
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
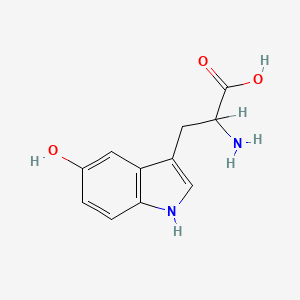

1. 5 Hydroxytryptophan
2. 5-htp
3. 5-hydroxy- Tryptophan
4. Hydroxytryptophan
5. Oxitriptan
6. Oxytryptophan
7. Tryptophan, 5 Hydroxy
8. Tryptophan, 5-hydroxy-
1. 56-69-9
2. Dl-5-hydroxytryptophan
3. 5-hydroxy-dl-tryptophan
4. 2-amino-3-(5-hydroxy-1h-indol-3-yl)propanoic Acid
5. 114-03-4
6. (+-)-5-hydroxytryptophan
7. Dl-hydroxytryptophan
8. Dl-5-htp
9. 5-htp
10. 5-hydroxytryptophan Dl-form
11. H-dl-trp(5-oh)-oh
12. D,l-5-hydroxytryptophan
13. Nsc-92523
14. (+/-)-5-hydroxytryptophan
15. 5-hydroxytryptophan (5-htp)
16. Chebi:28171
17. 9181p3oi6n
18. Mfcd00005651
19. Dl-pretonine
20. 72572-98-6
21. Dl-oxitriptan
22. 5-hydroxy Tryptophan
23. Usaf Cb-96
24. 5-hydroxytryptophan, Dl-
25. 5-hydroxytryptophan L Form
26. Hsdb 4295
27. Dl-5-hydroxytryptophane
28. Nci-c56644
29. Tryptophan, 5-hydroxy-, Dl
30. 5-22-14-00278 (beilstein Handbook Reference)
31. Einecs 200-284-8
32. Tryptophan, 5-hydroxy-, L-
33. Nsc 92523
34. Brn 0088199
35. Dl-tryptophan, 5-hydroxy-
36. Unii-9181p3oi6n
37. Mfcd00064341
38. 5-hydroxyltryptophan
39. 5-hydroxytryptophan #
40. H-l-trp(5-oh)-oh
41. 5-hydroxytryptophan(5-htp)
42. Schembl43242
43. Mls006011926
44. 2-amino-3-(5-hydroxy-1h-indol-3-yl)propanoicacid
45. (.+-.)-5-hydroxytryptophan
46. Chembl162789
47. 5-hydroxytryptophan, Aldrichcpr
48. Schembl19871232
49. 5-hydroxytryptophan [mi]
50. Dtxsid50859863
51. Hms3372g05
52. Hms3656m11
53. Hms3884h15
54. Wln: T56 Bmj D1yzvq Gq
55. 5-hydroxytryptophan [hsdb]
56. Hy-n0122
57. Nsc92523
58. Bbl012270
59. S2374
60. Stk803137
61. Akos003382640
62. Akos016050369
63. Ccg-235444
64. Ncgc00015526-02
65. Ncgc00015526-04
66. Ac-13632
67. Ac-23979
68. Smr000326745
69. Sy030569
70. Sy048412
71. Vs-03256
72. Db-051092
73. A2320
74. Cs-0007832
75. Ft-0620484
76. Ft-0620490
77. Ft-0620491
78. Ft-0620498
79. Ft-0627633
80. Sw219417-1
81. 56h699
82. C01017
83. Dl-2-amino-3-(5-hydroxyindolyl)propionic Acid
84. H-8280
85. N88462
86. Sr-01000003309
87. Q-102914
88. Sr-01000003309-1
89. Q27103541
| Molecular Weight | 220.22 g/mol |
|---|---|
| Molecular Formula | C11H12N2O3 |
| XLogP3 | -1.2 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Exact Mass | 220.08479225 g/mol |
| Monoisotopic Mass | 220.08479225 g/mol |
| Topological Polar Surface Area | 99.3 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 272 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
5-HTP has shown some usefulness in some conditions characterized, in part, by serotonin deficits, principally depression. It has also been shown to be useful in some with obesity, insomnia, fibromyalgia and chronic tension headache.
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.4 (2001)
It has been long known that brain serotonin systems contribute to the modulation of food intake and satiety. An increase of intrasynaptic serotonin tends to reduce food consumption. Thus, one might consider that individuals taking 5-HTP might experience increase satiety and weight loss over a period of time. There are few studies on the effects of 5-HTP on obesity and they suggest an anorectic effect of 5-HTP.
Coates, P.M., Blackman, M.R., Cragg, G.M., Levine, M., Moss, J., White, J.D. (Ed), Encyclopedia of Dietary Supplements. Marcel Dekker, New York, NY, p. 350 (2005)
There is some evidence that 5-HTP ... can improve postural equilibrium, dysarthria in patients with various inherited and acquired cerebellar ataxias, and particularly in those with lesions located precisely in the anterior lobe vermis. Improvements in coordination have been reported in patients with Friedreich"s ataxia; however, the effect is only partial and not clinically major.
Coates, P.M., Blackman, M.R., Cragg, G.M., Levine, M., Moss, J., White, J.D. (Ed), Encyclopedia of Dietary Supplements. Marcel Dekker, New York, NY, p. 352 (2005)
Exptl Ther: Rats of the Dahl salt-sensitive (DS) and Dahl salt-resistant (DR) strains were placed on a 4% NaCl diet and blood pressures were monitored. Chronic subcutaneous infusion L-5-hydroxytryptophan (L-5-HTP, 12.6 mg/day) by osmotic minipumps significantly decreased the elevated systolic blood pressure of DS rats on a 4% NaCl diet. Blood pressures of DR rats were unaffected by treatment with L-5-HTP. Cardiac hypertrophy was associated with Dahl salt-induced hypertension. However, treatment with L-5-HTP failed to reduce the weight of the heart significantly. These results suggest that chronic administration of L-5-HTP was effective in reducing the elevated blood pressure in the DS model. The specific mechanisms by which L-5-HTP reduces the elevated blood pressure in DS rats is not clear and remains for further study.
PMID:1829234 Baron A et al; Pharmacology 42 (1): 15-22 (1991)
For more Therapeutic Uses (Complete) data for 5-HYDROXYTRYPTOPHAN (6 total), please visit the HSDB record page.
Other reported side effects, include nausea, diarrhea, loss of appetite, vomiting and difficult breathing. Neurological side effects, including dilation of the pupils, abnormally sensitive reflexes, loss of muscle coordination and blurring of vision, have been reported in those taking large doses of 5-HTP. Cardiac dysrhythmias have also been reported.
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.5 (2001)
Eosinophilia and eosinophilia-myalgia syndrome (EMS) have been reported in those taking 5-HTP. The eosinophilia myalgia syndrome is similar to that caused by L-tryptophan and was linked to contaminants in the 5-HTP preparation, rather than 5-HTP itself. Changing the 5-HTP lot in one group of patients resolved the eosinophilia. A scleroderma-like skin condition has been reported in some taking a combination of 5-HTP and carbidopa.
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.5 (2001)
5-HTP should be avoided by pregnant women and nursing mothers.
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.5 (2001)
5-HTP should be avoided by those with ischemic heart disease (history of myocardial infarction, angina pectoris, documented silent ischemia), coronary artery spasm (e.g., Prinzmetal's angina), uncontrollable hypertension and any other significant cardiovascular disease.
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.4 (2001)
For more Drug Warnings (Complete) data for 5-HYDROXYTRYPTOPHAN (8 total), please visit the HSDB record page.
Antidepressive Agents, Second-Generation
A structurally and mechanistically diverse group of drugs that are not tricyclics or monoamine oxidase inhibitors. The most clinically important appear to act selectively on serotonergic systems, especially by inhibiting serotonin reuptake. (See all compounds classified as Antidepressive Agents, Second-Generation.)
The immediate precursor in the serotonin synthetic route, 5-hydroxytryptophan (5-HTP), labeled with 11C in the beta position, has become available for studies using positron emission tomography (PET) to examine serotonin formation in human brain. Normalized uptake and intracerebral utilization of tracer amounts of (beta-11C)5-HTP were studied twice in six healthy male volunteers, three of them before and after pharmacological pretreatments ... Pretreatments with benserazide, p-chlorophenylalanine (PCPA), and unlabeled 5-HTP all significantly increased uptake of (beta-11C)5-HTP. The utilization rates in both striatal and frontal cortex were higher than those in the surrounding brain, indicating that PET studies using (beta-11C)5-HTP as a ligand quantitate selective processes in the utilization of 5-HTP.
PMID:1292039 Reibring L et al; Psychiatry Res 45(4): 215-25 (1992)
The efficiency of absorption of 5HTP, as well as its decarboxylation product serotonin, is approximately 47% to 84%. Absorption of 5-HTP occurs by an active transport process. 5-HTP is transported by the portal circulation to the liver where approximately 25% of an administered dose is metabolized ... . 5-HTP that is not metabolized in the liver is transported by the general circulation to the various tissues of the body, including the brain. 5-HTP readily crosses the blood-brain barrier, and is converted to serotonin in brain cells.
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.4 (2001)
5-HTP is transported by the portal circulation to the liver where approximately 25% of an administered dose is metabolized via vitamin B6-dependent L-aromatic amino acid decarboxylase to 5-hydroxytryptamine (5-HT) /serotonin/. 5-HT is subsequently metabolized to 5-hydroxyindole acetaldehyde which is rapidly metabolized to 5-hydroxyindole acetaldehyde which is rapidly metabolized to 5-hydroxyindoleacetic acid (5-HIAA).
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.4 (2001)
The possible analgesic effect of 5-HTP may be accounted for, in part, by its conversion to serotonin. 5-HTP has also been found to increase plasma beta-endorphin and platelet met-enkephalin levels, which may signify a reinforcing effect upon an endogenous analgesic effect.
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.3 (2001)
The mechanism of the possible antidepressant activity of 5-HTP is accounted for by its conversion to the neurotransmitter serotonin which plays a central role in the affective state. Antidepressants may work by either binding to one or more of the family of serotonin 5-HT receptors (5-HT1 - 5-HT7) or by inhibiting the reuptake of serotonin. ...
Physicians Desk Reference (PDR) for Nutritional Supplements 1st ed, Medical Economics, Thomson Healthcare; Montvale, NJ p.3 (2001)
Market Place

ABOUT THIS PAGE
60
PharmaCompass offers a list of HSDB 4295 API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right HSDB 4295 manufacturer or HSDB 4295 supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred HSDB 4295 manufacturer or HSDB 4295 supplier.
A HSDB 4295 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of HSDB 4295, including repackagers and relabelers. The FDA regulates HSDB 4295 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. HSDB 4295 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of HSDB 4295 manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A HSDB 4295 supplier is an individual or a company that provides HSDB 4295 active pharmaceutical ingredient (API) or HSDB 4295 finished formulations upon request. The HSDB 4295 suppliers may include HSDB 4295 API manufacturers, exporters, distributors and traders.
click here to find a list of HSDB 4295 suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
HSDB 4295 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of HSDB 4295 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right HSDB 4295 GMP manufacturer or HSDB 4295 GMP API supplier for your needs.
A HSDB 4295 CoA (Certificate of Analysis) is a formal document that attests to HSDB 4295's compliance with HSDB 4295 specifications and serves as a tool for batch-level quality control.
HSDB 4295 CoA mostly includes findings from lab analyses of a specific batch. For each HSDB 4295 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
HSDB 4295 may be tested according to a variety of international standards, such as European Pharmacopoeia (HSDB 4295 EP), HSDB 4295 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (HSDB 4295 USP).

