Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
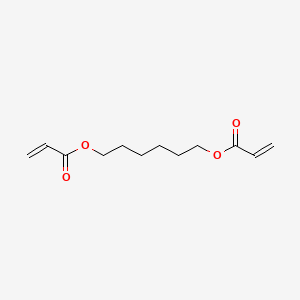

1. Hexamethylene Diacrylate
2. Hexanediol Diacrylate
1. 13048-33-4
2. Hexamethylene Diacrylate
3. Kayarad Hdda
4. 1,6-hexamethylene Diacrylate
5. Sartomer 238
6. Hdoda
7. Viscoat 230
8. Hexamethylene Acrylate
9. Photomer 4017
10. Hexamethylene Glycol Diacrylate
11. Setalux Uv 2243
12. Sartomer Sr 238
13. Hdda
14. Nk Ester A Hd
15. Acrylic Acid, Hexamethylene Ester
16. Hexaneglycol Diarcylate
17. 2-propenoic Acid, 1,6-hexanediyl Ester
18. Nk Ester A-hd
19. Sr 238
20. 1,6-hexanediol Di-2-propenoate
21. C 716
22. Fy751v5mmy
23. Dtxsid9025401
24. Laromer Hdda
25. Kayarad Ks-hdda
26. New Frontier Hdda
27. Actilane 425
28. Sartomer Sr-238
29. Photomer 4017f
30. 2-propenoic Acid, 1,1'-(1,6-hexanediyl) Ester
31. Hexamethyleneglycol Diacrylate
32. Genomer 1223
33. Hexaneglycol Diacrylate
34. Agisyn 2816
35. M-200 (acrylate)
36. Dtxcid705401
37. Em-221tf
38. Sr-238ns
39. 1,6-hexamethylenediol Diacrylate
40. Light Acrylate 1,6hx-a
41. Light Acrylate Sr-238f
42. 1,6hx-a
43. Sr-238f
44. 1,6-hexanediyl Diacrylate
45. Em-221
46. Sm-626
47. Sr-238
48. Sr-2389
49. M 200
50. V-230
51. Refchem:73627
52. 235-921-9
53. Hexanediol Diacrylate
54. Hexane-1,6-diyl Diacrylate
55. 6-prop-2-enoyloxyhexyl Prop-2-enoate
56. 1,6-bis(acryloyloxy)hexane
57. Mfcd00008631
58. Hexane-1,6-diyl Bisacrylate
59. Hexane-1,6-diyl Diacrylate (stabilized With Mehq)
60. Cas-13048-33-4
61. Hexanediol Diacrylate(9 Cp(25 Degrees C))
62. Ccris 4823
63. Hexamethylenediacrylate
64. Hsdb 6109
65. Einecs 235-921-9
66. Unii-fy751v5mmy
67. Brn 1870540
68. 1,6-hexanediol Diacrylate (stabilized With 100 Ppm Mehq) (>85%)
69. Ks-hdda
70. Nk Ester A-hd-n
71. 1,6-hexandiol Diacrylate
72. 1,6 Hexanediol Diacrylate
73. Hexamethylenediol Diacrylate
74. A-hd-n
75. Ec 235-921-9
76. C-6da
77. Schembl33725
78. 1,6-bis(acryloyloxy)hexane (stabilized With Mehq)
79. 1,6-hexanediol Diacrylate (stabilized With Mehq)
80. Light Ester 1.6hx-a
81. Chembl1530684
82. 6-(acryloyloxy)hexyl Acrylate #
83. 1,6-hexanediol Diacrylate, 90%
84. 1.6hx
85. Hexanediol Diacrylate (hdda)
86. Tox21_201349
87. Tox21_303168
88. Msk161342
89. Akos015904309
90. Hexamethylene Diacrylate, 1,6-
91. Ncgc00091257-01
92. Ncgc00091257-02
93. Ncgc00091257-03
94. Ncgc00091257-04
95. Ncgc00257183-01
96. Ncgc00258901-01
97. 1,6-hexanediol Diacrylate [hsdb]
98. As-75465
99. Db-254921
100. B2936
101. Cs-0158045
102. Ns00007428
103. 1,6-hexanediol Diacrylate, Technical Grade, 80%
104. F242822
105. 1,6-bis(acryloyloxy)hexane, (stabilized With Mehq)
106. Q1276266
107. 1,6-hexamethylene Diacrylate (1,6-hexanediol Diacrylate), Visc.5-8 Cps
| Molecular Weight | 226.27 g/mol |
|---|---|
| Molecular Formula | C12H18O4 |
| XLogP3 | 2.6 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 11 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 52.6 |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 219 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Acrylates and methacrylates are detoxified predominantly via conjugation with glutathione via the Michael addition reaction or glutathione-S-transferase. They are also likely to be hydrolyzed via carboxylesterases. The lower molecular weight esters are rapidly metabolized and eliminated, therefore, will not likely cause cumulative toxicity. /Acrylates/
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V6 600
...Dimethacrylates proved to be moderate to strong sensitizers in the guinea pig.
PMID:6839734 Van Der Walle HB et al; Contact Dermatitis 9(1): 10-20 (1983)
ABOUT THIS PAGE
35
PharmaCompass offers a list of EM-221 API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right EM-221 manufacturer or EM-221 supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred EM-221 manufacturer or EM-221 supplier.
A EM-221 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of EM-221, including repackagers and relabelers. The FDA regulates EM-221 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. EM-221 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A EM-221 supplier is an individual or a company that provides EM-221 active pharmaceutical ingredient (API) or EM-221 finished formulations upon request. The EM-221 suppliers may include EM-221 API manufacturers, exporters, distributors and traders.
EM-221 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of EM-221 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right EM-221 GMP manufacturer or EM-221 GMP API supplier for your needs.
A EM-221 CoA (Certificate of Analysis) is a formal document that attests to EM-221's compliance with EM-221 specifications and serves as a tool for batch-level quality control.
EM-221 CoA mostly includes findings from lab analyses of a specific batch. For each EM-221 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
EM-221 may be tested according to a variety of international standards, such as European Pharmacopoeia (EM-221 EP), EM-221 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (EM-221 USP).

