
Synopsis
Synopsis
0
KDMF
0
VMF
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
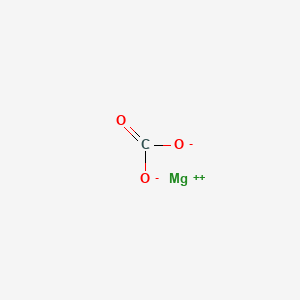

1. Anhydrous Magnesium Carbonate
2. C.i. 77713
3. Carbonic Acid, Magnesium Salt (1:1), Hydrate
4. Ci 77713
5. E-504
6. Magnesite
7. Magnesite (mg(co3))
8. Magnesium Carbonate (1:1) Hydrate
9. Magnesium Carbonate Anhydrous
10. Mgco3.3h2o
11. Nesquehonite
1. 546-93-0
2. Magnesite
3. Carbonic Acid, Magnesium Salt
4. 13717-00-5
5. Carbonic Acid, Magnesium Salt (1:1)
6. Magnesium Carbonate Anhydrous
7. Magnesite Dust
8. Magnesite (mg(co3))
9. Magnesium;carbonate
10. Hydromagnesite
11. Magmaster
12. Magnesium Carbonate (1:1)
13. 7757-69-9
14. Carbonate Magnesium
15. C.i. 77713
16. Stan-mag Magnesium Carbonate
17. Magnesium Carbonate,light
18. Magnesium Carbonate (mgco3)
19. Dci Light Magnesium Carbonate
20. Ins No.504(i)
21. Anhydrous Magnesium Carbonate
22. Ins-504(i)
23. Magnesium Carbonate Gold Star
24. Magnesium(ii) Carbonate (1:1)
25. Chebi:31793
26. E-504(i)
27. Mfcd00064632
28. Nsc-83511
29. 0ihc698356
30. Giobertite
31. Kimboshi
32. Apolda
33. Destab
34. Magfy
35. Magnesium Carbonate, Hydrated
36. Caswell No. 530
37. Gp 20 (carbonate)
38. Ma 70 (carbonate)
39. Gold Star (carbonate)
40. Magnesium Carbonate [usan]
41. Australian Magnesite
42. Hsdb 211
43. Einecs 208-915-9
44. Nsc 83511
45. Epa Pesticide Chemical Code 073503
46. Ci 77713
47. Ai3-00768
48. Magnesiumkarbonat
49. Unii-0ihc698356
50. Einecs 231-817-2
51. Carbonic Acid, Magnesium Salt (1:?)
52. Mgco3
53. Magnesium Carbonate Usp
54. Ec 208-915-9
55. Magnesium Carbonate, Usp Grade
56. Chembl1200736
57. Dtxsid4049660
58. Magnesium Carbonate Microparticles
59. Cs-b1764
60. Magnesium Carbonate [who-dd]
61. Akos015903527
62. Db09481
63. E504
64. Anhydrous Magnesium Carbonate [mart.]
65. Ft-0774766
66. Q407931
67. Cyclopentanecarboxylic Acid, 3-methyl-2-oxo-, Methyl Ester
68. 53678-75-4
| Molecular Weight | 84.31 g/mol |
|---|---|
| Molecular Formula | CMgO3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 83.9697855 g/mol |
| Monoisotopic Mass | 83.9697855 g/mol |
| Topological Polar Surface Area | 63.2 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 18.8 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
THE CARBONATE ... /SALT/ OF MAGNESIUM /IS/ USED AS /ANTACID/, USUALLY IN COMBINATION WITH ALUMINUM HYDROXIDE.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 909
ALTHOUGH 1 G CONTAINS APPROX 20 MEQ ONLY FRACTION MAY BE AVAILABLE FOR NEUTRALIZATION IN VIVO. USUAL ANTACID DOSE OF 500 MG TO 2 G MAY BE INADEQUATE
Gilman, A. G., L. S. Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 6th ed. New York: Macmillan Publishing Co., Inc. 1980., p. 993
MEDICATION (VET): WHEN INSOL IN DIGESTIVE TRACT IT IS ANTIDIARRHEAL & COATS MUCOSAE; WHEN SLIGHTLY SOL FORM IS USED IT CAN BE AS LAXATIVE AS MAGNESIUM OXIDE. ... COMMERCIALLY AVAILABLE COSMETIC GRADES EXIST FOR RARE USE AS TOPICAL PROTECTANT, MOISTURE & FAT ABSORBENT.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 319
AS AN ANTACID, IT IS RELATIVELY WEAK (1 G NEUTRALIZES APPROX 7 ML OF 0.1 N HCL IN 10 MIN & 17 ML IN 2 HR).
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 735
For more Therapeutic Uses (Complete) data for MAGNESIUM CARBONATE (8 total), please visit the HSDB record page.
3= MODERATELY TOXIC: PROBABLE ORAL LETHAL DOSE (HUMAN) 0.5-5 G/KG; BETWEEN 1 OZ & 1 PINT (OR 1 LB) FOR 70 KG PERSON (150 LB).
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-127
Used as an over the counter antacid.
FDA Label
Neutralizes acid in the stomach.
Bleaching Agents
Chemicals that are used to oxidize pigments and thus effect whitening. (See all compounds classified as Bleaching Agents.)
Hygroscopic Agents
Materials that readily absorb moisture from their surroundings. (See all compounds classified as Hygroscopic Agents.)
A - Alimentary tract and metabolism
A02 - Drugs for acid related disorders
A02A - Antacids
A02AA - Magnesium compounds
A02AA01 - Magnesium carbonate
A - Alimentary tract and metabolism
A06 - Drugs for constipation
A06A - Drugs for constipation
A06AD - Osmotically acting laxatives
A06AD01 - Magnesium carbonate
Absorption
About 40-60% of magnesium is absorbed following oral administration. Percent absorption decreases as dose increases.
Route of Elimination
Primarily eliminated in urine.
Volume of Distribution
Vd for magnesium is 0.2-0.4L/kg. About 50% distributes to bone.
Clearance
Maximum magnesium clearance is directly proportional to creatinine clearance.
IN SHEEP TRIAL REAGENT GRADE MATERIAL DEMONSTRATED 72% TRUE ABSORPTION VALUES, WHILE COMMERCIAL MAGNESITE HAD ONLY 14% VALUE DRAMATIZING NEED FOR MORE BIOLOGIC AVAILABILITY STUDIES ON MANY FEED INGREDIENTS.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 319
Magnesium does not appear to be metabolized in any way.
Half life of 27.7 hours reported with overdose of 400mEq of magnesium in an adult.
Magnesium carbonate reacts with hydrochloric acid in the stomach to form carbon dioxide and magnesium chloride thus neutralizing excess acid in the stomach.
... Rapidly reacts with hydrochloric acid to form ... carbon dioxide /and magnesium chloride/.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 2774

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Dosage Form : Tablet
Grade : Oral
Dosage Form : Cream / Lotion / Ointment
Grade : Topical, Parenteral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Tablet
Grade : Oral
Application : Controlled & Modified Release
Excipient Details : ACTILLETS are microcrystalline cellulose spheres developed using advanced drug delivery technology to enable effective loading & coating of particles.
Pharmacopoeia Ref : NA
Technical Specs : Not Available
Ingredient(s) : Microcrystalline Cellulose Excipients
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Taste Masking
Excipient Details : CM90 is a directly compressible, granulated calcium carbonate with maltodextrin used for swallow tablets due to its high density and compressibility.
Pharmacopoeia Ref : NA
Technical Specs : PSD D50: 490- 500 µm; Tapped Density: 1.50
Ingredient(s) : Calcium Carbonate Excipient
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Taste Masking
Excipient Details : CS90 is a directly compressible calcium carbonate with starch used for chewable tablets due to its smooth mouthfeel and creamy texture.
Pharmacopoeia Ref : NA
Technical Specs : PSD D50: 150-175 µm, Tapped Density: 0.85
Ingredient(s) : Calcium Carbonate Excipient
Dosage Form : Tablet
Grade : Oral
Application : Direct Compression
Excipient Details : DC SIM 100 is a directly compressible simethicone powder used for antacid and anti-gas tablets.
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Disintegrants & Superdisintegrants
Excipient Details : Mannogem 2080 exhibits excellent flow, disintegration and compression properties.
Dosage Form : Orodispersible Tablet
Grade : Oral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
38
PharmaCompass offers a list of Magnesium Carbonate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Magnesium Carbonate manufacturer or Magnesium Carbonate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Magnesium Carbonate manufacturer or Magnesium Carbonate supplier.
PharmaCompass also assists you with knowing the Magnesium Carbonate API Price utilized in the formulation of products. Magnesium Carbonate API Price is not always fixed or binding as the Magnesium Carbonate Price is obtained through a variety of data sources. The Magnesium Carbonate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A DTXSID4049660 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of DTXSID4049660, including repackagers and relabelers. The FDA regulates DTXSID4049660 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. DTXSID4049660 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of DTXSID4049660 manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A DTXSID4049660 supplier is an individual or a company that provides DTXSID4049660 active pharmaceutical ingredient (API) or DTXSID4049660 finished formulations upon request. The DTXSID4049660 suppliers may include DTXSID4049660 API manufacturers, exporters, distributors and traders.
click here to find a list of DTXSID4049660 suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A DTXSID4049660 DMF (Drug Master File) is a document detailing the whole manufacturing process of DTXSID4049660 active pharmaceutical ingredient (API) in detail. Different forms of DTXSID4049660 DMFs exist exist since differing nations have different regulations, such as DTXSID4049660 USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A DTXSID4049660 DMF submitted to regulatory agencies in the US is known as a USDMF. DTXSID4049660 USDMF includes data on DTXSID4049660's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The DTXSID4049660 USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of DTXSID4049660 suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The DTXSID4049660 Drug Master File in Japan (DTXSID4049660 JDMF) empowers DTXSID4049660 API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the DTXSID4049660 JDMF during the approval evaluation for pharmaceutical products. At the time of DTXSID4049660 JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of DTXSID4049660 suppliers with JDMF on PharmaCompass.
A DTXSID4049660 CEP of the European Pharmacopoeia monograph is often referred to as a DTXSID4049660 Certificate of Suitability (COS). The purpose of a DTXSID4049660 CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of DTXSID4049660 EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of DTXSID4049660 to their clients by showing that a DTXSID4049660 CEP has been issued for it. The manufacturer submits a DTXSID4049660 CEP (COS) as part of the market authorization procedure, and it takes on the role of a DTXSID4049660 CEP holder for the record. Additionally, the data presented in the DTXSID4049660 CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the DTXSID4049660 DMF.
A DTXSID4049660 CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. DTXSID4049660 CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of DTXSID4049660 suppliers with CEP (COS) on PharmaCompass.
A DTXSID4049660 written confirmation (DTXSID4049660 WC) is an official document issued by a regulatory agency to a DTXSID4049660 manufacturer, verifying that the manufacturing facility of a DTXSID4049660 active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting DTXSID4049660 APIs or DTXSID4049660 finished pharmaceutical products to another nation, regulatory agencies frequently require a DTXSID4049660 WC (written confirmation) as part of the regulatory process.
click here to find a list of DTXSID4049660 suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing DTXSID4049660 as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for DTXSID4049660 API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture DTXSID4049660 as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain DTXSID4049660 and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a DTXSID4049660 NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of DTXSID4049660 suppliers with NDC on PharmaCompass.
DTXSID4049660 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of DTXSID4049660 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right DTXSID4049660 GMP manufacturer or DTXSID4049660 GMP API supplier for your needs.
A DTXSID4049660 CoA (Certificate of Analysis) is a formal document that attests to DTXSID4049660's compliance with DTXSID4049660 specifications and serves as a tool for batch-level quality control.
DTXSID4049660 CoA mostly includes findings from lab analyses of a specific batch. For each DTXSID4049660 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
DTXSID4049660 may be tested according to a variety of international standards, such as European Pharmacopoeia (DTXSID4049660 EP), DTXSID4049660 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (DTXSID4049660 USP).

