
Synopsis
Synopsis
0
JDMF
0
EU WC
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Barosmin
2. Buchu Resin
3. Daflon
4. Resin, Buchu
5. Venosmine
1. 520-27-4
2. Barosmin
3. Diosimin
4. Venosmine
5. Diosmil
6. Diosmine
7. Flebosten
8. Daflon
9. Tovene
10. Ven-detrex
11. Diovenor
12. Buchu Resin
13. Litosmil
14. Veno-v
15. Flebosmil
16. Diosminum
17. Rioven
18. Diosmetin 7-o-rutinoside
19. Diosven
20. Flebaven
21. Flebavena
22. Hemerven
23. Insuven
24. Varinon
25. Dioven
26. Diosmetin-7-o-rutinoside
27. Diosmin [inn]
28. Diosmetin 7-rutinoside
29. 5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-7-[(2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-[[(2r,3r,4r,5r,6s)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxymethyl]oxan-2-yl]oxychromen-4-one
30. 7qm776wj5n
31. Chebi:4631
32. 3',5,7-trihydroxy-4'-methoxyflavone 7-rutinoside
33. 3',5,7-trihydroxy-4'-methoxyflavone-7-rutinoside
34. Diosmin (inn)
35. Nsc-758417
36. 5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-7-(((2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-((((2r,3r,4r,5r,6s)-3,4,5-trihydroxy-6-methyltetrahydro-2h-pyran-2-yl)oxy)methyl)tetrahydro-2h-pyran-2-yl)oxy)-4h-chromen-4-one
37. Diosmin, Analytical Standard
38. Dsstox_cid_25892
39. Dsstox_rid_81206
40. Dsstox_gsid_45892
41. 3',5,7-trihydroxy-4'-methoxyflavone-7-(6-o-(-deoxy-alpha-l-mannopyraonsyl)-beta-d-glucopyranoside
42. 5-hydroxy-2-(3-hydroxy-4-methoxy-phenyl)-7-[(2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-[[(2r,3r,4r,5r,6s)-3,4,5-trihydroxy-6-methyl-tetrahydropyran-2-yl]oxymethyl]tetrahydropyran-2-yl]oxy-chromen-4-one
43. 3',5,7-trihydroxy-4'-methoxyflavone 7-rhamnoglucoside
44. Cas-520-27-4
45. Diosmin [inn:ban]
46. Diosmin [inn-spanish]
47. Diosmine [inn-french]
48. Diosminum [inn-latin]
49. Sr-01000799147
50. Unii-7qm776wj5n
51. Ccris 7915
52. Ncgc00095022-01
53. 4h-1-benzopyran-4-one, 7-((6-o-(6-deoxy-.alpha.-l-mannopyranosyl)-.beta.-d-glucopyranosyl)oxy)-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-
54. 4h-1-benzopyran-4-one, 7-[[6-o-(6-deoxy-.alpha.-l-mannopyranosyl)-.beta.-d-glucopyranosyl]oxy]-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-
55. Einecs 208-289-7
56. Daflon (tn)
57. Mfcd00009772
58. Diosmin (8)
59. Se 4601
60. Diosmine [inci]
61. Diosmin [dsc]
62. Diosmin [mi]
63. Diosmin [mart.]
64. Diosmin [usp-rs]
65. Diosmin [who-dd]
66. Mls001304032
67. Schembl120870
68. Diosmin [ep Monograph]
69. Chembl231884
70. Dtxsid4045892
71. 3',5-dihydroxy-4'-methoxy-4-oxo-4h-chromen-7-ylrutosid
72. Bdbm153267
73. Hms2233p16
74. Hms3713l08
75. Act05288
76. Zinc4098512
77. Tox21_111392
78. 3',5-dihydroxy-4'-methoxy-4-oxo-4h-chromen-7-ylrutosid [iupac]
79. S2292
80. Akos015969767
81. Tox21_111392_1
82. Bcp9000612
83. Ccg-208570
84. Db08995
85. Nsc 758417
86. Smp1_000183
87. Ncgc00344564-01
88. 4h-1-benzopyran-4-one, 7-((6-o-(6-deoxy-alpha-l-mannopyranosyl)-beta-d-glucopyranosyl)oxy)-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-
89. 4h-1-benzopyran-4-one,7-[[6-o-(6-deoxy-a-l-mannopyranosyl)-b-d-glucopyranosyl]oxy]-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-
90. 5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-4-oxo-4h-1-benzopyran-7-yl 6-o-(6-deoxy-alpha-l-mannopyranosyl)-beta-d-glucopyranoside
91. 5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-7-((6-o-alpha-l-rhamnopyranosyl-beta-d-glycopyranosyl)oxy)-4-chromenon
92. As-13224
93. Bp-12422
94. Smr000718616
95. Bcp0726000067
96. C10039
97. D07858
98. 520d274
99. 3',5,7-trihydroxy-4'-methoxy Flavone-7-rutinoside
100. Q2607865
101. Sr-01000799147-4
102. Sr-01000799147-5
103. Sr-01000799147-6
104. Diosmin, European Pharmacopoeia (ep) Reference Standard
105. Diosmin, United States Pharmacopeia (usp) Reference Standard
106. Diosmin For System Suitability, European Pharmacopoeia (ep) Reference Standard
107. 5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-7-((2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(((2r,3r,4r,5r,6s)-3,4,5-trihydroxy-6-methyltetrahydro-2h-pyran-2-yloxy)methyl)tetrahydro-2h-pyran-2-yloxy)-4h-chromen-4-one
108. 5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-7-(o6-alpha-l-rhamnopyranosyl-beta-d-glucopyranosyloxy)chromen-4-one
109. 5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-7-[(2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-[[(2s,3s,4s,5s,6r)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxymethyl]oxan-2-yl]oxychromen-4-one
110. 7-((6-o-(6-deoxy-ga-l-mannopyranosyl)-.beta.-d-glucopyranosyl)oxy)-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-4h-1-benzopyran-4-one
111. 7-((6-o-(6-deoxy-ga-l-mannopyranosyl)-beta-d-glucopyranosyl)oxy)-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-4h-1-benzopyran-4-one
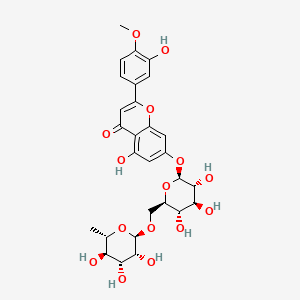
| Molecular Weight | 608.5 g/mol |
|---|---|
| Molecular Formula | C28H32O15 |
| XLogP3 | -0.8 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 7 |
| Exact Mass | 608.17412031 g/mol |
| Monoisotopic Mass | 608.17412031 g/mol |
| Topological Polar Surface Area | 234 Ų |
| Heavy Atom Count | 43 |
| Formal Charge | 0 |
| Complexity | 995 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 10 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Diosmin is used over-the-counter alone or with ingredients such as [hesperidin] and [diosmetin] to support vein and capillary function.
Diosmin is a venoactive drug supporting circulatory health through various actions on blood vessels; it supports lymphatic drainage and improves microcirculation while increasing venous tone and elasticity. For these reasons, diosmin is frequently taken by individuals with chronic venous disease to support vascular health and has been demonstrated to improve quality of life. In addition to the above effects, diosmin exerts antioxidant activity and scavenges oxygen free radicals, reducing levels of oxidative stress normally detected through biomarkers such as prostaglandins isoprostane precursors. In one clinical study, mean content of TNF alpha, VEGF-C, VEGF-A IL-6, in addition to FGF2 were decreased by after the therapy with diosmin; findings were statistically significant. Additionally, a decrease in edema and mean leg circumference of patients taking diosmin for three months was observed in a clinical study. Diosmin has been demonstrated to enhance the metabolism of glucose in diabetic disorders.
C - Cardiovascular system
C05 - Vasoprotectives
C05C - Capillary stabilizing agents
C05CA - Bioflavonoids
C05CA03 - Diosmin
Absorption
Diosmin is rapidly absorbed in the gastrointestinal tract. After a 900 mg single oral dose in a study using liquid chromatography with tandem mass spectrometry (LC-MS/MS) method, Cmax was 4.23.8 ngmL-1, Tmax was 18.79.9 hours, and AUC0~96 was 185.4166.2 ngmL-1 in healthy volunteers. Another pharmacokinetic study of 5 adults revealed a Cmax of 41794.1 ng/dL.
Route of Elimination
Pharmacokinetic data show absence of urinary elimination for diosmin and its aglycone diosmetin. Minor metabolites are found to be eliminated in the urine as glucuronic acid conjugates.
Volume of Distribution
A pharmacokinetic study of 5 adults revealed a volume of distribution of 62.17.9 L.
Degradation products of diosmin such as alkyl-phenolic acids confirm a metabolic pattern similar to that of other flavonoids.
Diosmin half-life ranges from 26 to 43 hours. One study using a liquid chromatography with tandem mass spectrometry (LC-MS/MS) method after a single 900 mg dose of diosmin demonstrated a half-life of 60.285.7 hours in healthy volunteers.
Diosmin helps to maintain circulatory system structure and function, particularly vein strength and competence. The molecular mechanism of action of diosmin has not been established. Several resources indicate that diosmin binds to the aryl hydrocarbon receptor, however clinical relevance to vascular function is unknown. One study demonstrates that oral diosmin exerts effects on the in vitro metabolism of noradrenaline by varicose veins, potentially benefitting vascular health.
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
36
PharmaCompass offers a list of Diosmin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Diosmin manufacturer or Diosmin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Diosmin manufacturer or Diosmin supplier.
A Diosmil manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Diosmil, including repackagers and relabelers. The FDA regulates Diosmil manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Diosmil API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Diosmil manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Diosmil supplier is an individual or a company that provides Diosmil active pharmaceutical ingredient (API) or Diosmil finished formulations upon request. The Diosmil suppliers may include Diosmil API manufacturers, exporters, distributors and traders.
click here to find a list of Diosmil suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Diosmil DMF (Drug Master File) is a document detailing the whole manufacturing process of Diosmil active pharmaceutical ingredient (API) in detail. Different forms of Diosmil DMFs exist exist since differing nations have different regulations, such as Diosmil USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Diosmil DMF submitted to regulatory agencies in the US is known as a USDMF. Diosmil USDMF includes data on Diosmil's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Diosmil USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Diosmil suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Diosmil Drug Master File in Korea (Diosmil KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Diosmil. The MFDS reviews the Diosmil KDMF as part of the drug registration process and uses the information provided in the Diosmil KDMF to evaluate the safety and efficacy of the drug.
After submitting a Diosmil KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Diosmil API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Diosmil suppliers with KDMF on PharmaCompass.
A Diosmil CEP of the European Pharmacopoeia monograph is often referred to as a Diosmil Certificate of Suitability (COS). The purpose of a Diosmil CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Diosmil EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Diosmil to their clients by showing that a Diosmil CEP has been issued for it. The manufacturer submits a Diosmil CEP (COS) as part of the market authorization procedure, and it takes on the role of a Diosmil CEP holder for the record. Additionally, the data presented in the Diosmil CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Diosmil DMF.
A Diosmil CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Diosmil CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Diosmil suppliers with CEP (COS) on PharmaCompass.
Diosmil Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Diosmil GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Diosmil GMP manufacturer or Diosmil GMP API supplier for your needs.
A Diosmil CoA (Certificate of Analysis) is a formal document that attests to Diosmil's compliance with Diosmil specifications and serves as a tool for batch-level quality control.
Diosmil CoA mostly includes findings from lab analyses of a specific batch. For each Diosmil CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Diosmil may be tested according to a variety of international standards, such as European Pharmacopoeia (Diosmil EP), Diosmil JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Diosmil USP).

