
Synopsis
Synopsis
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
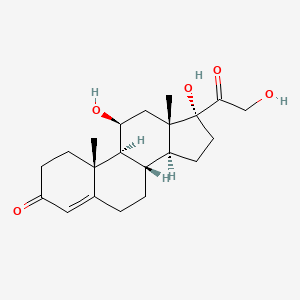

1. 11 Epicortisol
2. 11-epicortisol
3. Cortef
4. Cortifair
5. Cortisol
6. Cortril
7. Epicortisol
8. Hydrocortisone, (11 Alpha)-isomer
9. Hydrocortisone, (9 Beta,10 Alpha,11 Alpha)-isomer
10. Pregn-4-ene-3,20-dione, 11,17,21-trihydroxy-, (11beta)-
1. Cortisol
2. 50-23-7
3. Acticort
4. Cetacort
5. Cortef
6. Hydrasson
7. Hydrocortisyl
8. Hydrocortone
9. Cobadex
10. Hytone
11. Cortenema
12. Cortril
13. Dermacort
14. Proctocort
15. Hycort
16. Signef
17. 17-hydroxycorticosterone
18. Optef
19. Kendall's Compound F
20. Cort-dome
21. Cortanal
22. Corticreme
23. Cortifan
24. Cortiment
25. Cortispray
26. Cortonema
27. Dermolate
28. Efcorbin
29. Efcortelan
30. Eldecort
31. Ficortril
32. Genacort
33. Hycortol
34. Hycortole
35. Penecort
36. Permicort
37. Tarcortin
38. Traumaide
39. Alacort
40. Cleiton
41. Epicort
42. Dihydrocostisone
43. Hytone Lotion
44. Hidro-colisona
45. Hydro-adreson
46. Scheroson F
47. Incortin-h
48. Reichstein's Substance M
49. Ala-scalp
50. Domolene-hc
51. Epiderm H
52. Esiderm H
53. Otosone-f
54. Polcort H
55. Aeroseb-hc
56. Cortolotion
57. Cortoxide
58. Cremesone
59. Eldercort
60. Heb-cort
61. Maintasone
62. Nutracort
63. Delacort
64. Dioderm
65. Mildison
66. Rectoid
67. Synacort
68. Anflam
69. Hydrocorticosterone
70. Hydroxycortisone
71. H-cort
72. Hydro-colisona
73. Cortisol Alcohol
74. Incortin-hydrogen
75. Ala-cort
76. Barseb Hc
77. Dermocortal
78. Flexicort
79. Texacort
80. Timocort
81. Evacort
82. Komed Hc
83. Hydrocortisone Base
84. Lacticare-hc
85. Texacort Lotion 25
86. Anti-inflammatory Hormone
87. Hydrocortisone Alcohol
88. Algicirtis
89. Aquacort
90. Colocort
91. Cortesal
92. Cortisolonum
93. Hidalone
94. Hytisone
95. Kyypakkaus
96. Lactisona
97. Lubricort
98. Meusicort
99. Milliderm
100. Sanatison
101. Schericur
102. Sigmacort
103. Stiefcorcil
104. Amberin
105. Cutisol
106. Dermil
107. Glycort
108. Uniderm
109. Foille Insetti
110. Gyno-cortisone
111. Balneol-hc
112. Transderma H
113. Basan-corti
114. Clear Aid
115. Cremicort-h
116. Dome-cort
117. Stie-cort
118. Beta-hc
119. Neosporin-h Ear
120. Remederm Hc
121. Aquanil Hc
122. Cortisporin Otico
123. Derm-aid
124. Heb Cort
125. Nogenic Hc
126. Scalpicin Capilar
127. Systral Hydrocort
128. Prevex Hc
129. Cortisporin
130. Efcortelin
131. Fiocortril
132. Hidrocortisona
133. Hydrocortisone Free Alcohol
134. Hydrocortisonum
135. Proctofoam
136. Alphaderm
137. Hydracort
138. Medicort
139. Otocort
140. Zenoxone
141. Drotic
142. Vytone
143. 11beta-hydroxycortisone
144. Nystaform-hc
145. Aeroseb Hc
146. Caldecort Spray
147. Anusol Hc
148. Pediotic Suspension
149. Vosol Hc
150. Idrocortisone
151. 17alpha-hydroxycorticosterone
152. Hydrocortal
153. Hydroskin
154. Otalgine
155. Otobiotic
156. Plenadren
157. Protocort
158. Hysone
159. Racet
160. Ef Corlin
161. 11beta-hydrocortisone
162. Compound F
163. Lacticare Hc
164. Compound F (kendall)
165. 11-beta-hydrocortisone
166. 11-beta-hydroxycortisone
167. Hydrocortisone (cortisol)
168. Chronocort
169. Hydrocort
170. Preparation H Hydrocortisone Cream
171. Neo-cort-dome
172. 11beta,17alpha,21-trihydroxy-4-pregnene-3,20-dione
173. Otic-neo-cort-dome
174. 11beta,17,21-trihydroxypregn-4-ene-3,20-dione
175. Nsc 10483
176. Hc
177. [3h]cortisol
178. Nsc-10483
179. (11beta)-11,17,21-trihydroxypregn-4-ene-3,20-dione
180. Prestwick_265
181. (8s,9s,10r,11s,13s,14s,17r)-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-2,6,7,8,9,11,12,14,15,16-decahydro-1h-cyclopenta[a]phenanthren-3-one
182. 4-pregnene-11beta,17alpha,21-triol-3,20-dione
183. Chebi:17650
184. 11beta,17alpha,21-trihydroxypregn-4-ene-3,20-dione
185. Nsc10483
186. 11.beta.-hydrocortisone
187. Dermaspray
188. Wi4x0x7bpj
189. 11beta,17,21-trihydroxyprogesterone
190. 11.beta.-hydroxycortisone
191. Ophthocort
192. Terra-cortril
193. Mls000069609
194. 17.alpha.-hydroxycorticosterone
195. Pregn-4-ene-3,20-dione, 11,17,21-trihydroxy-, (11.beta.)-
196. Pregn-4-ene-3,20-dione, 11,17,21-trihydroxy-, (11beta)-
197. 4-pregnen-11beta,17alpha,21-triol-3,20-dione
198. Idrocortisone [dcit]
199. Genacort (lotion)
200. Anucort
201. Pregn-4-ene-3,20-dione, 11,17,21-trihydroxy-, (11b)-
202. Prepcort
203. Smr000059022
204. Dsstox_cid_714
205. Hydrocortisonum [inn-latin]
206. Proctozone Hc
207. Scalp-cort
208. Hidrocortisona [inn-spanish]
209. Rectasol-hc
210. Anucort-hc
211. Hydro-rx
212. Dsstox_rid_75753
213. Dsstox_gsid_20714
214. (8s,9s,10r,11s,13s,14s,17r)-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3(2h)-one
215. Hc (hydrocortisone)
216. 11.beta.,17,21-trihydroxypregn-4-ene-3,20-dione
217. Corhydron
218. (11alpha,14beta)-11,17,21-trihydroxypregn-4-ene-3,20-dione
219. Duocort
220. Hydrocortisone In Absorbase
221. Proctosol-hc
222. Hc #1
223. Hc #4
224. Acticort (tn)
225. Colocort (tn)
226. (1s,2r,10s,11s,14r,15s,17s)-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-6-en-5-one
227. Smr000653523
228. Cortef (tn)
229. Hytone (tn)
230. Ccris 5854
231. Component Of Otalgine
232. Anusol Hc (tn)
233. Component Of Lubricort
234. Cor-oticin
235. Hsdb 3339
236. Einecs 200-020-1
237. Unii-wi4x0x7bpj
238. Mfcd00011654
239. Component Of Neo-cort-dome
240. Cortizol
241. Efmody
242. Ai3-25006
243. 3h-cortisol
244. 11beta-cortisol
245. Cas-50-23-7
246. 11-hydrocortisone
247. Plenadren (tn)
248. Ncgc00022848-06
249. 11b-hydrocortisone
250. Hydrocortisone [usp:inn:ban:jan]
251. Drotic (salt/mix)
252. 11b-hydroxycortisone
253. Otocort (salt/mix)
254. Pregn-4-ene-3,20-dione, 11beta,17,21-trihydroxy-
255. Otalgine (salt/mix)
256. Hydrocortisone, 98%
257. 11,17,21-trihydroxypregn-4-ene-3,20-dione
258. Alkindi Sprinkle
259. Alphaderm (salt/mix)
260. Hydrocortisone, Topical
261. Otobiotic (salt/mix)
262. 4p6x
263. Cort-quin (salt/mix)
264. Cortisporin (salt/mix)
265. Vosol Hc (salt/mix)
266. 11a-hydroxycorticosterone
267. 17a-hydroxycorticosterone
268. Opera_id_1292
269. Pregn-4-ene-3,20-dione, 11,17,21-trihydroxy-, (11-beta)-
270. Prestwick0_000447
271. Prestwick1_000447
272. Prestwick2_000447
273. Prestwick3_000447
274. Epitope Id:174851
275. Upcmld-dp133
276. Ec 200-020-1
277. H 4001
278. Hydrocortisone [ii]
279. Hydrocortisone [mi]
280. Schembl4148
281. Neo-cort-dome (salt/mix)
282. Hydrocortisone [inn]
283. Hydrocortisone [jan]
284. Lopac0_000594
285. 11alpha-hydroxycorticosterone
286. Bspbio_000494
287. Hydrocortisone [hsdb]
288. Mls001148103
289. Mls002207135
290. Mls002222189
291. Mls002548868
292. Hydrocortisone [vandf]
293. Spbio_002433
294. Hydrocortisone [mart.]
295. Bpbio1_000544
296. Chembl389621
297. Gtpl2868
298. Hydrocortisone [usp-rs]
299. Hydrocortisone [who-dd]
300. Hydrocortisone [who-ip]
301. Pediotic Suspension (salt/mix)
302. Dtxsid7020714
303. Upcmld-dp133:001
304. Bdbm13775
305. Otic-neo-cort-dome (salt/mix)
306. 2v95
307. Hydrocortisone (jp17/usp/inn)
308. Hms1569i16
309. Hms2090m04
310. Hms2096i16
311. Hms2230b18
312. Hms2235f17
313. Hms3259c05
314. Hms3261h10
315. Hms3713i16
316. Hydrocortisone, >=98% (hplc)
317. Vioform-hydrocortisone (salt/mix)
318. 11b,17,21-trihydroxyprogesterone
319. Hydrocortisone [green Book]
320. Bcp09054
321. Hy-n0583
322. Hydrocortisone [orange Book]
323. Tox21_110883
324. Tox21_200815
325. Tox21_500594
326. Hydrocortisone [ep Monograph]
327. Lmst02030001
328. S1696
329. Zinc13540519
330. Hydrocortisone [usp Monograph]
331. Akos001582651
332. Hydrocortisonum [who-ip Latin]
333. Oticair Component Hydrocortisone
334. Otocort Component Hydrocortisone
335. Tox21_110883_1
336. Ccg-204683
337. Db00741
338. Lp00594
339. Nc00456
340. Sdccgsbi-0050576.p003
341. 11.beta.,17,21-trihydroxyprogesterone
342. Cipro Hc Component Hydrocortisone
343. Orlex Hc Component Hydrocortisone
344. Otobione Component Hydrocortisone
345. Pediotic Component Hydrocortisone
346. Pyocidin Component Hydrocortisone
347. Smp1_000156
348. Vosol Hc Component Hydrocortisone
349. Alphaderm Component Hydrocortisone
350. Ncgc00022848-07
351. Ncgc00022848-09
352. Ncgc00022848-10
353. Ncgc00022848-11
354. Ncgc00022848-12
355. Ncgc00022848-13
356. Ncgc00022848-14
357. Ncgc00022848-15
358. Ncgc00022848-17
359. Ncgc00022848-26
360. Ncgc00258369-01
361. Ncgc00261279-01
362. Otobiotic Component Hydrocortisone
363. (1s,10s,11s,15s,17s,2r,14r)-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,15-dimethyl Tetracyclo[8.7.0.0<2,7>.0<11,15>]heptadec-6-en-5-one
364. (8s,9s,10r,11s,13s,14s,17r)-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-1,2,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-3h-cyclopenta[a]phenanthren-3-one
365. Ac-12902
366. As-11651
367. Bp-20390
368. Hydrocortisone 100 Microg/ml In Methanol
369. Hydrocortisone Component Of Oticair
370. Hydrocortisone Component Of Otocort
371. Nci60_000118
372. Acetasol Hc Component Hydrocortisone
373. Calmurid Hc Component Hydrocortisone
374. Hydrocortisone Component Of Otobione
375. Hydrocortisone Component Of Pediotic
376. Hydrocortisone Component Of Pyocidin
377. 4-pregnene-11alpha,21-triol 3,20-dione
378. Hydrocortisone Component Of Alphaderm
379. Hydrocortisone Component Of Cipro Hc
380. Hydrocortisone Component Of Orlex Hc
381. Hydrocortisone Component Of Otobiotic
382. Hydrocortisone Component Of Vosol Hc
383. B1951
384. Eu-0100594
385. Prednisolone Impurity A [ep Impurity]
386. 4-pregnene-11b,17a,21-triol-3,20-dione
387. Hydrocortisone 100 Microg/ml In Acetonitrile
388. Hydrocortisone Component Of Acetasol Hc
389. Hydrocortisone Component Of Calmurid Hc
390. 50h237
391. C00735
392. D00088
393. U 1851
394. Hydrocortisone, Meets Usp Testing Specifications
395. Pregn-4-ene-3, 11.beta.,17,21-trihydroxy-
396. 11?,17?,21-trihydroxypregn-4-ene-3,20-dione
397. A929789
398. Hydrocortisone, Vetranal(tm), Analytical Standard
399. Q190875
400. Sr-01000000139
401. Q-201211
402. Sr-01000000139-3
403. Brd-k93568044-001-03-1
404. Brd-k93568044-001-11-4
405. Brd-k93568044-001-32-0
406. Hydrocortisone Acetate Impurity A [ep Impurity]
407. Hydrocortisone, Bioreagent, Suitable For Cell Culture
408. 4-pregnen-11.beta.,17.alpha.,21-triol-3,20-dione
409. 4-pregnene-11.beta.,17.alpha.,21-triol-3,20-dione
410. Pregn-4-ene-3,20-dione, 11.beta.,17,21-trihydroxy-
411. Z2786051549
412. (11beta)-11,17,21-trihydroxy-pregn-4-ene-3,20-dione
413. 11.beta.,17.alpha.,21-trihydroxy-4-pregnene-3,20-dione
414. 11.beta.,17.alpha.,21-trihydroxypregn-4-ene-3,20-dione
415. B48448a1-24ba-47ca-8d9e-43e5bc949386
416. Hydrocortisone, British Pharmacopoeia (bp) Assay Standard
417. Pregn-4-ene-3, 11,17,21-trihydroxy-, (11.beta.)-
418. 11,17,21-trihydroxypregn-4-ene-3,20-dione, (11.beta.)-
419. Hydrocortisone Sodium Succinate Impurity A [ep Impurity]
420. Hydrocortisone, European Pharmacopoeia (ep) Reference Standard
421. Hydrocortisone Hydrogen Succinate Impurity A [ep Impurity]
422. Hydrocortisone, United States Pharmacopeia (usp) Reference Standard
423. Hydrocortisone-water Soluble, Bioreagent, Suitable For Cell Culture
424. Hydrocortisone, Gamma-irradiated, Powder, Bioxtra, Suitable For Cell Culture
425. 4-(6-chloro-4-oxoquinazolin-3(4h)-yl)-n-(3-methoxyphenyl)piperidine-1-carboxamide
426. Cortisol Solution, 1.0 Mg/ml In Methanol, Ampule Of 1 Ml, Certified Reference Material
427. Hydrocortisone For Peak Identification, European Pharmacopoeia (ep) Reference Standard
428. Hydrocortisone Solution, 50 Mum, Sterile-filtered, Bioxtra, Suitable For Cell Culture
429. Hydrocortisone, Pharmaceutical Secondary Standard; Certified Reference Material
430. (10r,13s,17r)-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-2,6,7,8,9,11,12,14,15,16-decahydro-1h-cyclopenta[a]phenanthren-3-one
1. Efficort
2. Retef
3. Hydrocortisone Aceponate
4. Cortef Fluid
5. Hydrocortisone Cypionate
6. Hidroaltesona
7. Hydrocortone Phosphate
8. Hydrocortisone Phosphate
9. Hydrocortisone Sodium Phosphate
10. Aacicortisol
11. Hydrocortisone Hydrogen Succinate
12. Hydrocortisone Hemisuccinate Hydrate
13. Hydrocortisone Hemisuccinate
14. Hydrocortisone 21-hemisuccinate
| Molecular Weight | 362.5 g/mol |
|---|---|
| Molecular Formula | C21H30O5 |
| XLogP3 | 1.6 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 362.20932405 g/mol |
| Monoisotopic Mass | 362.20932405 g/mol |
| Topological Polar Surface Area | 94.8 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 684 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 7 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 36 | |
|---|---|
| Drug Name | Acetasol hc |
| PubMed Health | Acetic Acid/Hydrocortisone (Into the ear) |
| Drug Classes | Anti-Infective/Anti-Inflammatory Combination |
| Active Ingredient | Acetic acid, glacial; hydrocortisone |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 1%; 2% |
| Market Status | Prescription |
| Company | Actavis Mid Atlantic |
| 2 of 36 | |
|---|---|
| Drug Name | Ala-cort |
| PubMed Health | Hydrocortisone |
| Drug Classes | Anti-Inflammatory, Corticosteroid, Intermediate, Corticosteroid, Weak, Endocrine-Metabolic Agent, Gastrointestinal Agent, Hemorrhoidal Anti-Inflammatory, Hydrocortisone, Immune Suppressant |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Cream |
| Route | Topical |
| Strength | 1% |
| Market Status | Prescription |
| Company | Crown Labs |
| 3 of 36 | |
|---|---|
| Drug Name | Ala-scalp |
| PubMed Health | Hydrocortisone (On the skin) |
| Drug Classes | Corticosteroid, Intermediate, Corticosteroid, Weak |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Lotion |
| Route | Topical |
| Strength | 2% |
| Market Status | Prescription |
| Company | Crown Labs |
| 4 of 36 | |
|---|---|
| Drug Name | Anusol hc |
| PubMed Health | Hydrocortisone (By mouth) |
| Drug Classes | Anti-Inflammatory, Endocrine-Metabolic Agent, Immune Suppressant |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Cream |
| Route | Topical |
| Strength | 2.5% |
| Market Status | Prescription |
| Company | Salix Pharms |
| 5 of 36 | |
|---|---|
| Drug Name | Colocort |
| PubMed Health | Hydrocortisone Enema (Rectal) |
| Drug Classes | Anti-Inflammatory, Gastrointestinal Agent |
| Drug Label | Hydrocortisone is a white to practically white, odorless, crystalline powder, very slightly soluble in water. It has the chemical name Pregn-4-ene-3,20-dione, 11,17,21-trihydroxy-,(11)- and the following structural formula:Colocort is a convenien... |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Enema |
| Route | Rectal |
| Strength | 100mg/60ml |
| Market Status | Prescription |
| Company | Paddock |
| 6 of 36 | |
|---|---|
| Drug Name | Cortef |
| PubMed Health | Hydrocortisone (Rectal) |
| Drug Classes | Anti-Inflammatory, Hemorrhoidal Anti-Inflammatory |
| Drug Label | CORTEF Tablets contain hydrocortisone which is a glucocorticoid. Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic, which are readily absorbed from the gastrointestinal tract. Hydrocortisone USP is white to practical... |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 5mg; 10mg; 20mg |
| Market Status | Prescription |
| Company | Pharmacia And Upjohn |
| 7 of 36 | |
|---|---|
| Drug Name | Cortenema |
| PubMed Health | Neomycin, Polymyxin B, and Hydrocortisone |
| Drug Classes | Adrenal Glucocorticoid, Aminoglycoside/Corticosteroid Combination, Anti-Infective/Anti-Inflammatory Combination, Neomycin |
| Drug Label | Hydrocortisone is a white to practically white, odorless, crystalline powder, very slightly soluble in water. It has the chemical name Pregn-4-ene-3,20-dione,11,17,21-trihydroxy,(11)-and the following structural formula: CORTENEMA is a convenient... |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Enema |
| Route | Rectal |
| Strength | 100mg/60ml |
| Market Status | Prescription |
| Company | Ani Pharms |
| 8 of 36 | |
|---|---|
| Drug Name | Cortifoam |
| PubMed Health | Hydrocortisone |
| Drug Classes | Anti-Inflammatory, Corticosteroid, Intermediate, Corticosteroid, Weak, Endocrine-Metabolic Agent, Gastrointestinal Agent, Hemorrhoidal Anti-Inflammatory, Hydrocortisone, Immune Suppressant |
| Active Ingredient | Hydrocortisone acetate |
| Dosage Form | Aerosol, metered |
| Route | Rectal |
| Strength | 10% |
| Market Status | Prescription |
| Company | Meda Pharms |
| 9 of 36 | |
|---|---|
| Drug Name | Cortisporin |
| PubMed Health | Hydrocortisone |
| Drug Classes | Anti-Inflammatory, Corticosteroid, Intermediate, Corticosteroid, Weak, Endocrine-Metabolic Agent, Gastrointestinal Agent, Hemorrhoidal Anti-Inflammatory, Hydrocortisone, Immune Suppressant |
| Active Ingredient | Hydrocortisone acetate; polymyxin b sulfate; Hydrocortisone; Bacitracin zinc; hydrocortisone; neomycin sulfate |
| Dosage Form | Solution/drops; Suspension/drops; Ointment; Cream |
| Route | Topical; Otic |
| Strength | 400 units/gm; 10,000 units/gm; 0.5%; 10,000 units/ml; 1%; eq 3.5mg base/ml; eq 3.5mg base/gm; 5,000 units/gm |
| Market Status | Prescription |
| Company | Monarch Pharms; Citron Pharma |
| 10 of 36 | |
|---|---|
| Drug Name | Hydrocortisone |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Ointment; Enema; Tablet; Cream; Lotion |
| Route | Rectal; Oral; Topical |
| Strength | 1%; 5mg; 10mg; 2.5%; 100mg/60ml; 20mg |
| Market Status | Prescription |
| Company | Vintage Pharms; Amedra Pharms; Taro; Fougera; Lyne; Fougera Pharms; Teva Pharms; Actavis Mid Atlantic; Vintage; Perrigo New York |
| 11 of 36 | |
|---|---|
| Drug Name | Hydrocortisone and acetic acid |
| Drug Label | Topical corticosteroids constitute a class of primarily synthetic steroids used as anti-inflammatory and antipruritic agents. Texacort Topical Solution contains hydrocortisone as the active corticosteroid, having the chemical formula of Pregn-4-ene-3... |
| Active Ingredient | Acetic acid, glacial; hydrocortisone |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 1%; 2% |
| Market Status | Prescription |
| Company | Taro; Vintage |
| 12 of 36 | |
|---|---|
| Drug Name | Hydrocortisone in absorbase |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Ointment |
| Route | Topical |
| Strength | 1% |
| Market Status | Prescription |
| Company | Carolina Medcl |
| 13 of 36 | |
|---|---|
| Drug Name | Hydro-rx |
| PubMed Health | Acetic Acid/Hydrocortisone (Into the ear) |
| Drug Classes | Anti-Infective/Anti-Inflammatory Combination |
| Drug Label | Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic, which are readily absorbed from the gastrointestinal tract.Hydrocortisone is a white to practically white, odorless, crystalline powder, very slightly soluble in wat... |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Powder |
| Route | For rx compounding |
| Strength | 100% |
| Market Status | Prescription |
| Company | X Gen Pharms |
| 14 of 36 | |
|---|---|
| Drug Name | Nutracort |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Lotion |
| Route | Topical |
| Strength | 2.5% |
| Market Status | Prescription |
| Company | Dow Pharm |
| 15 of 36 | |
|---|---|
| Drug Name | Stie-cort |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Lotion |
| Route | Topical |
| Strength | 2.5% |
| Market Status | Prescription |
| Company | Perrigo |
| 16 of 36 | |
|---|---|
| Drug Name | Synacort |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Cream |
| Route | Topical |
| Strength | 1%; 2.5% |
| Market Status | Prescription |
| Company | Medicis |
| 17 of 36 | |
|---|---|
| Drug Name | Texacort |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Solution |
| Route | Topical |
| Strength | 2.5% |
| Market Status | Prescription |
| Company | Mission Pharma |
| 18 of 36 | |
|---|---|
| Drug Name | Vosol hc |
| Active Ingredient | Acetic acid, glacial; hydrocortisone |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 1%; 2% |
| Market Status | Prescription |
| Company | Hi Tech Pharma |
| 19 of 36 | |
|---|---|
| Drug Name | Acetasol hc |
| PubMed Health | Acetic Acid/Hydrocortisone (Into the ear) |
| Drug Classes | Anti-Infective/Anti-Inflammatory Combination |
| Active Ingredient | Acetic acid, glacial; hydrocortisone |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 1%; 2% |
| Market Status | Prescription |
| Company | Actavis Mid Atlantic |
| 20 of 36 | |
|---|---|
| Drug Name | Ala-cort |
| PubMed Health | Hydrocortisone |
| Drug Classes | Anti-Inflammatory, Corticosteroid, Intermediate, Corticosteroid, Weak, Endocrine-Metabolic Agent, Gastrointestinal Agent, Hemorrhoidal Anti-Inflammatory, Hydrocortisone, Immune Suppressant |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Cream |
| Route | Topical |
| Strength | 1% |
| Market Status | Prescription |
| Company | Crown Labs |
| 21 of 36 | |
|---|---|
| Drug Name | Ala-scalp |
| PubMed Health | Hydrocortisone (On the skin) |
| Drug Classes | Corticosteroid, Intermediate, Corticosteroid, Weak |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Lotion |
| Route | Topical |
| Strength | 2% |
| Market Status | Prescription |
| Company | Crown Labs |
| 22 of 36 | |
|---|---|
| Drug Name | Anusol hc |
| PubMed Health | Hydrocortisone (By mouth) |
| Drug Classes | Anti-Inflammatory, Endocrine-Metabolic Agent, Immune Suppressant |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Cream |
| Route | Topical |
| Strength | 2.5% |
| Market Status | Prescription |
| Company | Salix Pharms |
| 23 of 36 | |
|---|---|
| Drug Name | Colocort |
| PubMed Health | Hydrocortisone Enema (Rectal) |
| Drug Classes | Anti-Inflammatory, Gastrointestinal Agent |
| Drug Label | Hydrocortisone is a white to practically white, odorless, crystalline powder, very slightly soluble in water. It has the chemical name Pregn-4-ene-3,20-dione, 11,17,21-trihydroxy-,(11)- and the following structural formula:Colocort is a convenien... |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Enema |
| Route | Rectal |
| Strength | 100mg/60ml |
| Market Status | Prescription |
| Company | Paddock |
| 24 of 36 | |
|---|---|
| Drug Name | Cortef |
| PubMed Health | Hydrocortisone (Rectal) |
| Drug Classes | Anti-Inflammatory, Hemorrhoidal Anti-Inflammatory |
| Drug Label | CORTEF Tablets contain hydrocortisone which is a glucocorticoid. Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic, which are readily absorbed from the gastrointestinal tract. Hydrocortisone USP is white to practical... |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 5mg; 10mg; 20mg |
| Market Status | Prescription |
| Company | Pharmacia And Upjohn |
| 25 of 36 | |
|---|---|
| Drug Name | Cortenema |
| PubMed Health | Neomycin, Polymyxin B, and Hydrocortisone |
| Drug Classes | Adrenal Glucocorticoid, Aminoglycoside/Corticosteroid Combination, Anti-Infective/Anti-Inflammatory Combination, Neomycin |
| Drug Label | Hydrocortisone is a white to practically white, odorless, crystalline powder, very slightly soluble in water. It has the chemical name Pregn-4-ene-3,20-dione,11,17,21-trihydroxy,(11)-and the following structural formula: CORTENEMA is a convenient... |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Enema |
| Route | Rectal |
| Strength | 100mg/60ml |
| Market Status | Prescription |
| Company | Ani Pharms |
| 26 of 36 | |
|---|---|
| Drug Name | Cortifoam |
| PubMed Health | Hydrocortisone |
| Drug Classes | Anti-Inflammatory, Corticosteroid, Intermediate, Corticosteroid, Weak, Endocrine-Metabolic Agent, Gastrointestinal Agent, Hemorrhoidal Anti-Inflammatory, Hydrocortisone, Immune Suppressant |
| Active Ingredient | Hydrocortisone acetate |
| Dosage Form | Aerosol, metered |
| Route | Rectal |
| Strength | 10% |
| Market Status | Prescription |
| Company | Meda Pharms |
| 27 of 36 | |
|---|---|
| Drug Name | Cortisporin |
| PubMed Health | Hydrocortisone |
| Drug Classes | Anti-Inflammatory, Corticosteroid, Intermediate, Corticosteroid, Weak, Endocrine-Metabolic Agent, Gastrointestinal Agent, Hemorrhoidal Anti-Inflammatory, Hydrocortisone, Immune Suppressant |
| Active Ingredient | Hydrocortisone acetate; polymyxin b sulfate; Hydrocortisone; Bacitracin zinc; hydrocortisone; neomycin sulfate |
| Dosage Form | Solution/drops; Suspension/drops; Ointment; Cream |
| Route | Topical; Otic |
| Strength | 400 units/gm; 10,000 units/gm; 0.5%; 10,000 units/ml; 1%; eq 3.5mg base/ml; eq 3.5mg base/gm; 5,000 units/gm |
| Market Status | Prescription |
| Company | Monarch Pharms; Citron Pharma |
| 28 of 36 | |
|---|---|
| Drug Name | Hydrocortisone |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Ointment; Enema; Tablet; Cream; Lotion |
| Route | Rectal; Oral; Topical |
| Strength | 1%; 5mg; 10mg; 2.5%; 100mg/60ml; 20mg |
| Market Status | Prescription |
| Company | Vintage Pharms; Amedra Pharms; Taro; Fougera; Lyne; Fougera Pharms; Teva Pharms; Actavis Mid Atlantic; Vintage; Perrigo New York |
| 29 of 36 | |
|---|---|
| Drug Name | Hydrocortisone and acetic acid |
| Drug Label | Topical corticosteroids constitute a class of primarily synthetic steroids used as anti-inflammatory and antipruritic agents. Texacort Topical Solution contains hydrocortisone as the active corticosteroid, having the chemical formula of Pregn-4-ene-3... |
| Active Ingredient | Acetic acid, glacial; hydrocortisone |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 1%; 2% |
| Market Status | Prescription |
| Company | Taro; Vintage |
| 30 of 36 | |
|---|---|
| Drug Name | Hydrocortisone in absorbase |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Ointment |
| Route | Topical |
| Strength | 1% |
| Market Status | Prescription |
| Company | Carolina Medcl |
| 31 of 36 | |
|---|---|
| Drug Name | Hydro-rx |
| PubMed Health | Acetic Acid/Hydrocortisone (Into the ear) |
| Drug Classes | Anti-Infective/Anti-Inflammatory Combination |
| Drug Label | Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic, which are readily absorbed from the gastrointestinal tract.Hydrocortisone is a white to practically white, odorless, crystalline powder, very slightly soluble in wat... |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Powder |
| Route | For rx compounding |
| Strength | 100% |
| Market Status | Prescription |
| Company | X Gen Pharms |
| 32 of 36 | |
|---|---|
| Drug Name | Nutracort |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Lotion |
| Route | Topical |
| Strength | 2.5% |
| Market Status | Prescription |
| Company | Dow Pharm |
| 33 of 36 | |
|---|---|
| Drug Name | Stie-cort |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Lotion |
| Route | Topical |
| Strength | 2.5% |
| Market Status | Prescription |
| Company | Perrigo |
| 34 of 36 | |
|---|---|
| Drug Name | Synacort |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Cream |
| Route | Topical |
| Strength | 1%; 2.5% |
| Market Status | Prescription |
| Company | Medicis |
| 35 of 36 | |
|---|---|
| Drug Name | Texacort |
| Active Ingredient | Hydrocortisone |
| Dosage Form | Solution |
| Route | Topical |
| Strength | 2.5% |
| Market Status | Prescription |
| Company | Mission Pharma |
| 36 of 36 | |
|---|---|
| Drug Name | Vosol hc |
| Active Ingredient | Acetic acid, glacial; hydrocortisone |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 1%; 2% |
| Market Status | Prescription |
| Company | Hi Tech Pharma |
Anti-Inflammatory Agents, Steroidal
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
MEDICATION (VET): Acute urticaria /can be treated by/ rapid-acting adrenocorticosteroids, eg, hydrocortisone ... .
Kahn, C.M. (Ed.); The Merck Veterinary Manual 9th ed. Merck & Co. Whitehouse Station, NJ. 2005, p. 690
MEDICATION (VET): /USED/ IV, IN PREVENTING OR TREATING ADRENAL FAILURE & SHOCK-LIKE CONDITIONS IN SURGICAL CASES WHICH HAVE BEEN ON CORTICOSTEROIDS, IN ACUTE ALLERGIC REACTIONS...IN POOR SURGICAL RISKS, & IN CASES WHICH HAVE HAD OVERWHELMING SYSTEMIC INFECTIONS...IN DOGS OR CATTLE...
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 266
MEDICATION (VET) /EXPL:/: 5 Standardbreds and 4 Dutch Warmblood horses /were/ used to examine sensitivity of peripheral tissues to exogenous insulin 24 hours after administration of a single dose of hydrocortisone (0.06 mg/kg), eGH (20 ug/kg), or saline (0.9% NaCl) solution and after long-term administration (11 to 15 days) of eGH to horses. The amounts of metabolized glucose (M) and plasma insulin concentration (I) were determined. Values for M and the M-to-I ratio were significantly higher 24 hours after administration of a single dose of hydrocortisone than after single-dose administration of eGH or saline solution. After long-term administration of eGH, basal I concentration was increased and the mean M-to-I ratio was 22% lower, compared with values for horses treated with saline solution. Increases in M and the M-to-I ratio after a single dose of hydrocortisone imply that short-term hydrocortisone treatment increases glucose use by, and insulin sensitivity of, peripheral tissues. Assuming a single dose of hydrocortisone improves sensitivity of peripheral tissues to insulin, it may be an interesting candidate for use in reducing insulin resistance in peripheral tissues of horses with several disease states.
PMID:6334949 de Graaf-Roelfsema E et al; Am J Vet Res 66 (11): 1907-13 (2005)
For more Therapeutic Uses (Complete) data for HYDROCORTISONE (23 total), please visit the HSDB record page.
It is not known whether rectal corticosteroids are distributed into breast milk. Systemic corticosteroids are distributed into breast milk and may cause unwanted effects, such as growth suppression, in the infant. Rectal corticosteroids are not recommended for use by breast-feeding mothers. /Corticosteroids, rectal/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 913
The results of a prospective randomized controlled trial, which looked at the incidence of postoperative diabetes insipidus following the use of three different hydrocortisone protocols, and the results of a study, on the incidence of diabetes insipidus and cortisol response in patients not given hydrocortisone /was reported/. In study 1, 114 patients with pituitary macroadenoma were randomized into three groups: conventional dose (injected hydrocortisone 100 mg IV 6-hourly for 3 days); intermediate dose (injected hydrocortisone 100 mg IV 6-hourly on day 1, 100 mg IV 8-hourly on day 2, and 100 mg IV 12-hourly on day 3); low dose protocol (injected hydrocortisone 25 mg IV 6-hourly on day 1, 25 mg IV 8-hourly on day 2 and 25 mg IV 12-hourly on day 3). Radical excision was achieved in 92 patients. The incidence of diabetes insipidus with the conventional dose was 52%, intermediate dose, 36% and low dose, 24% (p = 0.025). Study 2 included 16 consecutive patients with Hardy's grade A & B pituitary adenoma. These patients were randomized to receive (Group I) or not receive (Group II) hydrocortisone. Patients in Group II demonstrated normal cortisol response intraoperatively and no patient developed features of hypocortisolism; the incidence of diabetes insipidus in this group was 14%. The low dose hydrocortisone protocol reduced the incidence of diabetes insipidus by 46% when compared with the conventional dose hydrocortisone protocol. In patients with grade A and B tumor with normal preoperative cortisol levels, the use of perioperative hydrocortisone can be avoided.
PMID:14635749 Rajaratnam S et al; Br J Neurosurg 17 (5): 437-42 (2003)
ACUTE ADRENAL INSUFFICIENCY RESULTS FROM TOO RAPID WITHDRAWAL OF CORTICOSTEROID THERAPY. /CORTICOSTEROIDS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 1496
POTENTIAL ADVERSE EFFECTS ON FETUS: Cleft palate, spontaneous abortions, and intrauterine growth retardation in animals. Potential for cleft palate formation and adrenal suppression in humans, although teratogenic effects have not been confirmed. POTENTIAL SIDE EFFECTS ON BREAST-FED INFANT: Passes into breast milk in small amounts. Administration of physiologic doses unlikely to adversely affect infant. FDA Category: C (C = Studies in laboratory animals have revealed adverse effects on the fetus (teratogenic, embryocidal, etc.) but there are no controlled studies in pregnant women. The benefits from use of the drug in pregnant women may be acceptable despite its potential risks, or there are no laboratory animal studies or adequate studies in pregnant women.) /Adrenocorticosteroids/ /from table II/
PMID:2195076 Stockton DL, Palle AS; J Am Acad Dermatol 23 (1): 87-103 (1990)
For more Drug Warnings (Complete) data for HYDROCORTISONE (31 total), please visit the HSDB record page.
Otic solutions are indicated for infections of the external auditory canal caused by susceptible organisms and with inflammation. Hydrocortisone tablets are indicated for certain endocrine, rheumatic, collagen, allergic, ophthalmic, respiratory, hematologic, neoplastic, edematous, gastrointestinal, and other conditions. A hydrocortisone enema is indicated for ulcerative colitis, a topical ointment with antibiotics is indicated for corticosteroid responsive dermatoses with infections, and a topical cream with [acyclovir] is indicated to treat cold sores. Oral granules of hydrocortisone are used as a replacement therapy for Adrenocortical Insufficiency (AI) in children under 17 years of age.
FDA Label
Replacement therapy of adrenal insufficiency in infants, children and adolescents (from birth to < 18 years old).
Treatment of adrenal insufficiency in adults.
Treatment of adrenocortical insufficiency
Treatment of congenital adrenal hyperplasia (CAH) in adolescents aged 12 years and over and adults.
Hydrocortisone binds to the glucocorticoid receptor leading to downstream effects such as inhibition of phospholipase A2, NF-kappa B, other inflammatory transcription factors, and the promotion of anti-inflammatory genes. Hydrocortisone has a wide therapeutic index and a moderate duration of action. Patients should stop taking the medication if irritation or sensitization occurs.
Anti-Inflammatory Agents
Substances that reduce or suppress INFLAMMATION. (See all compounds classified as Anti-Inflammatory Agents.)
H02AB09
H02AB09
H02AB09
D07AA02
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
A - Alimentary tract and metabolism
A01 - Stomatological preparations
A01A - Stomatological preparations
A01AC - Corticosteroids for local oral treatment
A01AC03 - Hydrocortisone
A - Alimentary tract and metabolism
A07 - Antidiarrheals, intestinal antiinflammatory/antiinfective agents
A07E - Intestinal antiinflammatory agents
A07EA - Corticosteroids acting locally
A07EA02 - Hydrocortisone
C - Cardiovascular system
C05 - Vasoprotectives
C05A - Agents for treatment of hemorrhoids and anal fissures for topical use
C05AA - Corticosteroids
C05AA01 - Hydrocortisone
D - Dermatologicals
D07 - Corticosteroids, dermatological preparations
D07A - Corticosteroids, plain
D07AA - Corticosteroids, weak (group i)
D07AA02 - Hydrocortisone
D - Dermatologicals
D07 - Corticosteroids, dermatological preparations
D07X - Corticosteroids, other combinations
D07XA - Corticosteroids, weak, other combinations
D07XA01 - Hydrocortisone
H - Systemic hormonal preparations, excl. sex hormones and insulins
H02 - Corticosteroids for systemic use
H02A - Corticosteroids for systemic use, plain
H02AB - Glucocorticoids
H02AB09 - Hydrocortisone
S - Sensory organs
S01 - Ophthalmologicals
S01B - Antiinflammatory agents
S01BA - Corticosteroids, plain
S01BA02 - Hydrocortisone
S - Sensory organs
S01 - Ophthalmologicals
S01C - Antiinflammatory agents and antiinfectives in combination
S01CB - Corticosteroids/antiinfectives/mydriatics in combination
S01CB03 - Hydrocortisone
S - Sensory organs
S02 - Otologicals
S02B - Corticosteroids
S02BA - Corticosteroids
S02BA01 - Hydrocortisone
Absorption
Oral hydrocortisone at a dose of 0.2-0.3mg/kg/day reached a mean Cmax of 32.69nmol/L with a mean AUC of 90.63h\*nmol/L A 0.4-0.6mg/kg/day dose reached a mean Cmax of 70.81nmol/L with a mean AUC of 199.11h\*nmol/L. However, the pharmacokinetics of hydrocortisone can vary by 10 times from patient to patient. Topical hydrocortisone cream is 4-19% bioavailable[8546995] with a Tmax of 24h. Hydrocortisone retention enemas are have a bioavailability of 0.810 for slow absorbers and 0.502 in rapid absorbers. Slow absorbers take up hydrocortisone at a rate of 0.3610.255/h while fast absorbers take up hydrocortisone at a rate of 1.050.255/h. A 20mg IV dose of hydrocortisone has an AUC of 1163277ng\*h/mL.
Route of Elimination
Corticosteroids are eliminated predominantly in the urine. However, data regarding the exact proportion is not readily available.
Volume of Distribution
Total hydrocortisone has a volume of distribution of 39.82L, while the free fraction has a volume of distribution of 474.38L.
Clearance
Total hydrocortisone by the oral route has a mean clearance of 12.85L/h, while the free fraction has a mean clearance of 235.78L/h. A 20mg IV dose of hydrocortisone has a clearance of 18.24.2L/h.
Following percutaneous penetration of a topical corticosteroid, the drug that is systemically absorbed probably follows the metabolic pathways of systemically administered corticosteroids. Corticosteroids usually are metabolized in the liver and excreted by the kidneys. Some topical corticosteroids and their metabolites are excreted in bile. /Topical corticosteroids/
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3525
Topical application of corticosteroids to the mucosa of the genitourinary or lower intestinal tract may result in substantial systemic absorption of the drugs. In healthy individuals, as much as 30-90% of rectally administered hydrocortisone as a retention enema may be absorbed. Greater amounts of hydrocortisone may be absorbed rectally if the intestinal mucosa is inflamed.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3525
Following topical application of a corticosteroid to most areas of normal skin, only minimal amounts of the drug reach the dermis and subsequently the systemic circulation; however, absorption is markedly increased when the skin has lost its keratin layer and can be increased by inflammation and/or diseases of the epidermal barrier (e.g., psoriasis, eczema). The drugs are absorbed to a greater degree from the scrotum, axilla, eyelid, face, and scalp than from the forearm, knee, elbow, palm, and sole. Even after washing the area being treated, prolonged absorption of the corticosteroid occurs, possibly because the drug is retained in the stratum corneum. /Topical corticosteroids/
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3525
Percutaneous penetration of corticosteroids varies among individual patients and can be increased by the use of occlusive dressings, by increasing the concentration of the corticosteroid, and by using different vehicles. The use of an occlusive dressing with hydrocortisone for 96 hours substantially enhances percutaneous penetration of the drug; however, such use for up to 24 hours does not appear to alter penetration of topically applied hydrocortisone.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3525
For more Absorption, Distribution and Excretion (Complete) data for HYDROCORTISONE (15 total), please visit the HSDB record page.
Hydrocortisone is metabolised to 6-beta hydrocortisol via CYP3A, 5-beta tetrahydrocortisol via 3-oxo-5-beta-steroid 4-dehydrogenase, 5-alpha tetrahydrocortisol via 3-oxo-5-alpha-steroid 4-dehydrogenase 2, cortisone via Corticosteroid 11-beta-dehydrogenase isozyme 1 and Corticosteroid 11-beta-dehydrogenase isozyme 2, and glucuronide products. Cortisone is further metabolized to tetrahydrocortisone and dihydrocortisol.
A study was made of the absorption of exogenous hydrocortisone and formation of its metabolites in isolated liver of intact and exposed rats in conditions of recirculating perfusion. It was shown that the absorption of the hormone by the liver of irradiated rats was greatly lowered but the content of most metabolites found in the perfused medium of irradiated liver increased as compared to the control. It is suggested that irradiation inhibits subsequent transformations of the hydrocortisone metabolism products.
PMID:4001319 Litskevich LA, Dokshina GA; Radiobiologiia 25 (2): 200-3 (1985)
Subcellular distribution of (3)H-hydrocortisone and its metabolites in the liver and kidney of intact and alloxan diabetic rats was investigated. Ten minutes after the administration of this hormone several metabolites (mostly tetrahydrocortisol) and the native hormone were found in liver cytosol, microsomes, mitochondria and nuclei, the relative content of individual compounds in various subcellular fractions being different. In liver mitochondria, microsomes and nuclei of alloxan diabetic rats the concentration of tetrahydrocortisol was decreased, while that of native hormone was increased as compared to normal animals. It was suggested that such changes found in diabetic animals may be one of the causes of increased sensitivity of transcription and translation processes to glucocorticoids. In kidney cytosol and microsomes of intact rats cortisone and tetrahydrocortisol were found. In diabetic animals, however, the concentration of tetrahydrocortisol increased, while that of cortisone was undetectable.
PMID:3259502 Minchenko AG, Tronjko ND; Endocrinol Exp 22 (1): 19-28 (1988)
Total hydrocortisone via the oral route has a half life of 2.15h while the free fraction has a half life of 1.39h. A 20mg IV dose of hydrocortisone has a terminal half life of 1.90.4h.
... After IV administration, hydrocortisone was eliminated with a total body clearance of 18 L/hr and a half-life of 1.7 hr.
PMID:2050835 Derendorf H et al; J Clin Pharmacol 31 (5): 473-6 (1991)
The short term effects of corticosteroids are decreased vasodilation and permeability of capillaries, as well as decreased leukocyte migration to sites of inflammation. Corticosteroids binding to the glucocorticoid receptor mediates changes in gene expression that lead to multiple downstream effects over hours to days. Glucocorticoids inhibit neutrophil apoptosis and demargination; they inhibit phospholipase A2, which decreases the formation of arachidonic acid derivatives; they inhibit NF-Kappa B and other inflammatory transcription factors; they promote anti-inflammatory genes like interleukin-10. Lower doses of corticosteroids provide an anti-inflammatory effect, while higher doses are immunosuppressive. High doses of glucocorticoids for an extended period bind to the mineralocorticoid receptor, raising sodium levels and decreasing potassium levels.
Following topical application, corticosteroids produce anti-inflammatory, antipruritic, and vasoconstrictor actions. The activity of the drugs is thought to result at least in part from binding with a steroid receptor. Corticosteroids decrease inflammation by stabilizing leukocyte lysosomal membranes, preventing release of destructive acid hydrolases from leukocytes; inhibiting macrophage accumulation in inflamed areas; reducing leukocyte adhesion to capillary endothelium; reducing capillary wall permeability and edema formation; decreasing complement components; antagonizing histamine activity and release of kinin from substrates; reducing fibroblast proliferation, collagen deposition, and subsequent scar tissue formation; and possibly by other mechanisms as yet unknown. Corticosteroids, especially the fluorinated corticosteroids, have antimitotic activity on cutaneous fibroblasts and the epidermis. /Corticosteroids/
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3525
Reactive oxygen species (ROS) generation by polymorphonuclear leukocytes (PMNL) and mononuclear cells (MNC) is inhibited following the intravenous administration of hydrocortisone. This is associated with a parallel decrease in intranuclear NFkappaB, known to modulate inflammatory responses including ROS generation. Plasma levels of interleukin-10 (IL-10), an anti-inflammatory and immunosuppressive cytokine produced by TH2 cells, are also increased after hydrocortisone administration. In this study, we have investigated the effect of hydrocortisone on p47(phox) subunit, a key component of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, in MNC and the pharmacodynamics of this effect with ROS generation and plasma IL-10 levels /were investigated/. p47(phox) subunit protein levels in MNC showed a progressive decrease after hydrocortisone administration. It reached a nadir at 4 hours and increased thereafter to a baseline level at 24 hours. ROS generation also decreased, reached a nadir between 2 and 4 hours, and returned to a baseline level at 24 hours. IL-10 concentrations increased, peaked at 4 hours, and reverted to the baseline levels at 24 hours. In conclusion, p47(phox) subunit suppression may contribute to the inhibition of ROS generation in MNC after hydrocortisone administration. This suppression occurs in parallel with the suppression of NFkappaB and an increase in IL-10 plasma levels. Therefore, it would appear that the decrease in intranuclear NFkappaB and an increase in IL-10 may cause the inhibitory modulation on p47(phox) subunit and ROS generation by MNC following hydrocortisone and other glucocorticoids.
PMID:11319715 Dandona P et al; Metabolism 50 (5): 548-52 (2001)

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
51
PharmaCompass offers a list of Hydrocortisone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Hydrocortisone manufacturer or Hydrocortisone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Hydrocortisone manufacturer or Hydrocortisone supplier.
PharmaCompass also assists you with knowing the Hydrocortisone API Price utilized in the formulation of products. Hydrocortisone API Price is not always fixed or binding as the Hydrocortisone Price is obtained through a variety of data sources. The Hydrocortisone Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Glycort manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Glycort, including repackagers and relabelers. The FDA regulates Glycort manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Glycort API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Glycort manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Glycort supplier is an individual or a company that provides Glycort active pharmaceutical ingredient (API) or Glycort finished formulations upon request. The Glycort suppliers may include Glycort API manufacturers, exporters, distributors and traders.
click here to find a list of Glycort suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Glycort DMF (Drug Master File) is a document detailing the whole manufacturing process of Glycort active pharmaceutical ingredient (API) in detail. Different forms of Glycort DMFs exist exist since differing nations have different regulations, such as Glycort USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Glycort DMF submitted to regulatory agencies in the US is known as a USDMF. Glycort USDMF includes data on Glycort's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Glycort USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Glycort suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Glycort Drug Master File in Japan (Glycort JDMF) empowers Glycort API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Glycort JDMF during the approval evaluation for pharmaceutical products. At the time of Glycort JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Glycort suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Glycort Drug Master File in Korea (Glycort KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Glycort. The MFDS reviews the Glycort KDMF as part of the drug registration process and uses the information provided in the Glycort KDMF to evaluate the safety and efficacy of the drug.
After submitting a Glycort KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Glycort API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Glycort suppliers with KDMF on PharmaCompass.
A Glycort CEP of the European Pharmacopoeia monograph is often referred to as a Glycort Certificate of Suitability (COS). The purpose of a Glycort CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Glycort EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Glycort to their clients by showing that a Glycort CEP has been issued for it. The manufacturer submits a Glycort CEP (COS) as part of the market authorization procedure, and it takes on the role of a Glycort CEP holder for the record. Additionally, the data presented in the Glycort CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Glycort DMF.
A Glycort CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Glycort CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Glycort suppliers with CEP (COS) on PharmaCompass.
A Glycort written confirmation (Glycort WC) is an official document issued by a regulatory agency to a Glycort manufacturer, verifying that the manufacturing facility of a Glycort active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Glycort APIs or Glycort finished pharmaceutical products to another nation, regulatory agencies frequently require a Glycort WC (written confirmation) as part of the regulatory process.
click here to find a list of Glycort suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Glycort as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Glycort API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Glycort as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Glycort and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Glycort NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Glycort suppliers with NDC on PharmaCompass.
Glycort Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Glycort GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Glycort GMP manufacturer or Glycort GMP API supplier for your needs.
A Glycort CoA (Certificate of Analysis) is a formal document that attests to Glycort's compliance with Glycort specifications and serves as a tool for batch-level quality control.
Glycort CoA mostly includes findings from lab analyses of a specific batch. For each Glycort CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Glycort may be tested according to a variety of international standards, such as European Pharmacopoeia (Glycort EP), Glycort JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Glycort USP).

