
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
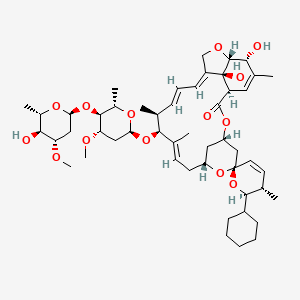

1. 25-cyclohexyl-5-o-demethyl-25-de(1-methylpropyl)avermectin A1a
2. Avermectin A1a, 25-cyclohexyl-5-o-demethyl-25-de(1-methylpropyl)-
3. Dectomax
4. L-701023
5. L701023
6. Uk-67,994
7. Uk-67994
1. 117704-25-3
2. Dectomax
3. Uk 67994
4. Uk-67,994
5. Kgd7a54h5p
6. Uk-67994
7. Avermectin A1a, 25-cyclohexyl-5-o-demethyl-25-de(1-methylpropyl)-
8. L-701023
9. Nsc-760342
10. L 701023
11. Doramectina
12. Doramectine
13. Doramectinum
14. Doramectine [inn-french]
15. Doramectinum [inn-latin]
16. Unii-kgd7a54h5p
17. Doramectina [inn-spanish]
18. Doramectin [usan:inn:ban]
19. Hsdb 7452
20. Ncgc00185761-01
21. Cyclohexylavermectin B1
22. Doramectin [mi]
23. Uk 67,994
24. Doramectin [inn]
25. Doramectin [jan]
26. Doramectin [hsdb]
27. Doramectin [usan]
28. Ec 601-490-4
29. Doramectin [mart.]
30. Dsstox_cid_28908
31. Dsstox_rid_83176
32. Dsstox_gsid_48982
33. Schembl118775
34. Doramectin [green Book]
35. Chembl2361641
36. Cyclohexylavermectin B1;dectomax
37. Dtxsid9048982
38. Ex-a3582
39. Tox21_113442
40. Mfcd00894747
41. S5117
42. Akos005146234
43. Doramectin 100 Microg/ml In Methanol
44. Zinc238809351
45. Zinc245253793
46. Ccg-270566
47. Cs-0736
48. Db11400
49. Nsc 760342
50. Ac-27772
51. As-17466
52. Doramectin 100 Microg/ml In Acetonitrile
53. Hy-17035
54. Cas-117704-25-3
55. 704d253
56. Doramectin, Antibiotic For Culture Media Use Only
57. Q-201041
58. Avermectin A(sub 1a), 25-cyclohexyl-5-o-demethyl-25-de(1-methylpropyl)-
59. (1'r,2s,4's,5s,6r,8'r,10'e,12'r,13's,14'e,20'r,21'r,24's)-6-cyclohexyl-21',24'-dihydroxy-12'-(((2r,4s,5s,6s)-5-(((2s,4s,5s,6s)-5-hydroxy-4-methoxy-6-methyloxan-2-yl)oxy)-4-methoxy-6-methyloxan-2-yl)oxy)-5,11',13',22'-tetramethyl-5,6-dihydro-3',7',19'-trioxaspiro(pyran-2,6'-tetracyclo(15.6.1.14,8.020,24)pentacosane)-10',14',16',22'-tetraen-2'-one
| Molecular Weight | 899.1 g/mol |
|---|---|
| Molecular Formula | C50H74O14 |
| XLogP3 | 4.5 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 7 |
| Exact Mass | 898.50785703 g/mol |
| Monoisotopic Mass | 898.50785703 g/mol |
| Topological Polar Surface Area | 170 Ų |
| Heavy Atom Count | 64 |
| Formal Charge | 0 |
| Complexity | 1790 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 19 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 3 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
MEDICATION (VET): Antiparasitic
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. 13th Edition, Whitehouse Station, NJ: Merck and Co., Inc., 2001., p. 603
VET: Doramectin is an ecto- and endoparasiticide for use in cattle and pigs. It is a semisynthetic member of the avermectin class, structurally similar to abamectin and ivermectin.
WHO; Evaluation of certain veterinary drug residues in food p.15 (2004) WHO Technical Report Series 925
VET: Doramectin (NADA 141-095) is approved for topical use to treat and control various worms (roundworms, lungworms, and eyeworms), grubs, lice, horn flies, and mange mites. It is also approved to control infections and to protect from reinfection with Cooperia oncophora and Dictyocaulus viviparus for 21 days, Ostertagia ostertagi, C. punctata, and Oesophagostomum radiatum for 28 days, and Haemonchus placei for 35 days after treatment.
US FDA, Center for Veterinary Medicine; CVM Update: Reminder - Doramectin not permitted for use in dairy cattle, June 21, 2004. Available from, as of July 19, 2006: https://www.fda.gov/cvm/CVM_Updates/milkresup.htm
VET: Objective: To determine effectiveness of a pour-on formulation of doramectin against Damalinia bovis, Haematopinus eurysternus, Linognathus vituli, Solenopotes capillatus, Chorioptes bovis, Sarcoptes scabiei, Hypoderma bovis, and Hypoderma lineatusm. Animals: Cattle of various ages with naturally acquired or artificial infestations with 1 or more species of lice, mites, or grubs. Procedure: In 10 louse and 6 mite studies, cattle were treated with doramectin (500 mug/kg, topicaly) on day 0, and p 28 days after treatment. Burdens of C. bovis and S. scabiei decreased to 0 in naturally infested cattle and approximately 0 in artificially infested cattle by day 14 to 15. In grub studies, 107 of 136 control cattle had warbles, whereas 2 of 136 doramectin-treated cattle had 1 warble each, which represented a cure rate of 98.5%.
PMID:10211679 Rooney KA et al; American Journal of Veterinary Research 60 (4): 402-4 (1999)
For more Therapeutic Uses (Complete) data for DORAMECTIN (6 total), please visit the HSDB record page.
Anthelmintics
Agents that kill parasitic worms. They are used therapeutically in the treatment of HELMINTHIASIS in man and animal. (See all compounds classified as Anthelmintics.)
Anti-Anxiety Agents
Agents that alleviate ANXIETY, tension, and ANXIETY DISORDERS, promote sedation, and have a calming effect without affecting clarity of consciousness or neurologic conditions. ADRENERGIC BETA-ANTAGONISTS are commonly used in the symptomatic treatment of anxiety but are not included here. (See all compounds classified as Anti-Anxiety Agents.)
Anticonvulsants
Drugs used to prevent SEIZURES or reduce their severity. (See all compounds classified as Anticonvulsants.)
Insecticides
Pesticides designed to control insects that are harmful to man. The insects may be directly harmful, as those acting as disease vectors, or indirectly harmful, as destroyers of crops, food products, or textile fabrics. (See all compounds classified as Insecticides.)
In the first study, 10 dairy Holstein cows were treated with a pour-on formulation of doramectin at a dose of 0.58 mg/kg bw and were retreated with the same dose 56 days later. ... Samples of milk were collected for 49 days and 10 days, respectively, after the first and second treatments. Samples were collected twice daily until day 7, and once daily on days 10, 13, 16, 19, 22, 25, 28, 32, 36, 40 and 49. On retreatment, samples were taken twice daily until day 7 and once at day 10. ... The concentrations of doramectin residue in milk increased to a maximum mean value of 22 mg/kg at 72 hr after treatment. Mean concentrations of doramectin residues decreased to below the limit of quantitation (3 mg/kg) at 384 hr (16 days). After retreatment, concentrations of doramectin residues increased gradually to a maximum mean value of 12 mg/kg at 48 hr after dosing; and decreased to <4 mg/kg at 240 hr (10 days) after dosing. The milk/fat analyses were conducted 1, 4, and 10 days after dosing. Mean concentrations of doramectin residues in the milk fat at these time points were 171 mg/kg, 501 mg/kg and 114 mg/kg, respectively. Concentration factors for doramectin residues in milk fat were 29.6, 32.2 and 24.7, respectively.
WHO; Evaluation of certain veterinary drug residues in food p.15 (2004) WHO Technical Report Series 925
In the second study, 10 cows were treated with doramectin by topical application of a pour-on formulation at a dose of 0.58 mg/kg and were re-treated with the same dose 56 days later. Samples of milk were collected twice daily. Concentrations of doramectin in milk increased to a maximum mean value of 9 mg/kg at 45 hr after treatment and decreased to below the LOQ by 237 hr (10 days) after treatment. After re-treatment on day 56, concentrations of residues increased to a mean maximum value of 8 mg/kg after 93 hr and decreased to less than the LOQ after 237 hr (10 days). Mean concentrations of doramectin residues in the milk fat at 1, 4, and 10 days were 91 mg/kg, 142 mg/kg and 55 mg/kg, respectively. Concentration factors for doramectin residues in milk fat versus milk were 14.2, 20.9 and 14.1, respectively.
WHO; Evaluation of certain veterinary drug residues in food p.16 (2004) WHO Technical Report Series 925
The third study determined the residue depletion profile of doramectin following the subcutaneous administration of doramectin formulation at 0.23 mg/kg bw in lactating cattle, followed by retreatment at the same dose 56 days later. ... Doramectin concentrations in milk increased gradually to a maximum mean value of 45 mg/kg at 67 hr. Subsequently, doramectin residues gradually declined, with mean residues below LOQ at 523 hr (22 days). After re-treatment, doramectin residues increased to a maximum mean value of 53 mg/kg at 56 hr. Residue concentrations then decreased to a mean value of 25 mg/kg at 237 hr (10 days) after re-treatment. Residues resulting from treatment by injection were consistently higher at any given timepoint than were those resulting from treatment with the pour-on formulation. Milk fat analyses were conducted using samples collected at the morning milking on days 1, day 4 and day 10 after treatment. Mean concentrations of doramectin residues in milk fat at these time-points were 557 mg/kg, 1036 mg/kg and 354 mg/kg, respectively. Milk fat concentration factors were 24, 24.2 and 23.4, respectively.
WHO; Evaluation of certain veterinary drug residues in food p.16 (2004) WHO Technical Report Series 925
Self-licking behavior in cattle has recently been identified as a determinant of the kinetic disposition of topically-administered ivermectin. /The present study documents/ the occurrence and extent of transfer between cattle of three topically-administered endectocides, as a consequence of allo-licking. Four groups of two Holstein cows each received one pour-on formulation of doramectin, ivermectin, or moxidectin, or no treatment. The cows were then kept together in a paddock. Systemic exposure to each topically-administered endectocide was observed in at least five of six non-treated cattle. Plasma and fecal drug concentration profiles in non-treated animals were highly variable between animals and within an animal, and sometimes attained those observed in treated animals. Drug exchanges were quantified by measuring plasma and fecal clearances after simultaneous i.v. administration of the three drugs as a cocktail. Plasma clearances were 185 + or - 43, 347 + or - 77 and 636 + or - 130 ml/kg/day, fecal clearances representing 75 + or - 26, 28 + or - 13, and 39 + or - 30% of the plasma clearance for doramectin, ivermectin and moxidectin, respectively. The amount of drug ingested by non-treated cattle attained 1.3-21.3% (doramectin), 1.3-16.1% (ivermectin), 2.4-10.6% (moxidectin) of a pour-on dose (500 ug/kg). The total amount of drug ingested by all non-treated cattle represented 29% (doramectin), 19% (ivermectin), and 8.6% (moxidectin) of the total amount of each drug poured on the backs of treated animals. The cumulative amounts of endectocide ingested by each non-treated cow ranged from 1.3 to 27.4% of a pour-on dose. Oral bioavailability after drug ingestion due to allo-licking was 13.5 + or - 9.4, 17.5 + or - 3.5 and 26.1+ or - 11.1% for doramectin, ivermectin and moxidectin, respectively. The extent of drug exchange demonstrated here raises concerns for drug efficacy and safety, emergence of drug resistance, presence of unexpectedly high residue levels in treated and/or untreated animals and high environmental burdens.
PMID:15491592 Bousquet-Melou A et al; Int J Parasitol 34 (11): 1299-307 (2004)
For more Absorption, Distribution and Excretion (Complete) data for DORAMECTIN (7 total), please visit the HSDB record page.
Doramectin labelled with tritium in the 5-position was administered as a single dose to Sprague-Dawley rats (2 males given 5 mg/kg bw in propylene glycol:glycerol by gavage), a beagle dog (1 female given 3.5 mg/kg bw in sesame oil by gavage) and cattle (5 males given 0.2 mg/kg bw subcutaneously). /The following metabolites were identified in/... the liver and feces from each species and the fat of cattle... /unchanged doramectin, 3"-O-desmethyl doramectin, 24-hydroxymethyl doramectin, and 24-hydroxymethyl-3"-O-desmethyl doramectin./
Joint FAO/WHO Expert Committee on Food Additives; WHO Food Additives Ser 36: Doramectin. Available from: //www.inchem.org/documents/jecfa/jecmono/v36je02.htm as of June 14, 2006.
The products of doramectin metabolism were similar in all species investigated /rats, dogs, pigs, cattle/. The metabolites were more polar than doramectin and were the result of O-demethylation in the distal saccharide ring, of hydroxylation of the 24-methyl group and a combination of both of these biotransformations.
Heitzman RJ; Doramectin. Addendum to the doramectin residue monograph prepared by the 45th meeting of the Committee and published in FAO Food and Nutrition Paper 41/8, Rome 1996 p. 29. Available from ftp://ftp.fao.org/ag/agn/jecfa/vetdrug/41-12-doramectin.pdf as of July 19, 2006
The plasma kinetics of doramectin were determined in eight pigs (4 male castrates and 4 females, each weighing approximately 40 kg) dosed im with (3H)doramectin at 0.3 mg/kg bw using a prototype commercial formulation (75% sesame oil/25% ethyl oleate). ... The apparent terminal half-lives of elimination from plasma of total (3H)labelled materials and unchanged doramectin were 7.7 and 6.4 days, respectively.
Heitzman RJ; Doramectin. Addendum to the doramectin residue monograph prepared by the 45th meeting of the Committee and published in FAO Food and Nutrition Paper 41/8, Rome 1996 p. 28. Available from ftp://ftp.fao.org/ag/agn/jecfa/vetdrug/41-12-doramectin.pdf as of July 19, 2006
Avermectins induce rapid, non-spastic paralysis in nematodes and arthropods. One common feature of avermectins appears to be the modulation of trans-membrane chloride ion (Cl-) channel activity in nematode nerve cells, and in both nerve and muscle cells of arthropods. These Cl- channels may be gated by a variety of neurotransmitter receptors including gamma-aminobutyric acid (GABA), glutamate and acetylcholine. Activation of the Cl- channels by avermectins leads to an increase in Cl- conductance which results in a changed membrane potential and this causes inhibition of electrical activity in the target nerve or muscle cell. GABA is also a major inhibitory neurotransmitter in the mammalian CNS and avermectins do have intrinsic activity on the mammalian GABA receptor/Cl- channel complex. Avermectins have been reported to bind to glycine receptor/Cl- channel complexes which are restricted to the CNS in mammals. Penetration of the blood brain barrier by avermectins is extremely poor and this may account for the wide margin of safety exhibited by these compounds following administration to mammals. /Avermectins/
Joint FAO/WHO Expert Committee on Food Additives; WHO Food Additives Ser 36: Doramectin. Available from: //www.inchem.org/documents/jecfa/jecmono/v36je02.htm as of June 14, 2006.
Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
79
PharmaCompass offers a list of Doramectin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Doramectin manufacturer or Doramectin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Doramectin manufacturer or Doramectin supplier.
A Doramectin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Doramectin, including repackagers and relabelers. The FDA regulates Doramectin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Doramectin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Doramectin manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Doramectin supplier is an individual or a company that provides Doramectin active pharmaceutical ingredient (API) or Doramectin finished formulations upon request. The Doramectin suppliers may include Doramectin API manufacturers, exporters, distributors and traders.
click here to find a list of Doramectin suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Doramectin as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Doramectin API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Doramectin as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Doramectin and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Doramectin NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Doramectin suppliers with NDC on PharmaCompass.
Doramectin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Doramectin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Doramectin GMP manufacturer or Doramectin GMP API supplier for your needs.
A Doramectin CoA (Certificate of Analysis) is a formal document that attests to Doramectin's compliance with Doramectin specifications and serves as a tool for batch-level quality control.
Doramectin CoA mostly includes findings from lab analyses of a specific batch. For each Doramectin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Doramectin may be tested according to a variety of international standards, such as European Pharmacopoeia (Doramectin EP), Doramectin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Doramectin USP).

