
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
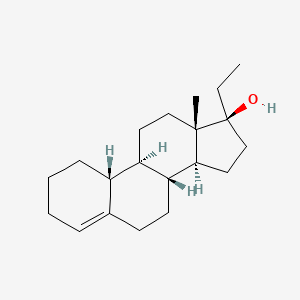

1. Ethyloestrenol
2. Maxibolin
1. Maxibolin
2. Orabolin
3. Ethylnandrol
4. Duraboral
5. Durabolin-o
6. Ethylestrenolum
7. Etilestrenol
8. Ethyloestrenol
9. 965-90-2
10. Neodurabolin
11. Maxibalin
12. Orgabolin
13. Orgaboral
14. 17alpha-ethylestr-4-en-17beta-ol
15. 17alpha-ethyl-17beta-hydroxy-4-estrene
16. 19-nor-17alpha-pregn-4-en-17beta-ol
17. 17beta-hydroxy-17alpha-ethyl-19-nor-4-androstene
18. (17alpha)-19-norpregn-4-en-17-ol
19. Adc79ek5q8
20. Chebi:31578
21. Ethylnandrol (jan)
22. Nsc-37726
23. Ncgc00167514-01
24. Org-483
25. Ethyloestrenolum
26. Nsc 37726
27. Dsstox_cid_3024
28. Ethylnandrol [jan]
29. Dsstox_rid_76835
30. Dsstox_gsid_23024
31. (8r,9s,10r,13s,14s,17s)-17-ethyl-13-methyl-2,3,6,7,8,9,10,11,12,14,15,16-dodecahydro-1h-cyclopenta[a]phenanthren-17-ol
32. Etilestrenolo [dcit]
33. Etilestrenolo
34. (17beta)-17-ethylestr-4-en-17-ol
35. Etilestrenol [inn-spanish]
36. Ethylestrenolum [inn-latin]
37. Cas-965-90-2
38. Smr000238204
39. Hsdb 3327
40. 19-norpregn-4-en-17-ol, (17alpha)-
41. Einecs 213-523-6
42. Estr-4-en-17beta-ol, 17-ethyl-
43. Unii-adc79ek5q8
44. (17-alpha)-19-norpregn-4-en-17-ol
45. Ethylestrenol [usan:inn:ban]
46. Orabolin (tn)
47. 19-nor-17alpha-pregn-4-en-17-ol
48. 19-nor-17.alpha.-pregn-4-en-17.beta.-ol
49. 19-nor-17-alpha-pregn-4-en-17-ol
50. 19-norpregn-4-en-17-ol, (17-alpha)-
51. Ethylestrenol [mi]
52. Ethylestrenol [inn]
53. Ethylestrenol (usan/inn)
54. Ethylestrenol [hsdb]
55. Ethylestrenol [usan]
56. Ethylestrenol [vandf]
57. Mls000759411
58. Mls001424123
59. Ethylestrenol [mart.]
60. Schembl147908
61. Ethylestrenol [who-dd]
62. Gtpl6948
63. Chembl1200623
64. Dtxsid6023024
65. Hms2051p08
66. Ethylestrenol [orange Book]
67. Zinc4215863
68. Tox21_112512
69. Tox21_112512_1
70. Ccg-101014
71. Db01493
72. Nc00264
73. Ncgc00167514-02
74. D01414
75. Q764283
76. 19-norpregn-4-en-17-ol, (17.alpha.)
77. W-100138
| Molecular Weight | 288.5 g/mol |
|---|---|
| Molecular Formula | C20H32O |
| XLogP3 | 5.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Exact Mass | 288.245315640 g/mol |
| Monoisotopic Mass | 288.245315640 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 453 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anabolic Steroids
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
/Ethylestrenol/ has been used in for the promotion of growth in boys with short stature or delayed bone growth.
Reynolds, J.E.F., Prasad, A.B. (eds.) Martindale-The Extra Pharmacopoeia. 28th ed. London: The Pharmaceutical Press, 1982., p. 1554
Use of anabolic steroids by athletes is not recommended. Objective evidence is conflicting and inconclusive as to whether these medications significantly increase athletic performance by increasing muscle strength. Weight gains reported by athletes are due in part to fluid retention, which is a potentially hazardous side effect of anabolic steroid therapy. The risk of other unwanted effects, such as testicular atrophy and suppression of spermatogenesis in males; menstrual disturbances and virilization, such as deepening of voice, development of acne, and unnatural growth of body hair in females; peliosis hepatis or other hepatotoxicity; and hepatic cancer outweigh any possible benefit received from anabolic steroids and make their use in athletes inappropriate. /Anabolic Steroids/
Thomson.Micromedex. Drug Information for the Health Care Professional. 25th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2005., p. 140
Hepatocellular carcinoma has been associated rarely with long-term, high-dose anabolic steroid therapy. /Anabolic Steroids/
Thomson.Micromedex. Drug Information for the Health Care Professional. 25th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2005., p. 141
Hepatic neoplasms have been associated rarely with long-term, high-dose anabolic steroid therapy. /Anabolic Steroids/
Thomson.Micromedex. Drug Information for the Health Care Professional. 25th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2005., p. 141
FDA Pregnancy Category X. /CONTRAINDICATED IN PREGNANCY. Studies in animals and or humans, or investigational or post-marketing reports, have demonstrated positive evidence of fetal abnormalities or risk which clearly outweighs any possible benefit to the patient./
Thomson.Micromedex. Drug Information for the Health Care Professional. 25th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2005., p. 141
For more Drug Warnings (Complete) data for ETHYLESTRENOL (17 total), please visit the HSDB record page.
Anabolic Agents
These compounds stimulate anabolism and inhibit catabolism. They stimulate the development of muscle mass, strength, and power. (See all compounds classified as Anabolic Agents.)
A - Alimentary tract and metabolism
A14 - Anabolic agents for systemic use
A14A - Anabolic steroids
A14AB - Estren derivatives
A14AB02 - Ethylestrenol
The absorption, distribution, metabolism and excretion of [3H]ethylestrenol were studied in the rat. 2. Approximately one third of an intragastric dose was absorbed; 17% of the dose was excreted in urine and 83% in faeces within 10 days. 3. The dose is distributed throughout the rat, and kidney and liver were found to contain respectively 2.5-3 and 5-7 times the average specific activity of all other tissues. 4. Unchanged ethylestrenol was the only component detected in urine. Ethylestrenol was also found in faeces, along with two different dihydroxylated dihydro derivatives and one trihydroxylated dihydro derivative.
PMID:7233967 Stelle JW et al; Xenobiotica 11(2):103-15 (1981)
It is not known whether anabolic steroids are distributed into breast milk. /Anabolic Steroids/
Thomson.Micromedex. Drug Information for the Health Care Professional. 25th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2005., p. 141
Ethylestrenol incubated with a post-mitochondrial supernatant fraction of rat liver plus co-factors gives norethandrolone as the major metabolite. A second (minor) metabolite was tentatively identified as 17 alpha-ethyl-5 epsilon-estrane-3 epsilon,17 beta-diol. A pathway is suggested for the metabolism of ethylestrenol in the rat.
PMID:7233968 Steele JW et al; Xenobiotica. 1981 Feb;11(2):117-21.
Orabolin was oxidized at C-3 to form nilevar which in turn was metabolized in man by a ring reduction and side chain hydroxylation to form a 19-norpregnatriol. Identification of this metabolite, detection in urine is test for athletes suspected of drug misuse.
PMID:410996 Ward et al; J Steroid Biochem 8 (10): 1057-63 (1977)
Anabolic steroids reverses catabolic processes and negative nitrogen balance by promoting protein anabolism and stimulating appetite if there is concurrently a proper intake of calories and proteins. /Anabolic Steroids/
Thomson.Micromedex. Drug Information for the Health Care Professional. 25th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2005., p. 141
ABOUT THIS PAGE
79
PharmaCompass offers a list of Ethylestrenol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ethylestrenol manufacturer or Ethylestrenol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ethylestrenol manufacturer or Ethylestrenol supplier.
A Ethylestrenol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ethylestrenol, including repackagers and relabelers. The FDA regulates Ethylestrenol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ethylestrenol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ethylestrenol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Ethylestrenol supplier is an individual or a company that provides Ethylestrenol active pharmaceutical ingredient (API) or Ethylestrenol finished formulations upon request. The Ethylestrenol suppliers may include Ethylestrenol API manufacturers, exporters, distributors and traders.
click here to find a list of Ethylestrenol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Ethylestrenol DMF (Drug Master File) is a document detailing the whole manufacturing process of Ethylestrenol active pharmaceutical ingredient (API) in detail. Different forms of Ethylestrenol DMFs exist exist since differing nations have different regulations, such as Ethylestrenol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Ethylestrenol DMF submitted to regulatory agencies in the US is known as a USDMF. Ethylestrenol USDMF includes data on Ethylestrenol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Ethylestrenol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Ethylestrenol suppliers with USDMF on PharmaCompass.
Ethylestrenol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ethylestrenol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ethylestrenol GMP manufacturer or Ethylestrenol GMP API supplier for your needs.
A Ethylestrenol CoA (Certificate of Analysis) is a formal document that attests to Ethylestrenol's compliance with Ethylestrenol specifications and serves as a tool for batch-level quality control.
Ethylestrenol CoA mostly includes findings from lab analyses of a specific batch. For each Ethylestrenol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ethylestrenol may be tested according to a variety of international standards, such as European Pharmacopoeia (Ethylestrenol EP), Ethylestrenol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ethylestrenol USP).

