
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
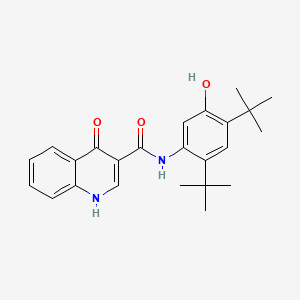

1. 3-quinolinecarboxamide, N-(2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl)-1,4-dihydro-4-oxo-
2. Kalydeco
3. N-(2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl)-4-oxo-1,4-dihydroquinoline-3-carboxamide
4. N-(2,4-di-tert-butyl-5-hydroxyphenyl)-4-oxo-1,4-dihydroquinoline-3-carboxamide
5. Vx-770
1. 873054-44-5
2. Vx-770
3. Kalydeco
4. Ivacaftor (vx-770)
5. N-(2,4-di-tert-butyl-5-hydroxyphenyl)-4-oxo-1,4-dihydroquinoline-3-carboxamide
6. Vx 770
7. N-[2,4-bis(tert-butyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxo-3-quinolinecarboxamide
8. Chebi:66901
9. N-(2,4-ditert-butyl-5-hydroxyphenyl)-4-oxo-1h-quinoline-3-carboxamide
10. 1y740ill1z
11. 1413431-05-6
12. 3-quinolinecarboxamide, N-(2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl)-1,4-dihydro-4-oxo-
13. N-(2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl)-4-oxo-1,4-dihydroquinoline-3-carboxamide
14. Vx770
15. 3-quinolinecarboxamide, N-[2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxo-
16. Kalydeco (tn)
17. Ivacaftor [usan]
18. Ivacaftor [usan:inn]
19. Ivacaftorum
20. Unii-1y740ill1z
21. Ivacaftor D18
22. Vx7
23. Ivacaftor [inn]
24. Ivacaftor [mi]
25. Ivacaftor (usan/inn)
26. Vx-770 - Ivacaftor
27. Ivacaftor [vandf]
28. Ivacaftor [who-dd]
29. Mls006011119
30. Schembl351373
31. Gtpl4342
32. Ivacaftor [orange Book]
33. Vx-770, Ivacaftor, Kalydeco
34. Chembl2010601
35. Dtxsid00236281
36. Ex-a441
37. Orkambi Component Ivacaftor
38. Symkevi Component Ivacaftor
39. Bcpp000199
40. Hms3654e10
41. Hms3744k05
42. Trikafta Component Ivacaftor
43. Bcp19794
44. Bdbm50032693
45. Mfcd17171361
46. S1144
47. Zinc52509463
48. Ivacaftor Component Of Orkambi
49. Akos015994762
50. Akos032950001
51. Ivacaftor Component Of Trikafta
52. Bcp9000799
53. Ccg-268562
54. Cs-0497
55. Db08820
56. Ex-7211
57. Le-0002
58. Sb16815
59. Ncgc00242480-01
60. Ncgc00242480-03
61. Ac-28324
62. Hy-13017
63. Smr004702900
64. Ft-0696681
65. Sw219620-1
66. Ec-000.2478
67. A25626
68. D09916
69. Ab01565806_02
70. Q6095693
71. Ctp-656; Ctp-656; Ctp-656; D9-ivacaftor;vx-561
72. Cystic Fibrosis Transmembrane Conductance Regulator Potentiator
73. N-(5-hydroxy-2,4-ditert-butyl-phenyl)-4-oxo-1h-quinoline-3-carboxamide
74. N-(2,4-ditert-butyl-5-hydroxyphenyl)-4-oxo-1,4-dihydroquinoline-3-carboxamide
75. N-(5-hydroxy-2,4-bis(2-methyl-2-propanyl)phenyl]-4-oxo-1,4-dihydro-3-quinolinecarboxamide
76. N-[2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxoquinoline-3-carboxamide
77. 1134822-00-6
78. Ivacaftor;n-(2,4-di-tert-butyl-5-hydroxyphenyl)-4-oxo-1,4-dihydroquinoline-3-carboxamide;ivacaftor
| Molecular Weight | 392.5 g/mol |
|---|---|
| Molecular Formula | C24H28N2O3 |
| XLogP3 | 5.6 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 392.20999276 g/mol |
| Monoisotopic Mass | 392.20999276 g/mol |
| Topological Polar Surface Area | 78.4 Ų |
| Heavy Atom Count | 29 |
| Formal Charge | 0 |
| Complexity | 671 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Kalydeco |
| PubMed Health | Ivacaftor (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | The active ingredient in KALYDECO tablets is ivacaftor, which has the following chemical name: N-(2,4-di-tert-butyl-5-hydroxyphenyl)-1,4-dihydro-4-oxoquinoline-3-carboxamide. Its molecular formula is C24H28N2O3 and its molecular weight is 392.49. Iva... |
| Active Ingredient | Ivacaftor |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 150mg |
| Market Status | Prescription |
| Company | Vertex Pharms |
| 2 of 2 | |
|---|---|
| Drug Name | Kalydeco |
| PubMed Health | Ivacaftor (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | The active ingredient in KALYDECO tablets is ivacaftor, which has the following chemical name: N-(2,4-di-tert-butyl-5-hydroxyphenyl)-1,4-dihydro-4-oxoquinoline-3-carboxamide. Its molecular formula is C24H28N2O3 and its molecular weight is 392.49. Iva... |
| Active Ingredient | Ivacaftor |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 150mg |
| Market Status | Prescription |
| Company | Vertex Pharms |
When used as monotherapy as the product Kalydeco, ivacaftor is indicated for the management of CF in patients age 2 years and older who have a mutation in the CFTR gene that is responsive to ivacaftor potentiation. Ivacaftor received expanded approval in May 2017 for the following 33 CFTR mutations: E56K, P67L, R74W, D110E, D110H, R117C, R117H, G178R, E193K, L206W, R347H, R352Q, A455E, S549N, S549R, G551D, G551S, D579G, S945L, S977F, F1052V, K1060T, A1067T, G1069R, R1070Q, R1070W, F1074L, D1152H, G1244E, S1251N, S1255P, D1270N, and G1349D. When used in combination with the drug [lumacaftor] as the product Orkambi, ivacaftor is indicated for the management of CF patients age 6 years and older who are shown to be homozygous for the F508del mutation in the CFTR gene. When used in combination with [tezacaftor] in the product Symdeko, it is used to manage CF in patients 12 years and older who have at least one mutation in the CFTR gene or patients aged 12 or older who are shown to be homozygous for the F508del mutation. When used in combination with tezacaftor and [elexacaftor] in the product Trikafta, it is indicated for the treatment of cystic fibrosis in patients 12 years of age and older who have at least one _F508del_ mutation in the CFTR gene.
FDA Label
Kalydeco tablets are indicated:
- As monotherapy for the treatment of adults, adolescents, and children aged 6 years and older and weighing 25 kg or more with cystic fibrosis (CF) who have an R117H CFTR mutation or one of the following gating (class III) mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene: G551D, G1244E, G1349D, G178R, G551S, S1251N, S1255P, S549N or S549R (see sections 4. 4 and 5. 1).
- In a combination regimen with tezacaftor/ivacaftor tablets for the treatment of adults, adolescents, and children aged 6 years and older with cystic fibrosis (CF) who are homozygous for the F508del mutation or who are heterozygous for the F508del mutation and have one of the following mutations in the CFTR gene: P67L, R117C, L206W, R352Q, A455E, D579G, 711+3AG, S945L, S977F, R1070W, D1152H, 2789+5GA, 3272 26AG, and 3849+10kbCT.
- In a combination regimen with ivacaftor/tezacaftor/elexacaftor tablets for the treatment of adults, adolescents, and children aged 6 years and older with cystic fibrosis (CF) who have at least one F508del mutation in the CFTR gene (see section 5. 1).
Kalydeco granules are indicated for the treatment of infants aged at least 4 months, toddlers and children weighing 5 kg to less than 25 kg with cystic fibrosis (CF) who have an R117H CFTR mutation or one of the following gating (class III) mutations in the CFTR gene: G551D, G1244E, G1349D, G178R, G551S, S1251N, S1255P, S549N or S549R (see sections 4. 4 and 5. 1).
The use of Ivacaftor has been shown to both improve CF symptoms and modulate underlying disease pathology. This is achieved by potentiating the channel opening probability (or gating) of CFTR protein in patients with impaired gating mechanisms. This is in contrast to [DB09280], another CF medication, that functions by preventing misfolding of the CFTR protein and thereby results in increased processing and trafficking of mature protein to the cell surface. Results from clinical trials indicated that treatment with ivacaftor results in improved lung function, reduced chance of experiencing a pulmonary exacerbation, reduced sweat chloride, increased weight gain, and improvements in CF symptoms and quality of life. When combined with tezacaftor, significant improvements in lung function have been observed in clinical studies. Ivacaftor was not found to increase the QTc interval in a clinically significant manner. Although ivacaftor given alone has not shown any significant improvements in patients with the delta-F508 mutation, it has shown significant improvements (>10% increase in FEV1 from baseline) in lung function for the following mutations: E56K, P67L, R74W, D110E, D110H, R117C, R117H, G178R, E193K, L206W, R347H, R352Q, A455E, S549N, S549R, G551D, G551S, D579G, S945L, S977F, F1052V, K1060T, A1067T, G1069R, R1070Q, R1070W, F1074L, D1152H, G1244E, S1251N, S1255P, D1270N, and G1349. This list was expanded by the FDA in May 2017 from 10 to 33 to accommodate more rare mutations. It is important to note that this drug may cause an increase in liver transaminases (ALT, AST). Ensure to assess liver transaminases before the initiation of treatment, every 3 months during the first year of administration, followed by every year thereafter.
Chloride Channel Agonists
A class of drugs that stimulate chloride ion influx through cell membrane channels. (See all compounds classified as Chloride Channel Agonists.)
R07AX02
R07AX30
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
R - Respiratory system
R07 - Other respiratory system products
R07A - Other respiratory system products
R07AX - Other respiratory system products
R07AX02 - Ivacaftor
Absorption
Ivacaftor is well absorbed in the gastrointestinal tract. Following administration of ivacaftor with fat-containing foods, peak plasma concentrations were reached at 4 hours (Tmax) with a maximum concentration (Cmax) of 768 ng/mL and AUC of 10600 ng * hr/mL. It is recommended that ivacaftor is taken with fat-containing foods as they increase absorption by approximately 2.5- to 4-fold.
Route of Elimination
After oral administration, ivacaftor is mainly eliminated in the feces after metabolic conversion and this elimination represents 87.8% of the dose. From the total eliminated dose, the metabolites M1 and M6 account for the majority of the eliminated dose, being 22% for M1 and 43% for M6. Ivacaftor shows negligible urinary excretion as the unchanged drug.
Volume of Distribution
After oral administration of 150 mg every 12 hours for 7 days to healthy volunteers in a fed state, the mean (SD) for apparent volume of distribution was 353 (122) L.
Clearance
The CL/F (SD) for the 150 mg dose was 17.3 (8.4) L/hr in healthy subjects.
Ivacaftor is extensively metabolized in humans. In vitro and clinical studies indicate that ivacaftor is primarily metabolized by CYP3A. From this metabolism, the major formed metabolites are M1 and M6. M1 is considered pharmacologically active even though it just presents approximately one-sixth the effect of the parent compound ivacaftor. On the other hand, M6 is not considered pharmacologically active as it represents less than one-fiftieth of the effect of the parent compound.
In a clinical study, the apparent terminal half-life was approximately 12 hours following a single dose of ivacaftor. One source mentions the half-life ranges from 12 to 14 hours.
A wide variety of CFTR mutations correlate to the Cystic Fibrosis phenotype and are associated with differing levels of disease severity. The most common mutation, affecting approximately 70% of patients with CF worldwide, is known as F508del-CFTR or delta-F508 (F508), in which a deletion in the amino acid phenylalanine at position 508 results in impaired production of the CFTR protein, thereby causing a significant reduction in the amount of ion transporter present on cell membranes. Ivacaftor as monotherapy has failed to show a benefit for patients with delta-F508 mutations, most likely due to an insufficient amount of protein available at the cell membrane for interaction and potentiation by the drug. The next most common mutation, G551D, affecting 4-5% of CF patients worldwide is characterized as a missense mutation, whereby there is sufficient amount of protein at the cell surface, but opening and closing mechanisms of the channel are altered. Ivacaftor is indicated for the management of CF in patients with this second type of mutation, as it binds to and potentiates the channel opening ability of CFTR proteins on the cell membrane. Ivacaftor exerts its effect by acting as a potentiator of the CFTR protein, an ion channel involved in the transport of chloride and sodium ions across cell membranes of the lungs, pancreas, and other organs. Alterations in the CFTR gene result in altered production, misfolding, or function of the protein and consequently abnormal fluid and ion transport across cell membranes. Ivacaftor improves CF symptoms and underlying disease pathology by potentiating the channel open probability (or gating) of CFTR protein in patients with impaired CFTR gating mechanisms. The overall level of ivacaftor-mediated CFTR chloride transport is dependent on the amount of CFTR protein at the cell surface and how responsive a particular mutant CFTR protein is to ivacaftor potentiation.
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 42858
Submission : 2025-11-01
Status : Active
Type : II

NDC Package Code : 70600-078
Start Marketing Date : 2025-10-20
End Marketing Date : 2027-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 Lupin Manufacturing Solutions – delivering high-quality APIs & end-to-end CDMO services for faster, cost-effective drug development.
Lupin Manufacturing Solutions – delivering high-quality APIs & end-to-end CDMO services for faster, cost-effective drug development.

GDUFA
DMF Review : Reviewed
Rev. Date : 2020-08-20
Pay. Date : 2020-07-02
DMF Number : 32610
Submission : 2019-03-29
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35551
Submission : 2021-01-25
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35587
Submission : 2021-02-09
Status : Active
Type : II


GDUFA
DMF Review : Reviewed
Rev. Date : 2023-10-17
Pay. Date : 2023-08-29
DMF Number : 29398
Submission : 2015-05-30
Status : Active
Type : II

Date of Issue : 2025-07-07
Valid Till : 2028-07-14
Written Confirmation Number : WC-0021
Address of the Firm :

NDC Package Code : 14501-0019
Start Marketing Date : 2015-01-13
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 42858
Submission : 2025-11-01
Status : Active
Type : II
 Lupin Manufacturing Solutions – delivering high-quality APIs & end-to-end CDMO services for faster, cost-effective drug development.
Lupin Manufacturing Solutions – delivering high-quality APIs & end-to-end CDMO services for faster, cost-effective drug development.
GDUFA
DMF Review : Complete
Rev. Date : 2020-08-20
Pay. Date : 2020-07-02
DMF Number : 32610
Submission : 2019-03-29
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2017-07-18
Pay. Date : 2017-06-28
DMF Number : 31849
Submission : 2017-06-30
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-05-19
Pay. Date : 2015-03-10
DMF Number : 28867
Submission : 2015-03-10
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35586
Submission : 2021-02-11
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-08-13
Pay. Date : 2021-07-07
DMF Number : 36094
Submission : 2021-07-07
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2023-10-17
Pay. Date : 2023-08-29
DMF Number : 29398
Submission : 2015-05-30
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35447
Submission : 2020-12-28
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2019-09-10
Pay. Date : 2019-07-25
DMF Number : 33783
Submission : 2019-07-26
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35587
Submission : 2021-02-09
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]4-oxo-l ,4-dihydroquinoline -3-carboxylic acid
CAS Number : 13593-94-7
End Use API : Ivacaftor
About The Company : Malladi Drugs & Pharmaceuticals Ltd, founded in 1980 by Mr. M L N Sastry, is a leader in manufacturing ephedrine and pseudoephedrine salts. It is now the top AP...
4-oxo-1,4-dihydroquinoline-3-carboxylic acid
CAS Number : 13721-01-2
End Use API : Ivacaftor
About The Company : Operational since 2024, ApiSyn Healthcare Pvt. Ltd. is a rapidly growing cGMP manufacturer and CDMO specializing in APIs, intermediates, peptides, and amino aci...
5-amino-2,4-di-tert-butylphenyl methyl carbonate
CAS Number : 1182822-31-6
End Use API : Ivacaftor
About The Company : Operational since 2024, ApiSyn Healthcare Pvt. Ltd. is a rapidly growing cGMP manufacturer and CDMO specializing in APIs, intermediates, peptides, and amino aci...
4-oxo-1,4-dihydroquinoline-3-carboxylic acid
CAS Number : 13721-01-2
End Use API : Ivacaftor
About The Company : We are into manufacturing of bulk drug Intermediates and Active Pharmaceutical Ingredients (API). Our core competence is contract manufacturing for our clients ...

5-Amino-2,4-ditert-butylphenol
CAS Number : 873055-58-4
End Use API : Ivacaftor
About The Company : We are into manufacturing of bulk drug Intermediates and Active Pharmaceutical Ingredients (API). Our core competence is contract manufacturing for our clients ...

CAS Number : 87-51-4
End Use API : Ivacaftor
About The Company : Blue Jet Healthcare is a global, science-driven pharmaceutical company specializing in collaboration, development, and manufacturing of advanced pharmaceutical ...

2,4-Di-tert-butyl-5 -nitrophenol
CAS Number : 873055-57-3
End Use API : Ivacaftor
About The Company : Established in 2011 and situated in Hangzhou, Zhejiang, China, Hangzhou Longshine Bio-Tech CO., Ltd is dedicated to providing services for pharmaceutical and ch...

4-Oxo-1,4-dihydroquinoline-3-carboxylic acid
CAS Number : 13721-01-2
End Use API : Ivacaftor
About The Company : Established in 2011 and situated in Hangzhou, Zhejiang, China, Hangzhou Longshine Bio-Tech CO., Ltd is dedicated to providing services for pharmaceutical and ch...

4-Oxo-1,4-dihydroquinoline-3-carboxylic
CAS Number : 13721-01-2
End Use API : Ivacaftor
About The Company : Soujanya Life Sciences is a new division of the Soujanya Group, a 40-year-old organization with a strong presence in 50 countries and expertise in pigments and ...

5-Amino-2,4-di-tert-butyl-phenol
CAS Number : 873055-58-4
End Use API : Ivacaftor
About The Company : VIDGAS is dedicated to delivering the highest quality SFC and HPLC purification and isolation solutions to clients across the pharmaceutical, biopharmaceutical,...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Dosage Form : Tablet
Grade : Oral
Application : Controlled & Modified Release
Excipient Details : ACTILLETS are microcrystalline cellulose spheres developed using advanced drug delivery technology to enable effective loading & coating of particles.
Pharmacopoeia Ref : NA
Technical Specs : Not Available
Ingredient(s) : Microcrystalline Cellulose Excipients
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Disintegrants & Superdisintegrants
Excipient Details : Mannogem 2080 exhibits excellent flow, disintegration and compression properties.
Dosage Form : Orodispersible Tablet
Grade : Oral
Dosage Form : Orodispersible Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
46
PharmaCompass offers a list of Ivacaftor API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ivacaftor manufacturer or Ivacaftor supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ivacaftor manufacturer or Ivacaftor supplier.
A Ivacaftor manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ivacaftor, including repackagers and relabelers. The FDA regulates Ivacaftor manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ivacaftor API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ivacaftor manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Ivacaftor supplier is an individual or a company that provides Ivacaftor active pharmaceutical ingredient (API) or Ivacaftor finished formulations upon request. The Ivacaftor suppliers may include Ivacaftor API manufacturers, exporters, distributors and traders.
click here to find a list of Ivacaftor suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Ivacaftor DMF (Drug Master File) is a document detailing the whole manufacturing process of Ivacaftor active pharmaceutical ingredient (API) in detail. Different forms of Ivacaftor DMFs exist exist since differing nations have different regulations, such as Ivacaftor USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Ivacaftor DMF submitted to regulatory agencies in the US is known as a USDMF. Ivacaftor USDMF includes data on Ivacaftor's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Ivacaftor USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Ivacaftor suppliers with USDMF on PharmaCompass.
A Ivacaftor written confirmation (Ivacaftor WC) is an official document issued by a regulatory agency to a Ivacaftor manufacturer, verifying that the manufacturing facility of a Ivacaftor active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Ivacaftor APIs or Ivacaftor finished pharmaceutical products to another nation, regulatory agencies frequently require a Ivacaftor WC (written confirmation) as part of the regulatory process.
click here to find a list of Ivacaftor suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Ivacaftor as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Ivacaftor API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Ivacaftor as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Ivacaftor and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Ivacaftor NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Ivacaftor suppliers with NDC on PharmaCompass.
Ivacaftor Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ivacaftor GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ivacaftor GMP manufacturer or Ivacaftor GMP API supplier for your needs.
A Ivacaftor CoA (Certificate of Analysis) is a formal document that attests to Ivacaftor's compliance with Ivacaftor specifications and serves as a tool for batch-level quality control.
Ivacaftor CoA mostly includes findings from lab analyses of a specific batch. For each Ivacaftor CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ivacaftor may be tested according to a variety of international standards, such as European Pharmacopoeia (Ivacaftor EP), Ivacaftor JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ivacaftor USP).

