ncgc00261234-01
171. (2s)-2,5-diamino-5-oxo-pentanoic Acid
172. As-11765
173. Bp-13284
174. Sbi-0050532.p003
175. L-glutamine, Bioultra, >=99.5% (nt)
176. Eu-0100549
177. G0063
178. L-glutamine, Saj Special Grade, >=99.0%
179. C00064
180. D00015
181. D70833
182. G 3126
183. M02960
184. Ab00173347-03
185. Ab00173347_04
186. L-glutamine, Reagentplus(r), >=99% (hplc)
187. L-glutamine, Vetec(tm) Reagent Grade, >=99%
188. 008g044
189. A831906
190. A937790
191. L-glutamine, Cell Culture Reagent (h-l-gln-oh)
192. Q181619
193. 7fba778c-d6b8-495c-bfe7-1cb8ec4abeab
194. J-521645
195. Q-100459
196. Brd-k83896451-001-01-8
197. F0001-1471
198. L-glutamine, Certified Reference Material, Tracecert(r)
199. Z1250208676
200. (s)-2,5-diamino-5-oxopentanoic Acid, L-glutamic Acid 5-amide
201. Glutamine, United States Pharmacopeia (usp) Reference Standard
202. L-glutamine, Gamma-irradiated, Bioxtra, Suitable For Cell Culture
203. L-glutamine, Pharmaceutical Secondary Standard; Certified Reference Material
204. L-glutamine Solution 200 Mm, 29.23 Mg/ml In Saline, Solution, Suitable For Cell Culture
205. L-glutamine Solution, 200 Mm, Hybri-max(tm), Sterile-filtered, Suitable For Hybridoma
206. L-glutamine Solution, 200 Mm, Solution, Sterile-filtered, Bioxtra, Suitable For Cell Culture
207. L-glutamine, Meets Usp Testing Specifications, Cell Culture Tested, 99.0-101.0%, From Non-animal Source
208. L-glutamine, Pharmagrade, Ajinomoto, Usp, Manufactured Under Appropriate Gmp Controls For Pharma Or Biopharmaceutical Production, Suitable For Cell Culture
| Molecular Weight | 146.14 g/mol |
|---|---|
| Molecular Formula | C5H10N2O3 |
| XLogP3 | -3.1 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 146.06914219 g/mol |
| Monoisotopic Mass | 146.06914219 g/mol |
| Topological Polar Surface Area | 106 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 146 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Nutrestore |
| PubMed Health | L-glutamine (By mouth) |
| Drug Classes | Amino Acid Supplement, Gastrointestinal Agent |
| Drug Label | NutreStore (L-glutamine powder for oral solution) for oral administration is formulated as a white crystalline powder in a paper-foil-plastic laminate packet. Each packet of NutreStore contains 5 g of L-glutamine. The amino acid glutamine is also kno... |
| Active Ingredient | Glutamine |
| Dosage Form | For solution |
| Route | Oral |
| Strength | 5gm/packet |
| Market Status | Prescription |
| Company | Emmaus Medcl |
| 2 of 2 | |
|---|---|
| Drug Name | Nutrestore |
| PubMed Health | L-glutamine (By mouth) |
| Drug Classes | Amino Acid Supplement, Gastrointestinal Agent |
| Drug Label | NutreStore (L-glutamine powder for oral solution) for oral administration is formulated as a white crystalline powder in a paper-foil-plastic laminate packet. Each packet of NutreStore contains 5 g of L-glutamine. The amino acid glutamine is also kno... |
| Active Ingredient | Glutamine |
| Dosage Form | For solution |
| Route | Oral |
| Strength | 5gm/packet |
| Market Status | Prescription |
| Company | Emmaus Medcl |
EXPL THER Glutamine depletion has negative effects on the functional integrity of the gut and leads to immunosuppression. Very low birth weight (VLBW) infants are susceptible to glutamine depletion, as enteral nutrition is limited in the first weeks of life. Enteral glutamine supplementation may have a positive effect on feeding tolerance, infectious morbidity and short-term outcome. The aim of the study was to determine the effect of enteral glutamine supplementation on plasma amino acid concentrations, reflecting one aspect of safety of enteral glutamine supplementation in VLBW infants. In a double-blind placebo-controlled randomized controlled trial, VLBW infants (gestational age <32 weeks or birth weight <1500 g) received enteral glutamine supplementation (0.3 g/kg per day) or isonitrogenous placebo supplementation (alanine) between day 3 and day 30 of life. Supplementation was added to breast milk or to preterm formula. Plasma amino acid concentrations were measured at four time points: before the start of the study and at days 7, 14 and 30 of life.Baseline patient and nutritional characteristics were not different in glutamine (n = 52) and control (n = 50) groups. Plasma concentrations of most essential and non-essential amino acids increased throughout the study period. There was no effect of enteral glutamine supplementation. In particular, the increase of plasma glutamine and glutamate concentrations was not different between the treatment groups (P = 0.49 and P = 0.34 respectively, day 30).Enteral glutamine supplementation in VLBW infants does not alter plasma concentrations of glutamine, glutamate or other amino acids. Enteral supplementation in a dose of 0.3 g/kg per day seems safe in VLBW infants.
van den Berg A et al; J Pediatr Gastroenterol Nutr. 41(1): 66-71(2005)
EXPL THER Critically ill patients have considerable oxidative stress. Glutamine and antioxidant supplementation may offer therapeutic benefit, although current data are conflicting.In this blinded 2-by-2 factorial trial, we randomly assigned 1223 critically ill adults in 40 intensive care units (ICUs) in Canada, the United States, and Europe who had multiorgan failure and were receiving mechanical ventilation to receive supplements of glutamine, antioxidants, both, or placebo. Supplements were started within 24 hours after admission to the ICU and were provided both intravenously and enterally. The primary outcome was 28-day mortality. Because of the interim-analysis plan, a P value of less than 0.044 at the final analysis was considered to indicate statistical significance. There was a trend toward increased mortality at 28 days among patients who received glutamine as compared with those who did not receive glutamine (32.4% vs. 27.2%; adjusted odds ratio, 1.28; 95% confidence interval [CI], 1.00 to 1.64; P=0.05). In-hospital mortality and mortality at 6 months were significantly higher among those who received glutamine than among those who did not. Glutamine had no effect on rates of organ failure or infectious complications. Antioxidants had no effect on 28-day mortality (30.8%, vs. 28.8% with no antioxidants; adjusted odds ratio, 1.09; 95% CI, 0.86 to 1.40; P=0.48) or any other secondary end point. There were no differences among the groups with respect to serious adverse events (P=0.83). Early provision of glutamine or antioxidants did not improve clinical outcomes, and glutamine was associated with an increase in mortality among critically ill patients with multiorgan failure.
Heyland D et al; N Engl J Med. 368 (16): 1489-97 (2013)
NutreStore (L-glutamine powder for oral solution) is indicated for the treatment of Short Bowel Syndrome (SBS) in patients receiving specialized nutritional support when used in conjunction with a recombinant human growth hormone that is approved for this indication. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
The safety and effectiveness of L-glutamine in pediatric patients have not been established.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
It is not known whether L-glutamine is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when L-glutamine is administered to a nursing woman.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
Glutamine is metabolized to glutamate and ammonia, which may increase in patients with hepatic dysfunction. Therefore, routine monitoring of renal and hepatic function is recommended in patients receiving intravenous parenteral nutrition (IPN) and NutreStore, particularly in those with renal or hepatic impairment.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
FDA Pregnancy Risk Category: C /RISK CANNOT BE RULED OUT. Adequate, well controlled human studies are lacking, and animal studies have shown risk to the fetus or are lacking as well. There is a chance of fetal harm if the drug is given during pregnancy; but the potential benefits may outweigh the potential risk./
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
For more Drug Warnings (Complete) data for Glutamine (7 total), please visit the HSDB record page.
Used for nutritional supplementation, also for treating dietary shortage or imbalance. Used to reduce the acute complications of sickle cell disease in adult and pediatric patients 5 years of age and older.
FDA Label
Like other amino acids, glutamine is biochemically important as a constituent of proteins. Glutamine is also crucial in nitrogen metabolism. Ammonia (formed by nitrogen fixation) is assimilated into organic compounds by converting glutamic acid to glutamine. The enzyme which accomplishes this is called glutamine synthetase. Glutamine can then be used as a nitrogen donor in the biosynthesis of many compounds, including other amino acids, purines, and pyrimidines. L-glutamine improves nicotinamide adenine dinucleotide (NAD) redox potential.
A - Alimentary tract and metabolism
A16 - Other alimentary tract and metabolism products
A16A - Other alimentary tract and metabolism products
A16AA - Amino acids and derivatives
A16AA03 - Glutamine
Absorption
Absorption is efficient and occurs by an active transport mechanism. Tmax is 30 minutes after a single dose. Absorption kinetics following multiple doses has not yet been determined.
Route of Elimination
Primarily eliminated by metabolism. While L-glutamine is filtered though the glomerulus, nearly all is reabsorbed by renal tubules.
Volume of Distribution
Volume of distribution is 200 mL/kg after intravenous bolus dose.
After an intravenous bolus dose in three subjects, the volume of distribution was estimated to be approximately 200 mL/kg.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
Following single dose oral administration of glutamine at 0.1 g/kg to six subjects, mean peak blood glutamine concentration was 1028uM (or 150 mcg/mL) occurring approximately 30 minutes after administration. The pharmacokinetics following multiple oral doses have not been adequately characterized.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
Metabolism is the major route of elimination for glutamine. Although glutamine is eliminated by glomerular filtration, it is almost completely reabsorbed by the renal tubules.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
Exogenous L-glutamine likely follows the same metabolic pathways as endogenous L-glutamine which is involved in the formation of glutamate, proteins, nucleotides, and amino acid sugars.
Glutamine plays an important role in nitrogen homeostasis and intestinal substrate supply. It has been suggested that glutamine is a precursor for arginine through an intestinal-renal pathway involving inter-organ transport of citrulline. The importance of intestinal glutamine metabolism for endogenous arginine synthesis in humans, however, has remained unaddressed. The aim of this study was to investigate the intestinal conversion of glutamine to citrulline and the effect of the liver on splanchnic citrulline metabolism in humans. Eight patients undergoing upper gastrointestinal surgery received a primed continuous intravenous infusion of [2-(15)N]glutamine and [ureido-(13)C-(2)H(2)]citrulline. Arterial, portal venous and hepatic venous blood were sampled and portal and hepatic blood flows were measured. Organ specific amino acid uptake (disposal), production and net balance, as well as whole body rates of plasma appearance were calculated according to established methods. The intestines consumed glutamine at a rate that was dependent on glutamine supply. Approximately 13% of glutamine taken up by the intestines was converted to citrulline. Quantitatively glutamine was the only important precursor for intestinal citrulline release. Both glutamine and citrulline were consumed and produced by the liver, but net hepatic flux of both amino acids was not significantly different from zero. Plasma glutamine was the precursor of 80% of plasma citrulline and plasma citrulline in turn was the precursor of 10% of plasma arginine. In conclusion, glutamine is an important precursor for the synthesis of arginine after intestinal conversion to citrulline in humans.
PMID:17347276 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2075174 van de Poll MC et al; J Physiol 581 (Pt 2): 819-27 (2007)
Endogenous glutamine participates in various metabolic activities, including the formation of glutamate, and synthesis of proteins, nucleotides, and amino sugars. Exogenous glutamine is anticipated to undergo similar metabolism.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
The half life of elimination is 1 h.
After an IV bolus dose in three subjects, the terminal half-life of glutamine was approximately 1 hour.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544
Supplemental L-glutamine's possible immunomodulatory role may be accounted for in a number of ways. L-glutamine appears to play a major role in protecting the integrity of the gastrointestinal tract and, in particular, the large intestine. During catabolic states, the integrity of the intestinal mucosa may be compromised with consequent increased intestinal permeability and translocation of Gram-negative bacteria from the large intestine into the body. The demand for L-glutamine by the intestine, as well as by cells such as lymphocytes, appears to be much greater than that supplied by skeletal muscle, the major storage tissue for L-glutamine. L-glutamine is the preferred respiratory fuel for enterocytes, colonocytes and lymphocytes. Therefore, supplying supplemental L-glutamine under these conditions may do a number of things. For one, it may reverse the catabolic state by sparing skeletal muscle L-glutamine. It also may inhibit translocation of Gram-negative bacteria from the large intestine. L-glutamine helps maintain secretory IgA, which functions primarily by preventing the attachment of bacteria to mucosal cells. L-glutamine appears to be required to support the proliferation of mitogen-stimulated lymphocytes, as well as the production of interleukin-2 (IL-2) and interferon-gamma (IFN-gamma). It is also required for the maintenance of lymphokine-activated killer cells (LAK). L-glutamine can enhance phagocytosis by neutrophils and monocytes. It can lead to an increased synthesis of glutathione in the intestine, which may also play a role in maintaining the integrity of the intestinal mucosa by ameliorating oxidative stress. The exact mechanism of the possible immunomodulatory action of supplemental L-glutamine, however, remains unclear. It is conceivable that the major effect of L-glutamine occurs at the level of the intestine. Perhaps enteral L-glutamine acts directly on intestine-associated lymphoid tissue and stimulates overall immune function by that mechanism, without passing beyond the splanchnic bed. The exact mechanism of L-glutamine's effect on NAD redox potential is unknown but is thought to involve increased amounts of reduced glutathione made available by glutamine supplementation. This improvement in redox potential reduces the amount of oxidative damage which sickle red blood cells are more susceptible to. The reduction in cellular damage is thought to reduce chronic hemolysis and vaso-occlusive events.
L-glutamine has important functions in regulation of gastrointestinal cell growth, function, and regeneration. Under normal conditions, glutamine concentration is maintained in the body by dietary intake and synthesis from endogenous glutamate. Data from clinical studies indicate that the role of and nutritional requirements for glutamine during catabolic illness, trauma, and infection may differ significantly from the role of and nutritional requirements for glutamine in healthy individuals. Glutamine concentrations decrease and tissue glutamine metabolism increases during many catabolic disease states, and thus glutamine is often considered a "conditionally essential" amino acid.
US Natl Inst Health; DailyMed. Current Medication Information for NUTRESTORE (glutamine) powder, for solution (July 2014). Available from, as of August 20, 2014: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=cd3fb572-c5b1-43da-aea2-31208985f544


