
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Europe
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
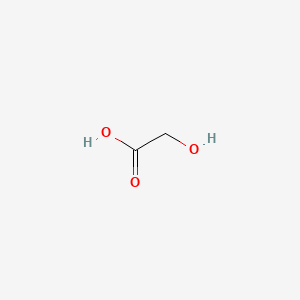

1. Glycolate
2. Glycolic Acid, 1-(14)c-labeled
3. Glycolic Acid, 2-(14)c-labeled
4. Glycolic Acid, Calcium Salt
5. Glycolic Acid, Monoammonium Salt
6. Glycolic Acid, Monolithium Salt
7. Glycolic Acid, Monopotassium Salt
8. Glycolic Acid, Monosodium Salt
9. Glycolic Acid, Potassium Salt
10. Hydroxyacetic Acid
11. Potassium Glycolate
1. 2-hydroxyacetic Acid
2. Hydroxyacetic Acid
3. 79-14-1
4. Glycollic Acid
5. Hydroxyethanoic Acid
6. Acetic Acid, Hydroxy-
7. Glycolate
8. Caswell No. 470
9. Glycocide
10. Alpha-hydroxyacetic Acid
11. Kyselina Glykolova
12. Kyselina Glykolova [czech]
13. Kyselina Hydroxyoctova
14. Hoch2cooh
15. 2-hydroxyethanoic Acid
16. Kyselina Hydroxyoctova [czech]
17. Epa Pesticide Chemical Code 000101
18. Glypure
19. Hsdb 5227
20. Nsc 166
21. Acetic Acid, 2-hydroxy-
22. Ai3-15362
23. Mfcd00004312
24. Glypure 70
25. Brn 1209322
26. Glycolic Acid Solution
27. Nsc-166
28. Acetic Acid, Hydroxy-, Homopolymer
29. 26124-68-5
30. .alpha.-hydroxyacetic Acid
31. Glycollate
32. 0wt12sx38s
33. Nsc166
34. Chebi:17497
35. Polyglycollic Acid
36. Goa
37. Glycolicacid
38. Dexon (polyester)
39. Poly(glycolic Acid)
40. Poly(l-glycolic Acid)
41. Glypure 70 Homopolymer
42. Glycolic Acid Homopolymer
43. Einecs 201-180-5
44. Unii-0wt12sx38s
45. Polyglycolide
46. Hydroxyacetic Acid Homopolymer
47. Hydroxyethanoate
48. A-hydroxyacetate
49. Ccris 9474
50. Hydroxy-acetic Acid
51. 2-hydroxyaceticacid
52. Alpha-hydroxyacetate
53. A-hydroxyacetic Acid
54. Acetic Acid, 2-hydroxy-, Homopolymer
55. Glycolic Acid 70%
56. 2-hydroxy Acetic Acid
57. 2-hydroxy-acetic Acid
58. Omega-hydroxy Fatty Acid
59. 2-hydroxyl Ethanoic Acid
60. Glycolic Acid, Polyesters
61. Ho-ch2-cooh
62. Dsstox_cid_5363
63. Hydroxyacetic Acid Solution
64. Bmse000245
65. Wln: Qv1q
66. Ec 201-180-5
67. Glycolic Acid [mi]
68. Dsstox_rid_77763
69. Glycolic Acid (7ci,8ci)
70. Dsstox_gsid_25363
71. Glycolic Acid [inci]
72. 4-03-00-00571 (beilstein Handbook Reference)
73. Glycolic Acid [vandf]
74. Glycolic Acid, P.a., 98%
75. Glycolic Acid [mart.]
76. Acetic Acid, Hydroxy- (9ci)
77. Chembl252557
78. Glycolic Acid [who-dd]
79. Dtxsid0025363
80. Glycolic Acid, Crystal, Reagent
81. Hydroxyacetic Acid [hsdb]
82. Glycolic Acid Solution, 56-58%
83. Bcp28762
84. Glycolic Acid, >=97.0% (t)
85. Str00936
86. Zinc4658557
87. Tox21_301298
88. S6272
89. Stl197955
90. Akos000118921
91. Glycolic Acid, Reagentplus(r), 99%
92. Cs-w016683
93. Db03085
94. Glycolic Acid 100 Microg/ml In Water
95. Glycolic Acid Solution, Puriss., 70%
96. Hy-w015967
97. Sb83760
98. Cas-79-14-1
99. Ncgc00160612-01
100. Ncgc00160612-02
101. Ncgc00257533-01
102. Glycolic Acid, 66-70% Aqueous Solution
103. Glycolic Acid Solution, Cp, 70% In H2o
104. Ft-0612572
105. Ft-0669047
106. G0110
107. G0196
108. Glycolic Acid 100 Microg/ml In Acetonitrile
109. Glycolic Acid, Saj Special Grade, >=98.0%
110. C00160
111. C03547
112. D78078
113. Glycolic Acid, Vetec(tm) Reagent Grade, 98%
114. Hydroxyacetic Acid; Hydroxyethanoic Acid
115. Glycolic Acid, Bioxtra, >=98.0% (titration)
116. Glycolic Acid Solution, Technical, ~55% In H2o
117. Q409373
118. J-509661
119. F2191-0224
120. Glycolic Acid Solution, High Purity, 70 Wt. % In H2o
121. Hydroxyacetic Acid; Hydroxyethanoic Acid; Glycollic Acid
122. Z1259155884
123. 287eb351-ff9f-4a67-b4b9-d626406c9b13
124. Glycolic Acid Solution, Technical Grade, 70 Wt. % In H2o
125. Glycolic Acid, Certified Reference Material, Tracecert(r)
126. Glycolic Acid, Anhydrous, Free-flowing, Redi-dri(tm), Reagentplus(r), 99%
127. Glycolic Acid, Pharmaceutical Secondary Standard; Certified Reference Material
128. O7z
| Molecular Weight | 76.05 g/mol |
|---|---|
| Molecular Formula | C2H4O3 |
| XLogP3 | -1.1 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Exact Mass | 76.016043985 g/mol |
| Monoisotopic Mass | 76.016043985 g/mol |
| Topological Polar Surface Area | 57.5 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 40.2 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Keratolytic Agents
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Glycolic acid is a member of the alpha-hydroxy acid (AHA) family, which ... has been used for centuries as a cutaneous rejuvenation treatment. Recently it has proved to be a versatile peeling agent and it is now widely used to treat many defects of the epidermis and papillary dermis in a variety of strengths, ranging from 20% to 70%, depending on the condition being treated. People of almost any skin type and color are candidates, and almost any area of the body can be peeled...
PMID:7600706 Murad H et al; Dermatol Clin 13 (2): 285-307 (1995)
Glycolic acid has been used by dermatologists for years to treat skin disorders and is a component of many over-the-counter personal care products. No systemic toxicity has been noted as a result of these uses.
Hayes AW, Stadler JC; Toxicologist 78 (1-S): 160 (2004)
Chemical peeling, also known as chemoexfoliation or dermapeeling, is performed to improve the skin's appearance as it reduces the wrinkles caused by aging and the features of photoaged skin. Although the best results are obtained with deep /phenol/ peels, the medium-depth peels allow to obtain excellent results without the dangerous side effects of deep peels. Medium-depth peelings are performed with trichloroacetic acid (TCA) at 35-50% alone or at 35% in combination with Jessner's solution, 70% glycolic acid, and solid CO(2)...
PMID:17166210 Camacho FM; J Cosmet Dermatol 4 (2): 117-28 (2005)
For more Therapeutic Uses (Complete) data for HYDROXYACETIC ACID (27 total), please visit the HSDB record page.
FDA has considered evidence that suggests that topically applied cosmetic products containing alpha hydroxy acids (AHAs) as ingredients may increase the sensitivity of skin to the sun while the products are used and for up to a week after use is stopped, and that this increased skin sensitivity to the sun may increase the possibility of sunburn. ... As an interim measure, while FDA continues to review the data on AHAs to address the potential for this increased skin sensitivity to the sun, FDA is recommending that the labeling of a cosmetic product that contains an AHA as an ingredient and that is topically applied to the skin or mucous membrane bear a statement that conveys the following information. The information in the AHA labeling statement is consistent with FDA's current thinking on sun protection. Sunburn Alert: This product contains an alpha hydroxy acid (AHA) that may increase your skin's sensitivity to the sun and particularly the possibility of sunburn. Use a sunscreen, wear protective clothing, and limit sun exposure while using this product and for a week afterwards. /Alpha hydroxy acids/
FDA; Center for Drug Evaluation and Research; Guidance for Industry. Labeling for Topically Applied Cosmetic Products Containing Alpha Hydroxy Acids as Ingredients. (January 10, 2005). Available from, as of July 30, 2008: https://www.cfsan.fda.gov/~dms/ahaguid2.html
1989-1996 Consumer adverse experience reports that were submitted to FDA headquarters and to FDA district offices on alpha hydroxy acid (AHA)-containing products /were evaluated/. Typical adverse reactions included "severe redness, swelling (especially in the area of the eyes), burning, blistering, bleeding, scarring, rash, itching, contact dermatitis, skin discoloration (reportedly permanent), and adverse neurological responses." Some of the individuals submitting an adverse experience report were seen by a physician, and at least one adverse report involved professional application and at least one involved a product prescribed by a dermatologist. FDA's submittal stated that "in addition to consumer reports of adverse reactions, letters have also been received from dermatologists treating patients suffering from injuries resulting from the use of these (AHA-containing) products". /Alpha hydroxy acids/
Cosmetic Ingredient Review; Final Report on the Safety Assessment of Glycolic Acid, Ammonium, Calcium, Potassium, and Sodium Glycolates, Methyl, Ethyl, Propyl, and Butyl Glycolates, and Lactic Acid, Ammonium, Calcium, Potassium, Sodium, and TEA-Lactates, Methyl, Ethyl, Isopropyl, and Butyl Lactate, and Lauryl, Myristyl, and Cetyl Lactates; Journal of American College of Toxicology 17(Suppl 1):1-242 (1998)
Keratolytic Agents
Agents that soften, separate, and cause desquamation of the cornified epithelium or horny layer of skin. They are used to expose mycelia of infecting fungi or to treat corns, warts, and certain other skin diseases. (See all compounds classified as Keratolytic Agents.)
The penetration of 10% aq. glycolic acid, adjusted to pH 3.8 using either ammonium or sodium hydroxide, was examined using separated Yucatan minipig epidermis and full thickness hairless mouse skin. A 200 uL-aliquot of each formulation was applied to an area of a Franz diffusion cell, and glycolic acid was analyzed using liquid scintillation counting. Using an occlusive patch, penetration was linear with a lag time of less than 15 mm. After 8 hr, 0.8 and 1.6% of the ammonium and sodium salts penetrated, respectively, using the pig skin model and 1.8 and 2.3% of the ammonium and sodium salts penetrated, respectively, using the mouse skin model. Under open patch conditions, penetration was not linear and lag time was greater than 15 mm. Using the pig skin model, 1.1 and 0.7% of the ammonium and sodium salts penetrated, respectively, and using the mouse skin model, 0.6 and 0.9% of the ammonium and sodium salts penetrated, respectively.
Cosmetic Ingredient Review; Final Report on the Safety Assessment of Glycolic Acid, Ammonium, Calcium, Potassium, and Sodium Glycolates, Methyl, Ethyl, Propyl, and Butyl Glycolates, and Lactic Acid, Ammonium, Calcium, Potassium, Sodium, and TEA-Lactates, Methyl, Ethyl, Isopropyl, and Butyl Lactate, and Lauryl, Myristyl, and Cetyl Lactates; Journal of American College of Toxicology 17(Suppl 1):1-242 (1998).
The skin penetration of (14)C-glycolic acid was studied using an in vitro system in which a cream formulation was applied to pig skin at a dose of 5 mg/0.79 sq cm skin without an occlusive patch. It was determined that 3.1% of the applied glycolic acid penetrated the skin.
Cosmetic Ingredient Review; Final Report on the Safety Assessment of Glycolic Acid, Ammonium, Calcium, Potassium, and Sodium Glycolates, Methyl, Ethyl, Propyl, and Butyl Glycolates, and Lactic Acid, Ammonium, Calcium, Potassium, Sodium, and TEA-Lactates, Methyl, Ethyl, Isopropyl, and Butyl Lactate, and Lauryl, Myristyl, and Cetyl Lactates; Journal of American College of Toxicology 17(Suppl 1):1-242 (1998).
Two female rhesus monkeys were dosed orally with 4 mL/kg of 500 mg/kg homogenous 1-(14)C-glycolic acid, 0.73 uC/mmol, in aq. solution via stomach tube. Urine was collected at intervals of 0-8, 8-24, 24-48, 48-72, and, for one monkey, 72-96 hr. Over a 72 hr period one animal excreted, as a percentage of the dose, 53.2% (14)C, 51.4% of which was excreted in the urine; 51.4% of the dose was excreted in the first 24 hr. The second animal excreted a total of 42.2% (14)C over 96 hr, 36.6% of which was excreted in the urine; 34.1% of the dose was excreted in the first 24 hr. (The greater amount of fecal radioactivity observed for this monkey could have been due to urinary radioactivity contamination.) Very little of the dose was converted to radioactive glyoxylic, hippuric, or oxalic acid.
Cosmetic Ingredient Review; Final Report on the Safety Assessment of Glycolic Acid, Ammonium, Calcium, Potassium, and Sodium Glycolates, Methyl, Ethyl, Propyl, and Butyl Glycolates, and Lactic Acid, Ammonium, Calcium, Potassium, Sodium, and TEA-Lactates, Methyl, Ethyl, Isopropyl, and Butyl Lactate, and Lauryl, Myristyl, and Cetyl Lactates; Journal of American College of Toxicology 17(Suppl 1):1-242 (1998).
Skin penetration of 10% aq. Glycolic acid was determined in vitro using human female (age 87 years) abdominal skin. The aq. solution was prepared by adding 0.8 mL 12.473% glycolic acid solution to 0.2 mL of (2-(14)C) glycolic acid solution, 44 mCi/mmol or 250 iCi/mL that contained 0.216 mg glycolic acid. The pH of a mixture containing 0.8 mL of the 12.473% glycolic acid solution and 0.2 mL of water was 3.72. Skin integrity was assessed by determining the permeability coefficient of tritiated water. Twenty uL of 10% aq. glycolic acid solution, 2 mg active, was placed on the stratum corneum surface; 13 replicates were used. Samples of 200 uL, which were taken 1, 2, 4, 6, 8, and 24 hr after application, were counted using a liquid scintillation counter. The skin surface was rinsed 3 times after the 24 hr sample was taken. The average total absorption over 24 hr 2.6 +/= 0.37 ug/sq cm representing 0.15 +/= 0.02% of the applied dose. A lag time of approximately 3.8 hr was followed by a period of steady-state diffusion at a rate of 0.13 ug/sq cm/hr. After 24 hr, 48 +/= 0.05% of the dose was recovered in the skin and 0.15 +/= 0.02% was found in the receptor phase. Total recovery was 102.9% +/= 2.9%.
Cosmetic Ingredient Review; Final Report on the Safety Assessment of Glycolic Acid, Ammonium, Calcium, Potassium, and Sodium Glycolates, Methyl, Ethyl, Propyl, and Butyl Glycolates, and Lactic Acid, Ammonium, Calcium, Potassium, Sodium, and TEA-Lactates, Methyl, Ethyl, Isopropyl, and Butyl Lactate, and Lauryl, Myristyl, and Cetyl Lactates; Journal of American College of Toxicology 17(Suppl 1):1-242 (1998).
For more Absorption, Distribution and Excretion (Complete) data for HYDROXYACETIC ACID (14 total), please visit the HSDB record page.
The kinetics of orally administered ethylene glycol (EG) and its major metabolites, glycolic acid (GA) and oxalic acid (OX), in pregnant (P; gestation day 10 at dosing, GD 10) rats were compared across doses, and between pregnant and nonpregnant (NP) rats. Groups of 4 jugular vein-cannulated female rats were administered 10 (P and NP), 150 (P), 500 (P), 1000 (P), or 2500 (P and NP) mg (13)C-labelled EG/kg body weight. Serial blood samples and urine were collected over 24-hr postdosing, and analyzed for EG, GA, and OX using GC/MS techniques. Pharmacokinetic parameters including Cmax, Tmax, AUC, and beta-t(1/2) were determined for EG and GA. Pregnancy status (GD 10-11) had no impact on the pharmacokinetic parameters investigated. Blood levels of GA were roughly dose-proportional from 10 to 150 mg EG/kg, but increased disproportionately from 500 to 1000 mg EG/kg. EG and GA exhibited dose-dependent urinary elimination at doses > or = 500 mg EG/kg, probably due to saturation of metabolic conversion of EG to GA, and of GA to downstream metabolites. The shift to nonlinear kinetics encompassed the NOEL (500 mg EG/kg) and LOEL (1000 mg EG/kg) for developmental toxicity of EG in rats, providing additional evidence for the role of GA in EG developmental toxicity. The peak maternal blood concentration of GA associated with the LOEL for developmental toxicity in the rat was quite high (363 microg/g or 4.8 mM blood). OX was a very minor metabolite in both blood and urine at all dose levels, suggesting that OX is not important for EG developmental toxicity.
PMID:11399788 Pottenger LH et al; Toxicol Sci 62 (1): 10-9 (2001)
The disposition of dichloroacetic acid (DCA) was investigated in Fischer 344 rats over the 48 hr after oral gavage of 282 mg/kg of 1- or 2-(14C)DCA (1-DCA or 2-DCA) and 28.2 mg/kg of 2-DCA... The major urinary metabolites were glycolic acid, glyoxylic acid, and oxalic acid. DCA and its metabolites accumulated in the tissues and were eliminated slowly....
PMID:8421320 Lin EL et al; J Toxicol Environ Health 38 (1): 19-32 (1993)
The accumulation of glycolate and the elimination kinetics of ethylene glycol (EG) /was examined in/ ... male Sprague-Dawley rats and mixed breed dogs... . EG was administered by gavage ... . The peak plasma level of EG occurred at 2 hr after dosing and that of glycolate between 4-6 hr. The rate of EG elimination was somewhat faster in rats with a half-life of 1.7 hr compared to 3.4 hr in dogs. The maximum plasma level of glycolate was greater in rats, although the pattern of accumulation was similar to that in dogs. Glycolate disappeared from the plasma at the same time as EG, suggesting a slower rate of elimination of the metabolite than that of EG. Renal excretion of EG was an important route for its elimination, accounting for 20-30% of the dose. Renal excretion of glycolate represented about 5% of the dose... /Glycolate/
PMID:2929116 Hewlett TP et al; Vet Hum Toxicol 31 (2): 116-20 (1989)
1,2-(14)C-Ethylene glycol (EG) was given to female CD (Sprague-Dawley) rats and CD-1 mice in order to determine tissue distribution and metabolic fate after intravenous (iv), peroral (po), and percutaneous (pc) doses. Rats were given doses of 10 or 1000 mg/kg by each route, and additional pc doses of 400, 600 or 800 mg/kg. Mice were also given iv and po doses of 10 or 1000 mg/kg, and intermediate po doses of 100, 200 or 400 mg/kg. Mice were given po doses of 100 or 1000 mg/kg, and both species were given a 50% (w/w) aqueous po dose to simulate antifreeze exposure. For both species, EG is very rapidly and almost completely adsorbed after po doses. ... The tissue distribution of EG following either iv or po routes was essentially the same, with similar percentages recovered for each dose by both routes and for either species. Cutaneously-applied EG was slowly and rather poorly adsorbed in both species, in comparison with po-dose administration, and urinalysis after undiluted po doses indicated that EG probably penetrates rat skin in the parent form. There was an absence in both species of dose-dependent changes in disposition and elimination following the pc application of EG. (14)C-labelled EG, glycolic acid and/or oxalic acid accounted for the majority of the detectable radioactivity in the urine samples from all dose routes in the rat, while glycoaldehyde and glyoxylic acid were not detected in any of the urine fractions evaluated. Similar increases in glycolate production with increasing dose were also observed in mouse urine samples from iv and po dosing. Also, glyoxylate and oxalate were absent from mouse urine...
PMID:8948094 Frantz SW et al; Xenobiotica 26 (11): 1195-220 (1996)
For more Metabolism/Metabolites (Complete) data for HYDROXYACETIC ACID (9 total), please visit the HSDB record page.
... ethylene glycol and glycolate were distributed in total body water with plasma half-lives of 8.4 and 7.0 hr respectively.
Jacobsen D et al; Am J Med 84: 145-52 (Jan) (1988)
Rats given 1, 5, and 10 mL/kg diethylene glycol eliminated diethylene glycol in their urine with half lives of 6, 6, and 12 hr assuming first order kinetics. More detailed analysis showed that 6, 9, and 18 hr after dosing with 1, 5, and 10 mL/kg diethylene glycol elimination of (14)C activity followed zero order kinetics then changed to first order kinetics with a half life of 3 hr. Rats dosed with 3 and 5 mL/kg ethylene glycol excreted unchanged ethylene glycol in their urine with half lives of 4.5 and 4.1 hr respectively.
PMID:2815837 Lenk W et al; Xenobiotica 19 (9): 961-79 (1989)
Ethylene glycol toxicity results from its metabolism to glycolic acid and other toxic metabolites. The accumulation of glycolate and the elimination kinetics of ethylene glycol and its metabolites are not well understood, so studies with male Sprague-Dawley rats and mixed breed dogs have been carried out. Ethylene glycol was administered by gavage to rats and dogs which were placed in metabolic cages for urine and blood sample collection at timed intervals. The peak plasma level of ethylene glycol occurred at 2 hr after dosing and that of glycolate between 4-6 hr. The rate of ethylene glycol elimination was somewhat faster in rats with a half-life of 1.7 hr compared to 3.4 hr in dogs. The maximum plasma level of glycolate was greater in rats although the pattern of accumulation was similar to that in dogs. Glycolate disappeared from the plasma at the same time as ethylene glycol, suggesting a slower rate of elimination of the metabolite than that of ethylene glycol. Renal excretion of ethylene glycol was an important route for its elimination accounting for 20-30% of the dose. Renal excretion of glycolate represented about 5% of the dose. Ethylene glycol induced an immediate, but short lived diuresis compared to that in control rats. Minimal clinical effects (mild acidosis with no sedation) were noted at these doses of ethylene glycol (1-2 g/kg) in both rats and dogs. The results indicate that the toxicokinetics of ethylene glycol and glycolate were similar in both species.
PMID:2929116 Hewlett TP et al; Vet Hum Toxicol 31 (2): 116-20 (1989)
The effect of 0.35 to 0.8 mmol/kg glycolic acid and 1.0 to 4.4 mmol/kg sodium glycolate on cyclopropane-epinephrine induced cardiac arrhythmias was examined using dogs. Doses of 0.35 to 0.5 mmol/kg glycolic acid increased the duration of arrhythmias in the 13 dogs tested, whereas doses >0.5 mmol/kg decreased or totally eliminated the arrhythmias in each of 11 dogs. Depression was observed for many of the dogs at higher doses. Sodium glycolate was much less effective in decreasing the arrhythmias, with 3 mmol/kg being required and its action being transient.
Cosmetic Ingredient Review; Final Report on the Safety Assessment of Glycolic Acid, Ammonium, Calcium, Potassium, and Sodium Glycolates, Methyl, Ethyl, Propyl, and Butyl Glycolates, and Lactic Acid, Ammonium, Calcium, Potassium, Sodium, and TEA-Lactates, Methyl, Ethyl, Isopropyl, and Butyl Lactate, and Lauryl, Myristyl, and Cetyl Lactates; Journal of American College of Toxicology 17(Suppl 1):1-242 (1998).
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
85
PharmaCompass offers a list of Glycolic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Glycolic Acid manufacturer or Glycolic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Glycolic Acid manufacturer or Glycolic Acid supplier.
A Glycolic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Glycolic Acid, including repackagers and relabelers. The FDA regulates Glycolic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Glycolic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Glycolic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Glycolic Acid supplier is an individual or a company that provides Glycolic Acid active pharmaceutical ingredient (API) or Glycolic Acid finished formulations upon request. The Glycolic Acid suppliers may include Glycolic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Glycolic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
Glycolic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Glycolic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Glycolic Acid GMP manufacturer or Glycolic Acid GMP API supplier for your needs.
A Glycolic Acid CoA (Certificate of Analysis) is a formal document that attests to Glycolic Acid's compliance with Glycolic Acid specifications and serves as a tool for batch-level quality control.
Glycolic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Glycolic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Glycolic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Glycolic Acid EP), Glycolic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Glycolic Acid USP).

