
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
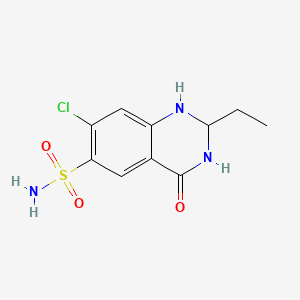

1. Quinethazone, (+)-isomer
2. Quinethazone, (+-)-isomer
3. Quinethazone, (-)-isomer
1. 73-49-4
2. Chinethazonum
3. Hydromox
4. Quinethazon
5. Quinethazonum
6. Aquamox
7. Idrokin
8. Chinetazone
9. Chinetazone [dcit]
10. Quinetazona [inn-spanish]
11. Quinethazonum [inn-latin]
12. Cl 36010
13. 7-chloro-2-ethyl-4-oxo-2,3-dihydro-1h-quinazoline-6-sulfonamide
14. 7-chloro-2-ethyl-4-oxo-1,2,3,4-tetrahydroquinazoline-6-sulfonamide
15. Quinethazone (1.5 G)
16. 7-chloro-2-ethyl-1,2,3,4-tetrahydro-4-oxo-6-quinazolinesulfonamide
17. 6-quinazolinesulfonamide, 7-chloro-2-ethyl-1,2,3,4-tetrahydro-4-oxo-
18. Nsc-759904
19. 7-chloro-2-ethyl-1,2,3,4-tetrahydro-4-oxo-6-sulfamoylquinazoline
20. 7-chloro-2-ethyl-6-sulfamoyl-1,2,3,4-tetrahydro-4-quinazolinone
21. Chebi:8717
22. 455e0s048w
23. Quinetazona
24. Dsstox_cid_3548
25. Dsstox_rid_77074
26. Dsstox_gsid_23548
27. Quinethazone (aquamox)
28. Chinethazone
29. 2-ethyl-7-chloro-1,2,3,4-tetrahydro-4-oxoquinazoline-6-sulfonamide
30. Hydromox (tn)
31. Ccris 6098
32. Hsdb 3392
33. Einecs 200-801-7
34. Quinethazone (jan/inn)
35. Brn 0818554
36. Unii-455e0s048w
37. Cas-73-49-4
38. 2-ethyl-7-chloro-1,2,3,4-tetrahydro-4-oxochinazolin-6-sulfonamid
39. Ncgc00016313-01
40. Quinethazone [usp:inn:ban:jan]
41. Starbld0009648
42. Prestwick0_001050
43. Prestwick1_001050
44. Prestwick2_001050
45. Prestwick3_001050
46. Quinethazone [mi]
47. Quinethazone [inn]
48. Quinethazone [jan]
49. Quinethazone [hsdb]
50. Chembl1532
51. Quinethazone [vandf]
52. Bspbio_000980
53. Quinethazone [mart.]
54. 5-25-09-00214 (beilstein Handbook Reference)
55. Mls002154126
56. Quinethazone [who-dd]
57. Schembl301310
58. Spbio_002910
59. Bpbio1_001078
60. Gtpl7289
61. Dtxsid9023548
62. Bdbm25898
63. Hms1571a22
64. Hms2098a22
65. Hms2234o04
66. Hms3370m01
67. Hms3715a22
68. Pharmakon1600-01503877
69. Quinethazone [orange Book]
70. Bcp07451
71. Hy-b1364
72. Tox21_110366
73. Ac8883
74. Mfcd00867329
75. Nsc759904
76. Zinc00000686
77. 6-quinazolinesulfonamide, 1,2,3,4-tetrahydro-7-chloro-2-ethyl-4-oxo-
78. Tox21_110366_1
79. Ccg-213232
80. Db01325
81. Nsc 759904
82. Hydromox R Component Quinethazone
83. Ncgc00179351-03
84. Smr001233433
85. Sy250224
86. Quinethazone Component Of Hydromox R
87. Ab00514022
88. Cs-0013105
89. Ft-0674271
90. C07342
91. D00461
92. Ab00514022_06
93. Sr-01000841190
94. Q7272222
95. Sr-01000841190-2
96. Brd-a59303141-001-03-9
97. 2-ethyl-7-chloro-2,3-dihydro-4(1h)-quinazolone-6-sulfonamide
98. 7-chloro-2-ethyl-4-hydroxy-1,2-dihydroquinazoline-6-sulfonamide
99. 7-chloro-2-ethyl-1,2,3,4-tetrahydro-4-oxo-6-quinzaolinesulfonamide
| Molecular Weight | 289.74 g/mol |
|---|---|
| Molecular Formula | C10H12ClN3O3S |
| XLogP3 | 1.2 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 289.0287901 g/mol |
| Monoisotopic Mass | 289.0287901 g/mol |
| Topological Polar Surface Area | 110 Ų |
| Heavy Atom Count | 18 |
| Formal Charge | 0 |
| Complexity | 436 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Diuretics, Sulfamyl
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
A QUINAZOLINE DERIVATIVE WITH DIURETIC & ANTIHYPERTENSIVE ACTIONS SIMILAR TO THIAZIDES. ... AVAILABLE CLINICAL EVIDENCE INDICATES THAT ITS SITE, MECHANISM & DURATION OF ACTION, ELECTROLYTE EXCRETION PATTERN, THERAPEUTIC ACTIONS, & UNTOWARD EFFECTS ARE SIMILIAR TO THOSE OF CHLOROTHIAZIDE & RELATED AGENTS.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 870
MOST OF THE THIAZIDES ARE GIVEN IN DIVIDED DAILY DOSES FOR TREATMENT OF HYPERTENSION, BUT A SINGLE DAILY DOSE MAY BE PREFERABLE FOR THE MOBILIZATION OF EDEMA FLUID. ... THE ACTION OF QUINETHAZONE...MAY PERSIST UP TO 24 HR.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 832
Initial antihypertensive agents Diuretics...hydrochlorothiazide or chlorthalidone is generally preferred; used in most clinical trials /from table/
Ellenhorn, M.J., S. Schonwald, G. Ordog, J. Wasserberger. Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore, MD: Williams and Wilkins, 1997., p. 552
For more Therapeutic Uses (Complete) data for QUINETHAZONE (8 total), please visit the HSDB record page.
PLASMA POTASSIUM CONCN SHOULD BE DETERMINED PERIODICALLY IN PT WHO RECEIVE THIAZIDE DIURETICS FOR EXTENDED PERIODS. /BENZOTHIADIAZIDES/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 832
... Long-term effects include slight decrease in extracellular fluid volume ... /from table/
Ellenhorn, M.J., S. Schonwald, G. Ordog, J. Wasserberger. Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore, MD: Williams and Wilkins, 1997., p. 552
May suppress lactation ... /Thiazide diuretics; from table/
Young, L.Y., M.A. Koda-Kimble (eds.). Applied Therapeutics. The Clinical Use of Drugs. 6th ed. Vancouver, WA., Applied Therapeutics, Inc. 1995., p. 45-29
Many experts consider diuretics contraindicated in pregnancy except for patients with heart disease, since they do not prevent or alter course of toxemia and may decrease placental perfusion. /Chlorothiazide; from table/
Young, L.Y., M.A. Koda-Kimble (eds.). Applied Therapeutics. The Clinical Use of Drugs. 6th ed. Vancouver, WA., Applied Therapeutics, Inc. 1995., p. 45-9
For more Drug Warnings (Complete) data for QUINETHAZONE (9 total), please visit the HSDB record page.
3. 3= MODERATELY TOXIC: PROBABLE ORAL LETHAL DOSE (HUMAN) 0.5-5 G/KG, BETWEEN 1 OUNCE & 1 PINT (OR 1 LB) FOR 70 KG PERSON (150 LB). /BENZOTHIADIAZIDE DIURETICS/
Gosselin, R.E., H.C. Hodge, R.P. Smith, and M.N. Gleason. Clinical Toxicology of Commercial Products. 4th ed. Baltimore: Williams and Wilkins, 1976., p. II-239
Used to treat hypertension.
Quinethazone is a thiazide diuretic used to treat hypertension. It inhibits Na+/Cl- reabsorption from the distal convoluted tubules in the kidneys. Thiazides also cause loss of potassium and an increase in serum uric acid. Thiazides are often used to treat hypertension, but their hypotensive effects are not necessarily due to their diuretic activity. Thiazides have been shown to prevent hypertension-related morbidity and mortality although the mechanism is not fully understood. Thiazides cause vasodilation by activating calcium-activated potassium channels (large conductance) in vascular smooth muscles and inhibiting various carbonic anhydrases in vascular tissue.
Sodium Chloride Symporter Inhibitors
Agents that inhibit SODIUM CHLORIDE SYMPORTERS. They act as DIURETICS. Excess use is associated with HYPOKALEMIA. (See all compounds classified as Sodium Chloride Symporter Inhibitors.)
C - Cardiovascular system
C03 - Diuretics
C03B - Low-ceiling diuretics, excl. thiazides
C03BA - Sulfonamides, plain
C03BA02 - Quinethazone
...RAPIDLY ABSORBED FROM GI TRACT...
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 81
As a diuretic, quinethazone inhibits active chloride reabsorption at the early distal tubule via the Na-Cl cotransporter, resulting in an increase in the excretion of sodium, chloride, and water. Thiazides like quinethazone also inhibit sodium ion transport across the renal tubular epithelium through binding to the thiazide sensitive sodium-chloride transporter. This results in an increase in potassium excretion via the sodium-potassium exchange mechanism. The antihypertensive mechanism of quinethazone is less well understood although it may be mediated through its action on carbonic anhydrases in the smooth muscle or through its action on the large-conductance calcium-activated potassium (KCa) channel, also found in the smooth muscle.
...BENZOTHIADIAZIDES HAVE DIRECT EFFECT ON RENAL TUBULAR TRANSPORT OF SODIUM & CHLORIDE INDEPENDENT OF CARBONIC ANHYDRASE INACTIVATION. ...HAVE PARALLEL DOSE-RESPONSE CURVES & COMPARABLE MAX CHLORURETIC EFFECTS. /BENZOTHIADIAZIDES/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 828
RENAL ACTIONS OF THIAZIDE DIURETICS DECR EXTRACELLULAR FLUID & PLASMA VOL, CARDIAC OUTPUT, & TOTAL EXCHANGEABLE SODIUM IN INDIVIDUALS WITHOUT ANY EVIDENCE OF CARDIAC FAILURE. ...SODIUM & WATER DEPLETION...BASIS FOR ANTIHYPERTENSIVE EFFECT. ...DIURETIC THIAZIDES RELAX PERIPHERAL ARTERIOLAR SMOOTH MUSCLE. /BENZOTHIADIAZIDES/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 712
Decreased plasma volume and decreased extracellular fluid volume; decreased cardiac output initially, followed by decreased total peripheral resistance with normalization of cardiac output ... /from table/
Ellenhorn, M.J., S. Schonwald, G. Ordog, J. Wasserberger. Ellenhorn's Medical Toxicology: Diagnosis and Treatment of Human Poisoning. 2nd ed. Baltimore, MD: Williams and Wilkins, 1997., p. 552
Thiazide diuretics increase urinary excretion of sodium and water by inhibiting sodium reabsorption in the early distal tubules. They increase the rate of delivery of tubular fluid and electrolytes to the distal sites of hydrogen and potassium ion secretion, while plasma volume contraction increases aldosterone production. The increased delivery and increase in aldosterone levels promote sodium reabsorption at the distal tubules, thus increasing the loss of potassium and hydrogen ions. /Thiazide diuretics/
USP. Convention. USPDI - Drug Information for the Health Care Professional. 19th ed. Volume I.Micromedex, Inc. Englewood, CO., 1999. Content Prepared by the U.S. Pharmacopieal Convention, Inc., p. 1274
For more Mechanism of Action (Complete) data for QUINETHAZONE (7 total), please visit the HSDB record page.
ABOUT THIS PAGE
81
PharmaCompass offers a list of Quinethazone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Quinethazone manufacturer or Quinethazone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Quinethazone manufacturer or Quinethazone supplier.
PharmaCompass also assists you with knowing the Quinethazone API Price utilized in the formulation of products. Quinethazone API Price is not always fixed or binding as the Quinethazone Price is obtained through a variety of data sources. The Quinethazone Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A DSSTox_CID_3548 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of DSSTox_CID_3548, including repackagers and relabelers. The FDA regulates DSSTox_CID_3548 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. DSSTox_CID_3548 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A DSSTox_CID_3548 supplier is an individual or a company that provides DSSTox_CID_3548 active pharmaceutical ingredient (API) or DSSTox_CID_3548 finished formulations upon request. The DSSTox_CID_3548 suppliers may include DSSTox_CID_3548 API manufacturers, exporters, distributors and traders.
DSSTox_CID_3548 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of DSSTox_CID_3548 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right DSSTox_CID_3548 GMP manufacturer or DSSTox_CID_3548 GMP API supplier for your needs.
A DSSTox_CID_3548 CoA (Certificate of Analysis) is a formal document that attests to DSSTox_CID_3548's compliance with DSSTox_CID_3548 specifications and serves as a tool for batch-level quality control.
DSSTox_CID_3548 CoA mostly includes findings from lab analyses of a specific batch. For each DSSTox_CID_3548 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
DSSTox_CID_3548 may be tested according to a variety of international standards, such as European Pharmacopoeia (DSSTox_CID_3548 EP), DSSTox_CID_3548 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (DSSTox_CID_3548 USP).

