
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Gliphosate
2. Glyphosate Hydrochloride (2:1)
3. Glyphosate, Calcium Salt
4. Glyphosate, Calcium Salt (1:1)
5. Glyphosate, Copper (2+) Salt
6. Glyphosate, Dilithium Salt
7. Glyphosate, Disodium Salt
8. Glyphosate, Magnesium Salt
9. Glyphosate, Magnesium Salt (2:1)
10. Glyphosate, Monoammonium Salt
11. Glyphosate, Monopotassium Salt
12. Glyphosate, Monosodium Salt
13. Glyphosate, Sodium Salt
14. Glyphosate, Zinc Salt
15. Kalach 360 Sl
16. N-(phosphonomethyl)glycine
17. Roundup
18. Yerbimat
1. 1071-83-6
2. N-(phosphonomethyl)glycine
3. Roundup
4. N-phosphonomethyl-glycine
5. Glyphosphate
6. 2-(phosphonomethylamino)acetic Acid
7. Glycine, N-(phosphonomethyl)-
8. N-phosphonomethylglycine
9. Glyfos
10. Pondmaster
11. N-phosphomethylglycine
12. Mon 2139
13. N-(phosphonomethyl) Glycine
14. Nsc151063
15. 2-((phosphonomethyl)amino)acetic Acid
16. Glialka
17. Glyphomax
18. Accord
19. Cp 67573
20. Mon 6000
21. Phosphonomethyliminoacetic Acid
22. 994-61-6
23. Nsc-151063
24. Chembl95764
25. Chebi:27744
26. Glyphosate 40% Aqueous Solution
27. 4632ww1x5a
28. [(phosphonomethyl)amino]acetic Acid
29. 2-[(phosphonomethyl)amino]acetic Acid
30. Silglif
31. Mon 0573
32. Roundup Max
33. Caswell No. 661a
34. 2-[(phosphonomethyl)amino]acetic Acid, 40% Aqueous Solution
35. Buccaneer
36. Folusen
37. Glifoglex
38. Herbatop
39. Hm 2028; Herbatop; Hockey; Kickdown; Klinik;nsc 151063
40. Kickdown
41. Phorsat
42. Atila
43. Forsat
44. Hockey
45. Klinik
46. Leone
47. Safal
48. ((phosphonomethyl)amino)acetic Acid
49. Gliz
50. Glygran
51. Buccaneer Plus
52. Glyphosate 100 Microg/ml In Water
53. Rebel Garden
54. Glyphodin A
55. Glyphosate Ct
56. Mfcd00055350
57. Ground Bio
58. Mon 39
59. Glyphosate [iso]
60. Lancer (herbicide)
61. Glialka 36
62. Glifosan 747
63. Glyphosate [ansi:bsi:iso]
64. Phosphonomethylglycine
65. C-k Yuyos Fav
66. Ccris 1587
67. Phosphonomethyl Glycine
68. Hsdb 3432
69. (carboxymethylamino)methylphosphonic Acid
70. Gliz 480cs
71. Cp 70139
72. Einecs 213-997-4
73. Epa Pesticide Chemical Code 417300
74. Nsc 151063
75. Brn 2045054
76. Unii-4632ww1x5a
77. N-phosphonomethyl Glycine
78. 2gga
79. 2ggd
80. N-(phosphonomethyl)-glycine
81. Gpf
82. Gpj
83. Mon 3539
84. Roundup (tn)
85. Carboxymethylaminomethanephosphinic Acid
86. Glyphosate [m+h+]
87. Hm 2028
88. Spectrum_001867
89. Glyphosate [mi]
90. Specplus_000478
91. Glyphosate-2-[13c]
92. Glyphosate-3-[13c]
93. Spectrum2_001847
94. Spectrum3_000839
95. Spectrum4_000679
96. Spectrum5_001992
97. Glyphosate [hsdb]
98. Glyphosate [iarc]
99. Wln: Qv1m1pqqo
100. Glyphosate [mart.]
101. Ncistruc1_000002
102. Ncistruc2_000104
103. Schembl16440
104. Bspbio_002397
105. Kbiogr_001117
106. Kbioss_002384
107. Spectrum330047
108. Divk1c_006574
109. Spbio_001634
110. Glyphosate, Isopropylamine Salt
111. Glyphosate-[2-13c,15n]
112. Dtxsid1024122
113. Kbio1_001518
114. Kbio2_002380
115. Kbio2_004948
116. Kbio2_007516
117. Kbio3_001897
118. Xddaorkbjwwyjs-uhfffaoysa-
119. N-(phosphonomethyl)glycine, 96%
120. Glyphosate 10 Microg/ml In Water
121. 2-(phosphonatomethylammonio)acetate
122. Amy22521
123. Bcp32091
124. Hy-b0863
125. Zinc3872713
126. 2-(phosphonomethylamino)ethanoic Acid
127. Bdbm50281349
128. Ccg-38069
129. Ncgc00014409
130. Nci151063
131. Akos004915878
132. Ks-5110
133. Ncgc00014409-02
134. Ncgc00014409-03
135. Ncgc00014409-04
136. Ncgc00094536-01
137. Ncgc00094536-02
138. Ncgc00094536-03
139. Nci60_001060
140. Cs-0012877
141. Ft-0651304
142. Glyphosate (40% In Aqueous Solution)
143. Glyphosate, Pestanal(r), Analytical Standard
144. 71g836
145. C01705
146. H10288
147. A801602
148. Q407232
149. Q-201175
150. 86491fad-2f76-4cde-90ee-6131d3fcf0bb
151. Glyphosate Isopropyl Amine Salt Aqueous Solution(41%)
152. Glyphosate, Certified Reference Material, Tracecert(r)
153. Glyphosate (n-(phosphonomethyl)glycine) 100 Microg/ml In Water
154. N-(phosphonomethyl)glycine, Plant Cell Culture Tested, Bioreagent
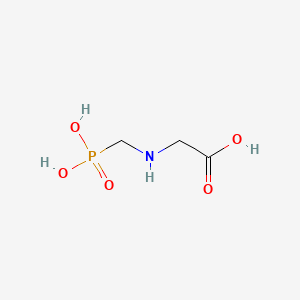
| Molecular Weight | 169.07 g/mol |
|---|---|
| Molecular Formula | C3H8NO5P |
| XLogP3 | -4.6 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Exact Mass | 169.01400935 g/mol |
| Monoisotopic Mass | 169.01400935 g/mol |
| Topological Polar Surface Area | 107 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 162 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Herbicides
Pesticides used to destroy unwanted vegetation, especially various types of weeds, grasses (POACEAE), and woody plants. Some plants develop HERBICIDE RESISTANCE. (See all compounds classified as Herbicides.)
Uncoupling Agents
Chemical agents that uncouple oxidation from phosphorylation in the metabolic cycle so that ATP synthesis does not occur. Included here are those IONOPHORES that disrupt electron transfer by short-circuiting the proton gradient across mitochondrial membranes. (See all compounds classified as Uncoupling Agents.)
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Antifungal Agents
Substances that destroy fungi by suppressing their ability to grow or reproduce. They differ from FUNGICIDES, INDUSTRIAL because they defend against fungi present in human or animal tissues. (See all compounds classified as Antifungal Agents.)
The toxicokinetics of glyphosate after single 100 mg/kg intravenous (i.v.) and 400 mg/kg oral doses were studied in rats. Serial blood samples were obtained after i.v. and oral administration. Plasma concentrations of glyphosate and its metabolite aminomethyl phosphonic acid (AMPA) were determined by HPLC method. After i.v. and oral administration, plasma concentration-time curves were best described by a two-compartment open model. For glyphosate, the elimination half-lives (T(1/2beta)) from plasma were 9.99 hr after i.v. and 14.38 hr after oral administration. The total plasma clearance was not influenced by dose concentration or route and reached a value of 0.995 L/hr/kg. After i.v. administration, the apparent volume of distribution in the second compartment (V(2)) and volume of distribution at steady state (V(ss)) were 2.39 and 2.99 L/kg, respectively, suggesting a considerable diffusion of the herbicide into tissues. After oral administration, glyphosate was partially and slowly absorbed with a T(max) of 5.16 hr. The oral bioavailability of glyphosate was found to be 23.21%. Glyphosate was converted to AMPA. The metabolite AMPA represented 6.49% of the parent drug plasma concentrations. The maximum plasma concentrations of glyphosate and AMPA were 4.62 and 0.416 microg/mL, respectively. The maximum plasma concentration of AMPA was achieved at 2.42 hr. For AMPA, the elimination half-life (T(1/2beta)) was 15.08 hr after oral administration of glyphosate parent compound.
PMID:19607892 Anadon A et al; Toxicol Lett 190 (1): 91-5 (2009)
The disposition of glyphosate was studied in rats. Male F344/N rats were gavaged with 5.6 or 56 mg/kg radiolabeled glyphosate. Urine and feces were collected at 24 hour intervals for 72 hr and analyzed for activity. Selected rats were killed 3 to 96 hr post dosing to determine the tissue distribution of radioactivity. Approximately 20 to 30% of either dose was eliminated in the urine and 70 to 80% in the feces over 72 hr. Only about 1% of the dose remained in the tissues, mostly in the liver and small intestine.
DHHS/NTP; Toxicology and Carcinogenesis Studies of Glyphosate p.55 (1992). Technical Rpt Series No.16. NIH Pub #92-3135. Available from, as of November 10, 2014: https://ntp-server.niehs.nih.gov/
... There is rapid elimination, no biotransformation, and minimal tissue retention of glyphosate in various species, including mammals, birds, and fish.
Krieger, R. (ed.). Handbook of Pesticide Toxicology. Volume 2, 2nd ed. 2001. Academic Press, San Diego, California., p. 1668
Greater than 90% of an orally administered dose of glyphosate is rapidly eliminated in 72 hr /by laboratory animals/. ... Typically, approximately 70% of the administered dose is eliminated in the feces, with the remainder eliminated in the urine. In all cases, less than 0.5% of the administered dose is found in the tissue and organs, demonstrating that glyphosate does not bioaccumulate in edible tissues.
Krieger, R. (ed.). Handbook of Pesticide Toxicology. Volume 2, 2nd ed. 2001. Academic Press, San Diego, California., p. 1668
For more Absorption, Distribution and Excretion (Complete) data for GLYPHOSATE (11 total), please visit the HSDB record page.
...In rats, > 97% of the (14)C/glyphosate/ in excreta, after a single oral dose, was shown to be unchanged compound. AMPA was the only metabolite, covering only 0.2-0.3% of the applied (14)C...
WHO/International Programme on Chemical Safety; Environmental Health Criteria 159, Glyphosate, (1994). Available from, as of November 10, 2014: https://www.inchem.org/pages/ehc.html
Following a single oral dose of 14C-glyphosate, amino methyl phosphonic acid (AMPA) was the only metabolite found in urine (0.2-0.3% of the administered dose) and feces (0.2-0.4% of the administered dose) /of male and female Sprague-Dawley rats/.
USEPA; Reregistration Eligibility Decision (RED) Database for Glyphosate (38641-94-0). EPA 738-R-93-014 (September 1993). Available from, as of January 25, 2006: https://www.epa.gov/pesticides/reregistration/status.htm
Studies of the metabolism of glyphosate in experimental animals (rats, rabbits, lactating goats, and chickens) indicate that it is not biotransformed, with essentially all the administered dose excreted as unchanged parent molecule.
Krieger, R. (ed.). Handbook of Pesticide Toxicology. Volume 2, 2nd ed. 2001. Academic Press, San Diego, California., p. 1669
Biotransformation of glyphosate occurs to a very low degree only. In rats it was shown that all of the carbon-14 in urine and feces, after a single oral application of (14)C-glyphosate, was present as unchanged parent compound. Also in rats, > 97% of the carbon-14 in excreta, after a single oral dose, was shown to be unchanged compound. AMPA was the only metabolite, covering only 0.2-0.3% of the applied carbon-14. In laying hens also, AMPA was the only metabolite, accounting for only a minor part of the applied amount.
WHO/International Programme on Chemical Safety; Environmental Health Criteria 159, Glyphosate, (1994). Available from, as of November 10, 2014: https://www.inchem.org/pages/ehc.html
Male and female Sprague-Dawley rats received single intraperitoneal injections of radiolabeled (14)C glyphosate. The dose level of glyphosate used for male and female rats was 1150 mg/kg. Blood samples were collected 0.25, 0.50, 1, 2, 4, 6 and 10 hours after injection. ... Assuming first order kinetics, the decrease in radioactivity in bone marrow occurred with a half-life of 7.6 and 4.2 hours for males and females, respectively. Similarly, the half-lives of the radioactivity in plasma were approximately 1 hour for both sexes.
USEPA; Reregistration Eligibility Decision (RED) Database for Glyphosate (38641-94-0). EPA 738-R-93-014 (September 1993). Available from, as of January 25, 2006: https://www.epa.gov/pesticides/reregistration/status.htm
The kinetics of whole body elimination were estimated using the radioactivity (14)C measured in urine and feces after a single oral dose of (14)C-glyphosate (10 or 1000 mg/kg body weight). Because of the lack of biotransformation of glyphosate it is valid to base kinetics on total radioactivity. The elimination appeared to be biphasic. The half-life of the alpha elimination phase at 10 mg/kg body weight was 5.87 hr (males) or 6.22 hr (females); at 1000 mg/kg body weight this was 5.26 hr (males)or 6.44 hr (females). The half-life of the beta phase at 10 mg/kg body weight was 79 hr (males) or 106 hr (females); at 1000 mg/kg body weight this was 181 hr (males) or 337 hr (females).
WHO/International Programme on Chemical Safety; Environmental Health Criteria 159, Glyphosate, (1994). Available from, as of November 10, 2014: https://www.inchem.org/pages/ehc.html
The toxicokinetics of glyphosate after single 100 mg/kg intravenous (i.v.) and 400 mg/kg oral doses were studied in rats. ... For glyphosate, the elimination half-lives (T(1/2beta)) from plasma were 9.99 hr after i.v. and 14.38 hr after oral administration.
PMID:19607892 Anadon A et al; Toxicol Lett 190 (1): 91-5 (2009)
Previous studies demonstrate that glyphosate exposure is associated with oxidative damage and neurotoxicity. Therefore, the mechanism of glyphosate-induced neurotoxic effects needs to be determined. The aim of this study was to investigate whether Roundup (a glyphosate-based herbicide) leads to neurotoxicity in hippocampus of immature rats following acute (30min) and chronic (pregnancy and lactation) pesticide exposure. Maternal exposure to pesticide was undertaken by treating dams orally with 1% Roundup (0.38% glyphosate) during pregnancy and lactation (till 15-day-old). Hippocampal slices from 15 day old rats were acutely exposed to Roundup (0.00005-0.1%) during 30min and experiments were carried out to determine whether glyphosate affects (45)Ca(2+) influx and cell viability. Moreover, /this study/ investigated the pesticide effects on oxidative stress parameters, (14)C-alpha-methyl-amino-isobutyric acid ((14)C-MeAIB) accumulation, as well as glutamate uptake, release and metabolism. Results showed that acute exposure to Roundup (30min) increases (45)Ca(2+) influx by activating NMDA receptors and voltage-dependent Ca(2+) channels, leading to oxidative stress and neural cell death. The mechanisms underlying Roundup-induced neurotoxicity also involve the activation of CaMKII and ERK. Moreover, acute exposure to Roundup increased (3)H-glutamate released into the synaptic cleft, decreased GSH content and increased the lipoperoxidation, characterizing excitotoxicity and oxidative damage. /This study/ also observed that both acute and chronic exposure to Roundup decreased (3)H-glutamate uptake and metabolism, while induced (45)Ca(2+) uptake and (14)C-MeAIB accumulation in immature rat hippocampus. Taken together, these results demonstrated that Roundup might lead to excessive extracellular glutamate levels and consequently to glutamate excitotoxicity and oxidative stress in rat hippocampus.
PMID:24636977 Cattani D et al; Toxicology 320: 34-45 (2014)
Glyphosate is the primary active constituent of the commercial pesticide Roundup. The present results show that acute Roundup exposure at low doses (36 ppm, 0.036 g/L) for 30 min induces oxidative stress and activates multiple stress-response pathways leading to Sertoli cell death in prepubertal rat testis. The pesticide increased intracellular Ca(2+) concentration by opening L-type voltage-dependent Ca(2+) channels as well as endoplasmic reticulum IP3 and ryanodine receptors, leading to Ca(2+) overload within the cells, which set off oxidative stress and necrotic cell death. Similarly, 30 min incubation of testis with glyphosate alone (36 ppm) also increased (45)Ca(2+) uptake. These events were prevented by the antioxidants Trolox and ascorbic acid. Activated protein kinase C, phosphatidylinositol 3-kinase, and the mitogen-activated protein kinases such as ERK1/2 and p38MAPK play a role in eliciting Ca(2+) influx and cell death. Roundup decreased the levels of reduced glutathione (GSH) and increased the amounts of thiobarbituric acid-reactive species (TBARS) and protein carbonyls. Also, exposure to glyphosate-Roundup stimulated the activity of glutathione peroxidase, glutathione reductase, glutathione S-transferase, gamma-glutamyltransferase, catalase, superoxide dismutase, and glucose-6-phosphate dehydrogenase, supporting downregulated GSH levels. Glyphosate has been described as an endocrine disruptor affecting the male reproductive system; however, the molecular basis of its toxicity remains to be clarified. We propose that Roundup toxicity, implicated in Ca(2+) overload, cell signaling misregulation, stress response of the endoplasmic reticulum, and/or depleted antioxidant defenses, could contribute to Sertoli cell disruption in spermatogenesis that could have an impact on male fertility.
PMID:23820267 de Liz Oliveira Cavalli VL et al; Free Radic Biol Med 65: 335-46 (2013)
A deregulation of programmed cell death mechanisms in human epidermis leads to skin pathologies. We previously showed that glyphosate, an extensively used herbicide, provoked cytotoxic effects on cultured human keratinocytes, affecting their antioxidant capacities and impairing morphological and functional cell characteristics. The aim of the present study, carried out on the human epidermal cell line HaCaT, was to examine the part of apoptosis plays in the cytotoxic effects of glyphosate and the intracellular mechanisms involved in the apoptotic events. /This study/ conducted different incubation periods to reveal the specific events in glyphosate-induced cell death. /It/ observed an increase in the number of early apoptotic cells at a low cytotoxicity level (15%), and then, a decrease, in favor of late apoptotic and necrotic cell rates for more severe cytotoxicity conditions. At the same time, /the study/ showed that the glyphosate-induced mitochondrial membrane potential disruption could be a cause of apoptosis in keratinocyte cultures.
PMID:22522424 Heu C et al; Environ Toxicol Pharmacol 34 (2): 144-53 (2012)
Herbicides have been recognized as the main environmental factor associated with human neurodegenerative disorders such as Parkinson's disease(PD). Previous studies indicated that the exposure to glyphosate, a widely used herbicide, is possibly linked to Parkinsonism, however the underlying mechanism remains unclear. We investigated the neurotoxic effects of glyphosate in differentiated PC12 /rat/ cells and discovered that it inhibited viability of differentiated PC12 cells in dose-and time-dependent manners. Furthermore, the results showed that glyphosate induced cell death via autophagy pathways in addition to activating apoptotic pathways. Interestingly, deactivation of Beclin-1 gene attenuated both apoptosis and autophagy in glyphosate treated differentiated PC12 cells, suggesting that Beclin-1 gene is involved in the crosstalk between the two mechanisms.
PMID:22504123 Gui YX et al; Neurotoxicol Teratol 34 (3): 344-9 (2012)
For more Mechanism of Action (Complete) data for GLYPHOSATE (7 total), please visit the HSDB record page.
Market Place

ABOUT THIS PAGE
13
PharmaCompass offers a list of Glyphosate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Glyphosate manufacturer or Glyphosate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Glyphosate manufacturer or Glyphosate supplier.
A Glyphosate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Glyphosate, including repackagers and relabelers. The FDA regulates Glyphosate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Glyphosate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Glyphosate supplier is an individual or a company that provides Glyphosate active pharmaceutical ingredient (API) or Glyphosate finished formulations upon request. The Glyphosate suppliers may include Glyphosate API manufacturers, exporters, distributors and traders.
Glyphosate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Glyphosate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Glyphosate GMP manufacturer or Glyphosate GMP API supplier for your needs.
A Glyphosate CoA (Certificate of Analysis) is a formal document that attests to Glyphosate's compliance with Glyphosate specifications and serves as a tool for batch-level quality control.
Glyphosate CoA mostly includes findings from lab analyses of a specific batch. For each Glyphosate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Glyphosate may be tested according to a variety of international standards, such as European Pharmacopoeia (Glyphosate EP), Glyphosate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Glyphosate USP).

