
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
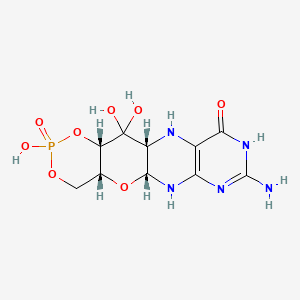

1. Alxn1101
2. Cpmp Cpd
3. Fosdenopterin
4. Nulibry
1. Fosdenopterin
2. Cpmp
3. Precursor Z Hydrate
4. C(pmp)
5. Alxn1101
6. 150829-29-1
7. Precursor Z, Hydrated
8. Fosdenopterin [usan]
9. Chebi:60210
10. 4x7k2681y7
11. Alxn-1101 Anhydrous Free Base
12. Fosdenopterin (usan)
13. (4ar,5ar,11ar,12as)-8-amino-2,12,12-trihydroxy-4a,5a,6,9,11,11a,12,12a-octahydro[1,3,2]dioxaphosphinino[4',5':5,6]pyrano[3,2-g]pteridin-10(4h)-one 2-oxide
14. Unii-4x7k2681y7
15. Fosdenopterin [inn]
16. Fosdenopterin [who-dd]
17. Cyclic Pyranopterin Monophoshate
18. Chembl2338675
19. Schembl16156371
20. Dtxsid90934067
21. Who 11150
22. Zinc34962340
23. Hy-109145
24. Cs-0115961
25. C18239
26. D11779
27. Q5198226
28. (1r,10r,12s,17r)-5-amino-11,11,14-trihydroxy-14-oxo-13,15,18-trioxa-2,4,6,9-tetraza-14lambda5-phosphatetracyclo[8.8.0.03,8.012,17]octadeca-3(8),4-dien-7-one
29. 1,3,2-dioxaphosphorino(4',5':5,6)pyrano(3,2-g)pteridin-10(4h)-one, 8-amino-4a,5a,6,7,11,11a,12,12a-octahydro-2,12,12-trihydroxy-, 2-oxide, (4ar,5ar,11ar,12as)-
| Molecular Weight | 363.22 g/mol |
|---|---|
| Molecular Formula | C10H14N5O8P |
| XLogP3 | -4 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 0 |
| Exact Mass | 363.05799942 g/mol |
| Monoisotopic Mass | 363.05799942 g/mol |
| Topological Polar Surface Area | 197 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 722 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Fosdenopterin is indicated to reduce the risk of mortality in patients with molybdenum cofactor deficiency (MoCD) type A.
NULIBRY is indicated for the treatment of patients with molybdenum cofactor deficiency (MoCD) Type A.
Fosdenopterin replaces an intermediate substrate in the synthesis of molybdenum cofactor, a compound necessary for the activation of several molybdenum-dependent enzymes including sulfite oxidase (SOX). Given that SOX is responsible for detoxifying sulfur-containing acids and sulfites such as S-sulfocysteine (SSC), urinary levels of SSC can be used as a surrogate marker of efficacy for fosdenopterin. Long-term therapy with fosdenopterin has been shown to result in a sustained reduction in urinary SSC normalized to creatinine. Animal studies have identified a potential risk of phototoxicity in patients receiving fosdenopterin - these patients should avoid or minimize exposure to sunlight and/or artificial UV light. If sun exposure is necessary, use protective clothing, hats, and sunglasses, in addition to seeking shade whenever practical. Consider the use of a broad-spectrum sunscreen in patients 6 months of age or older.
A16AX19
A - Alimentary tract and metabolism
A16 - Other alimentary tract and metabolism products
A16A - Other alimentary tract and metabolism products
A16AX - Various alimentary tract and metabolism products
A16AX19 - Fosdenopterin
Absorption
In healthy adult subjects, the observed Cmax and AUC0-inf following the intravenous administration of 0.68 mg/kg (0.76x the maximum recommended dose) were 2800 ng/mL and 5960 ng*h/mL, respectively. Both Cmax and AUC0-inf appear to increase proportionally with increasing doses.
Route of Elimination
Renal clearance of fosdenopterin accounts for approximately 40% of total clearance.
Volume of Distribution
The volume of distribution of fosdenopterin is approximately 300 mL/kg.
Clearance
Total body clearance of fosdenopterin ranges from 167 to 195 mL/h/kg.
Fosdenopterin metabolism occurs mainly via non-enzymatic degradation into Compound Z, which is a pharmacologically inactive product of endogenous cyclic pyranopterin monophosphate.
The mean half-life of fosdenopterin ranges from 1.2 to 1.7 hours.
Molybdenum cofactor deficiency (MoCD) is a rare autosomal-recessive disorder in which patients are deficient in three molybdenum-dependent enzymes: sulfite oxidase (SOX), xanthine dehydrogenase, and aldehyde dehydrogenase. The loss of SOX activity appears to be the main driver of MoCD morbidity and mortality, as the build-up of neurotoxic sulfites typically processed by SOX results in rapid and progressive neurological damage. In MoCD type A, the disorder results from a mutation in the _MOCS1_ gene leading to deficient production of MOCS1A/B, a protein that is responsible for the first step in the synthesis of molybdenum cofactor: the conversion of guanosine triphosphate into cyclic pyranopterin monophosphate (cPMP). Fosdenopterin is an exogenous form of cPMP, replacing endogenous production and allowing for the synthesis of molybdenum cofactor to proceed.
Patents & EXCLUSIVITIES
ABOUT THIS PAGE
84
PharmaCompass offers a list of Fosdenopterin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Fosdenopterin manufacturer or Fosdenopterin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Fosdenopterin manufacturer or Fosdenopterin supplier.
A Fosdenopterin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Fosdenopterin, including repackagers and relabelers. The FDA regulates Fosdenopterin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Fosdenopterin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Fosdenopterin supplier is an individual or a company that provides Fosdenopterin active pharmaceutical ingredient (API) or Fosdenopterin finished formulations upon request. The Fosdenopterin suppliers may include Fosdenopterin API manufacturers, exporters, distributors and traders.
click here to find a list of Fosdenopterin suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Fosdenopterin as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Fosdenopterin API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Fosdenopterin as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Fosdenopterin and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Fosdenopterin NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Fosdenopterin suppliers with NDC on PharmaCompass.
Fosdenopterin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Fosdenopterin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Fosdenopterin GMP manufacturer or Fosdenopterin GMP API supplier for your needs.
A Fosdenopterin CoA (Certificate of Analysis) is a formal document that attests to Fosdenopterin's compliance with Fosdenopterin specifications and serves as a tool for batch-level quality control.
Fosdenopterin CoA mostly includes findings from lab analyses of a specific batch. For each Fosdenopterin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Fosdenopterin may be tested according to a variety of international standards, such as European Pharmacopoeia (Fosdenopterin EP), Fosdenopterin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Fosdenopterin USP).

