
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
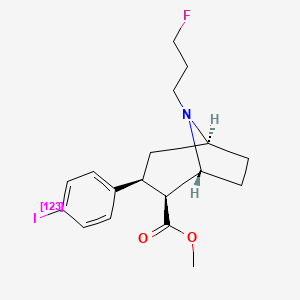

1. (18f)fp-cit
2. (o-methyl-11c)beta-cit-fp
3. 123i-fp-cit
4. 2-carbomethoxy-8-(3-fluoropropyl)-3-(4-iodophenyl)tropane
5. Beta-cit-fp
6. Fluoropropylcarbomethoxyiodophenylnortropane
7. Fp-cit
8. Fpcit
9. I(123)-n-fluoropropyl-2-beta-carbomethoxy-3-(4-iodophenyl)nortropane
10. I(123)-n-omega-fluoropropyl-2-beta-carbomethoxy-3-beta(4-iodophenyl)nortropane
11. N-omega-fluoropropyl-2beta-carbomethoxy-3beta-(4-iodophenyl)tropane
12. (1r,2s,3s,5s)-8-(3-fluoropropyl)-3-(4-iodophenyl)-8-azabicyclo(3,2,1)octane-2-carboxylic Acid Methyl Ester
13. 123i-ioflupane
14. Ioflupane
15. Rti 313
16. Rti-313
1. Ioflupane I 123
2. Ioflupane (123i)
3. 123i-ioflupane
4. Datscan
5. 123i-fp-cit
6. Ioflupane I(123)
7. V09ab03
8. Ioflupane ((123)i)
9. Ioflupane
10. V-09ab03
11. (123i)fp-cit
12. 155798-07-5
13. Ioflupane (123l)
14. Ioflupane, I-123
15. Fp-cit I-123
16. Ioflupane (sup 123)i
17. Iodine Ioflupane (123i)
18. Ioflupane (123i) [inn]
19. 3mm99t8r5q
20. Ioflupane I 123 [usan]
21. Methyl (1r,2s,3s,5s)-8-(3-fluoropropyl)-3-[4-((123)i)iodophenyl]-8-azabicyclo[3.2.1]octane-2-carboxylate
22. Methyl 8-(3-fluoropropyl)-3beta-(p-iodo-i(sup 123)-phenyl)-1alphah, 5alphah-nortropane-2beta-carboxylate
23. 123i-beta-cit-fp)
24. Unii-3mm99t8r5q
25. Ioflupane I-123 And Iodine
26. V 09ab03
27. Ioflupane I 123 [usan:inn:ban]
28. Ioflupano (123i)
29. Ioflupanum (123i)
30. Ioflupane ((sup 123)i)
31. Gtpl7653
32. Chembl3989517
33. Schembl16472968
34. Chebi:68855
35. Dtxsid90935291
36. Ioflupane I 123 [mi]
37. Ioflupane I-123 [vandf]
38. Ioflupane (123i) [who-dd]
39. Db08824
40. Ioflupane (123l) [ema Epar]
41. Ioflupane I-123 [orange Book]
42. Methyl (1s,3s,4s,5r)-8-(3-fluoropropyl)-3-(4-iodanylphenyl)-8-azabicyclo[3.2.1]octane-4-carboxylate
43. Ioflupane (sup 123)i [ema Epar]
44. Q3801359
45. 8-azabicyclo(3.2.1)octane-2-carboxylic Acid, 8-(3-fluoropropyl)-3-(4-(iodo-(sup 123)i)phenyl)-, Methyl Ester, (1r,2s,3s,5s)-
46. Methyl (1r,2s,3s,5s)-8-(3-fluoropropyl)-3-(4-(123i)iodanylphenyl)-8-azabicyclo[3.2.1]octane-2-carboxylate
47. Methyl 8-(3-fluoropropyl)-3.beta.-(p-iodo-(sup 123)i-phenyl)-1.alpha.h,5.alpha.h-nortropane-2.beta.-carboxylate
48. Methyl 8-(3-fluoropropyl)-3beta-(p-iodo-(sup 123)i-phenyl)-1alphah,5alphah-nortropane-2beta-carboxylate
| Molecular Weight | 427.3 g/mol |
|---|---|
| Molecular Formula | C18H23FINO2 |
| XLogP3 | 4 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Exact Mass | 427.07687 g/mol |
| Monoisotopic Mass | 427.07687 g/mol |
| Topological Polar Surface Area | 29.5 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 414 |
| Isotope Atom Count | 1 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Ioflupane I-123 is a SPECT (single photon emission computerized tomography) agent used to distinguish between Parkinsons syndrome tremors and essential tremor.
FDA Label
This medicinal product is for diagnostic use only.
Striascan is indicated for detecting loss of functional dopaminergic neuron terminals in the striatum:
- In adult patients with clinically uncertain parkinsonian syndromes, for example those with early symptoms, in order to help differentiate essential tremor from parkinsonian syndromes related to idiopathic Parkinsons disease, multiple system atrophy and progressive supranuclear palsy. Striascan is unable to discriminate between Parkinson's disease, multiple system atrophy and progressive supranuclear palsy.
- In adult patients, to help differentiate probable dementia with Lewy bodies from Alzheimers disease. Striascan is unable to discriminate between dementia with Lewy bodies and Parkinsons disease dementia.
This medicinal product is for diagnostic use only.
DaTSCAN is indicated for detecting loss of functional dopaminergic neuron terminals in the striatum:
- In adult patients with clinically uncertain Parkinsonian syndromes, for example those with early symptoms, in order to help differentiate essential tremor from Parkinsonian syndromes related to idiopathic Parkinsons disease, multiple system atrophy and progressive supranuclear palsy. DaTSCAN is unable to discriminate between Parkinson's disease, multiple system atrophy and progressive supranuclear palsy.
- In adult patients, to help differentiate probable dementia with Lewy bodies from Alzheimers disease. DaTSCAN is unable to discriminate between dementia with Lewy bodies and Parkinsons disease dementia.
This medicinal product is for diagnostic use only.
Celsunax is indicated for detecting loss of functional dopaminergic neuron terminals in the striatum:
- In adult patients with clinically uncertain parkinsonian syndromes, for example those with early symptoms, in order to help differentiate essential tremor from parkinsonian syndromes related to idiopathic Parkinsons disease, multiple system atrophy and progressive supranuclear palsy. Celsunax is unable to discriminate between Parkinson's disease, multiple system atrophy and progressive supranuclear palsy.
- In adult patients, to help differentiate probable dementia with Lewy bodies from Alzheimers disease. Celsunax is unable to discriminate between dementia with Lewy bodies and Parkinsons disease dementia.
iodine-123 labeled ioflupanebinds selectively to striatal presynaptic dopamine neurons by binding reversibly to presynaptic dopamine transporters.
Diagnostic Uses of Chemicals
Chemicals administered to patients in order to diagnose or study the pathology of medical conditions, diseases, or syndromes. (See all compounds classified as Diagnostic Uses of Chemicals.)
V09AB03
V09AB03
V09AB03
V - Various
V09 - Diagnostic radiopharmaceuticals
V09A - Central nervous system
V09AB - Iodine (123i) compounds
V09AB03 - Iodine ioflupane (123I)
Absorption
Absorption is 100% because administered I.V.
Route of Elimination
About 60% is excreted in the urine and 14% in the feces after 48 hours.
Volume of Distribution
Compared to the entire brain, about 30% of radioactivity is taken up by the striatum.
Half life is 13.2 hours.
Iodine-123 labeled ioflupane binds to presynaptic dopamine transporters. When Iodine-123 decays, a gammay ray is emmitted and detected through SPECT.
Related Excipient Companies

Excipients by Applications
ABOUT THIS PAGE
88
PharmaCompass offers a list of Ioflupane API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ioflupane manufacturer or Ioflupane supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ioflupane manufacturer or Ioflupane supplier.
A IOFLUPANE I 123 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of IOFLUPANE I 123, including repackagers and relabelers. The FDA regulates IOFLUPANE I 123 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. IOFLUPANE I 123 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A IOFLUPANE I 123 supplier is an individual or a company that provides IOFLUPANE I 123 active pharmaceutical ingredient (API) or IOFLUPANE I 123 finished formulations upon request. The IOFLUPANE I 123 suppliers may include IOFLUPANE I 123 API manufacturers, exporters, distributors and traders.
IOFLUPANE I 123 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of IOFLUPANE I 123 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right IOFLUPANE I 123 GMP manufacturer or IOFLUPANE I 123 GMP API supplier for your needs.
A IOFLUPANE I 123 CoA (Certificate of Analysis) is a formal document that attests to IOFLUPANE I 123's compliance with IOFLUPANE I 123 specifications and serves as a tool for batch-level quality control.
IOFLUPANE I 123 CoA mostly includes findings from lab analyses of a specific batch. For each IOFLUPANE I 123 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
IOFLUPANE I 123 may be tested according to a variety of international standards, such as European Pharmacopoeia (IOFLUPANE I 123 EP), IOFLUPANE I 123 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (IOFLUPANE I 123 USP).

