
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
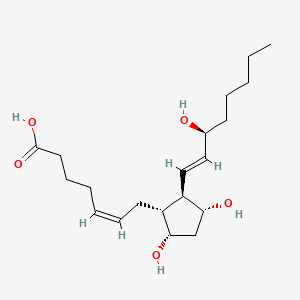

1. 9alpha,11beta Pgf2
2. 9alpha,11beta-pgf2
3. Alpha, Pgf2
4. Enzaprost F
5. Estrofan
6. F2 Alpha, Prostaglandin
7. F2alpha, Prostaglandin
8. Pgf2
9. Pgf2 Alpha
10. Pgf2alpha
11. Prostaglandin F2
12. Prostaglandin F2 Alpha
13. Prostaglandin F2alpha
1. Prostaglandin F2alpha
2. 551-11-1
3. Pgf2alpha
4. Prostaglandin F2a
5. Amoglandin
6. Enzaprost
7. Enzaprost F
8. Panacelan
9. L-pgf2-alpha
10. Prostaglandin F2 Alpha
11. Prostaglandin F2-alpha
12. Protamodin
13. L-prostaglandin F2-alpha
14. Cyclosin
15. Dinoprostum
16. Dinoprosta
17. Prostin F2alpha
18. Prosmon
19. Prostaglandin F2.alpha.
20. U-14583
21. Pgf2a
22. Prostarmon F
23. U 14583
24. Chembl815
25. B7in85g1hy
26. (z)-7-[(1r,2r,3r,5s)-3,5-dihydroxy-2-[(e,3s)-3-hydroxyoct-1-enyl]cyclopentyl]hept-5-enoic Acid
27. (z)-7-((1r,2r,3r,5s)-3,5-dihydroxy-2-((s,e)-3-hydroxyoct-1-en-1-yl)cyclopentyl)hept-5-enoic Acid
28. Chebi:15553
29. U-14,583
30. Ncgc00160385-01
31. 7-(3,5-dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl)-5-heptenoic Acid
32. Prostin F2-alpha
33. Pgf-2alpha
34. Pgf2 Alpha
35. Pgf2-alpha
36. 9s,11r,15s-trihydroxy-5z,13e-prostadienoic Acid
37. (+)-prostaglandin F2a
38. 9alpha,11alpha-pgf2
39. Pgf2
40. Prostin F 2 Alpha
41. Pgf2.alpha.
42. Prostaglandin F(sub 2a)
43. Dinoprostum [inn-latin]
44. (5z,13e,15s)-9alpha,11alpha,15-trihydroxyprosta-5,13-dien-1-oic Acid
45. 7-[3,5-dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl]-5-heptenoic Acid
46. Dinoprosta [inn-spanish]
47. (5z,9alpha,11alpha,13e,15s)-9,11,15-trihydroxyprosta-5,13-dien-1-oic Acid
48. Ccris 4253
49. Hsdb 3315
50. Dinoprost [usan:inn:ban:jan]
51. (+-)-pgf2alpha
52. 9a,11a-pgf2
53. Sr-01000721847
54. (e,z)-(1r,2r,3r,5s)-7-(3,5-dihydroxy-2-((3s)-(3-hydroxy-1-octenyl))cyclopentyl)-5-heptenoic Acid
55. (+-)-pgf2-alpha
56. Dl-prostaglandin F2-alpha
57. Pgf2-alpha Racemic Mixt.
58. Unii-b7in85g1hy
59. (+)-prostaglandin F2alpha
60. Prostaglandin F2alpha, Dl-
61. Brn 2225571
62. 9,11,15-trihydroxyprosta-5,13-dien-1-oic Acid
63. Prosmon (tn)
64. Prosta-5,13-dien-1-oic Acid, 9,11,15-trihydroxy-, (5z,9alpha,11alpha,13e,15s)-
65. 15-epi Pgf2alpha
66. Prostaglandin F2-alpha, Racemic Mixt.
67. Brn 4153898
68. Dinoprost [inn]
69. Dinoprost [jan]
70. Dinoprost [hsdb]
71. Dinoprost [usan]
72. Dsstox_cid_2946
73. Dinoprost [mart.]
74. (5z,9alpha,11alpha,13e,15s)-9,11,15-trihydroxyprosta-3,13-dien-1-oic Acid
75. 5-heptenoic Acid, 7-(3,5-dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl)-, L-
76. Dinoprost [who-dd]
77. Dsstox_rid_76800
78. Dsstox_gsid_22946
79. Schembl24292
80. Bspbio_001494
81. 23518-25-4
82. 5-heptenoic Acid, 7-(3,5-dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl)-, (1r-(1alpha(z),2beta(s*,e),3alpha,5alpha))-
83. Bml1-f08
84. Gtpl1884
85. Dinoprost (jp17/usan/inn)
86. 15(r)-prostaglandin F2 Alpha
87. 9,11,15-trihydroxy-(5z,9a,11a,13e,15s)-prosta-5,13-dien-1-oic Acid
88. Dtxsid9022946
89. Ex-a4115a
90. Hms1361k16
91. Hms1791k16
92. Hms1989k16
93. Hms2089f11
94. Hms3402k16
95. Hms3648n11
96. Amy30098
97. Zinc3830709
98. Tox21_111777
99. Bdbm50035622
100. Lmfa03010002
101. Mfcd00135231
102. Pdsp2_000079
103. Prostaglandin F2.alpha. [mi]
104. Ccg-208257
105. Cs-4232
106. Db12789
107. 5-heptenoic Acid, 7-(3,5-dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl)-, (+-)-
108. 5-heptenoic Acid, 7-(3,5-dihydroxy-2-(3-hydroxy-1-octenyl)cyclopentyl)-, Dl-
109. Idi1_033964
110. Ncgc00160385-02
111. Ncgc00160385-04
112. Ncgc00160385-05
113. Ncgc00160385-06
114. As-56892
115. Cas-551-11-1
116. Hy-12956
117. P1885
118. 62p015
119. C00639
120. D00081
121. A830494
122. Q421375
123. Sr-01000946457
124. Sr-01000721847-2
125. Sr-01000721847-4
126. Sr-01000946457-1
127. (5z,13e)-(15s)-9alpha,11alpha,15-trihydroxyprosta-5,13-dienoate
128. (5z,9alpha,11alpha,13e,15s)-9,11,15-trihydroxyprosta-5,13-dienoate
129. (5z,9alpha,11alpha,13e,15s)-9,11,15-trihydroxyprosta-5,13-dienoic Acid
130. Prosta-10,13-dien-1-oic Acid, 9,11,15-trihydroxy-, (5z,9.alpha.,11.alpha.,13e,15s)
131. (5z)-7-[(1r,2r,3r,5s)-3,5-dihydroxy-2-[(1e,3s)-3-hydroxyoct-1-en-1-yl]cyclopentyl]hept-5-enoic Acid
132. (z)-7-((1r,2r,3r,5s)-3,5-dihydroxy-2-((s,e)-3-hydroxyoct-1-enyl)-cyclopentyl)-hept-5-enoic Acid
133. (z)-7-[(1r,2r,3r,5s)-3,5-dihydroxy-2-((s)-(e)-3-hydroxyoct-1-enyl)-cyclopentyl]hept-5-enoic Acid
134. (z)-7-[(1r,2r,3r,5s)-3,5-dihydroxy-2-{(e)-(3s)-3-hydroxy-1-octenyl}-cyclopentyl]-5-heptenoic Acid
135. 13535-33-6
136. Prosta-5,13-dien-1-oic Acid, 9,11,15-trihydroxy-, (5z,9-alpha,11-alpha,13e,15s)-(+-)-
| Molecular Weight | 354.5 g/mol |
|---|---|
| Molecular Formula | C20H34O5 |
| XLogP3 | 2.7 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 12 |
| Exact Mass | 354.24062418 g/mol |
| Monoisotopic Mass | 354.24062418 g/mol |
| Topological Polar Surface Area | 98 Ų |
| Heavy Atom Count | 25 |
| Formal Charge | 0 |
| Complexity | 432 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 2 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Abortifacient Agents, Nonsteroidal; Oxytocics
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
...SOME PHYSICIANS PREFER TO DEFER ABORTION UNTIL AFTER 16TH WK, WHEN INTRA-AMNIOTIC ROUTE CAN BE EMPLOYED MORE SAFELY. /DINOPROST TROMETHAMINE/
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 625
EXTRA-AMNIOTIC TECHNIQUE, WHICH IS STILL INVESTIGATIONAL, REQUIRES TRANSVAGINAL INTRAUTERINE EXTRAOVULAR PLACEMENT OF INDWELLING CATHETER. ... RESERVED FOR TERMINATION OF PREGNANCIES BETWEEN 13TH & 15TH WK... /DINOPROST TROMETHAMINE/
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 625
DOSAGE THAT ACHIEVES EFFECTIVE CONCN WITHIN MYOMETRIUM IS LOWEST WITH INTRAUTERINE ADMIN. INTRA-AMNIOTIC INSTILLATION IS ACCOMPLISHED WITH LONG SPINAL NEEDLE INSERTED TRANSABDOMINALLY INTO AMNIOTIC SAC. /DINOPROST TROMETHAMINE/
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 625
For more Therapeutic Uses (Complete) data for PROSTAGLANDIN F2ALPHA (15 total), please visit the HSDB record page.
ASTHMATIC INDIVIDUALS ARE PARTICULARLY SENSITIVE, & PGF2ALPHA HAS CAUSED INTENSE BRONCHOSPASM.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 644
SOME INVESTIGATORS HAVE INDICATED THAT PROSTAGLANDINS MAY SHOW NARROWER DOSE-RESPONSE RANGE FOR PRODN OF PHYSIOLOGICAL CONTRACTIONS & OCCURRENCE OF UTERINE HYPERTONUS, POTENTIAL HAZARD THAT MAY BE AVOIDED BY VERY CAUTIOUS STEPWISE INCREMENTS IN RATE OF INFUSION. /PROSTAGLANDINS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 871
FOR ABORTION IN 2ND TRIMESTER...RESULTS IN...FREQUENT BUT TOLERABLE SIDE EFFECTS. HOWEVER, FOR VERY EARLY ABORTION (MENSES DELAYED UP TO SEVERAL WK), RATE OF SUCCESS REPORTED IS LOW & SERIOUS SIDE EFFECTS HAVE RESULTED FROM DOSES REQUIRED. /PROSTAGLANDINS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 871
...CONTRAINDICATED IN ACUTE PELVIC INFLAMMATION. IT SHOULD BE USED CAUTIOUSLY IN PT WITH HISTORY OF HYPERTENSION, ASTHMA, GLAUCOMA, EPILEPSY, OR CARDIOVASCULAR DISEASE.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 878
For more Drug Warnings (Complete) data for PROSTAGLANDIN F2ALPHA (11 total), please visit the HSDB record page.
Abortifacient Agents, Nonsteroidal
Non-steroidal chemical compounds with abortifacient activity. (See all compounds classified as Abortifacient Agents, Nonsteroidal.)
Oxytocics
Drugs that stimulate contraction of the myometrium. They are used to induce LABOR, OBSTETRIC at term, to prevent or control postpartum or postabortion hemorrhage, and to assess fetal status in high risk pregnancies. They may also be used alone or with other drugs to induce abortions (ABORTIFACIENTS). Oxytocics used clinically include the neurohypophyseal hormone OXYTOCIN and certain prostaglandins and ergot alkaloids. (From AMA Drug Evaluations, 1994, p1157) (See all compounds classified as Oxytocics.)
G - Genito urinary system and sex hormones
G02 - Other gynecologicals
G02A - Uterotonics
G02AD - Prostaglandins
G02AD01 - Dinoprost
AMONG COMPD OCCURRING IN SEMEN THAT ARE OF CURRENT RESEARCH INTEREST ARE PROSTAGLANDINS (25 MG/ML). /PROSTAGLANDINS/
Casarett, L.J., and J. Doull. Toxicology: The Basic Science of Poisons. New York: MacMillan Publishing Co., 1975., p. 263
APART FROM GENERALLY SLOW & SPECIES-DEPENDENT ISOMERIZATION, PGS E & F ARE RATHER STABLE IN BLOOD, BUT THEY ARE RAPIDLY DEGRADED & INACTIVATED BY TISSUE-BOUND ENZYMES; SOME 80-90% OR MORE IS DESTROYED DURING SINGLE PASSAGE THROUGH LIVER OR LUNGS. /PROSTAGLANDINS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 641
DEG OF BINDING OF PROSTAGLANDINS TO PLASMA PROTEINS APPEARS TO HAVE LITTLE INFLUENCE ON THEIR RATES OF METABOLISM & ELIMINATION. ... CLEARANCE RATES OF (3)H DID NOT CHANGE WHETHER PROSTAGLANDINS WERE ADMIN IN FREE FORM OR BOUND TO RAT PLASMA ALBUMIN. /PROSTAGLANDINS/
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 165
...PROSTAGLANDINS ARE RAPIDLY METABOLIZED... /PROSTAGLANDINS/
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 626
...THERE IS NO SINGLE PROSTAGLANDIN RECEPTOR. ... IT IS POSSIBLE TO DEMONSTRATE CALCIUM DEPENDENCE OF DIVERSE RESPONSES TO PROSTAGLANDINS & TO OBSERVE ALTERED CALCIUM FLUXES. ...PROSTAGLANDINS MAY EITHER STIMULATE OR INHIBIT ACCUM OF CYCLIC AMP. /PROSTAGLANDINS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 645
RESPONSES TO PROSTAGLANDIN F2ALPHA SHOW SPECIES VARIATION, BUT VASODILATATION HAS BEEN OBSERVED FOLLOWING INJECTION INTO HUMAN BRACHIAL ARTERY OF PGF2ALPHA... SUPERFICIAL VEINS OF HAND ARE CONTRACTED BY PFG2ALPHA...
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 642
PROMPT SUBSIDENCE OF PROGESTERONE OUTPUT & REGRESSION OF CORPUS LUTEUM FOLLOWS PARENTERAL INJECTION OF PROSTAGLANDIN F2ALPHA... THIS EFFECT INTERRUPTS EARLY PREGNANCY, WHICH IS DEPENDENT ON LUTEAL RATHER THAN PLACENTAL PROGESTERONE.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 645
OXYTOCIN & PROSTAGLANDINS AFFECT UTERINE SMOOTH MUSCLES BY DIFFERENT MECHANISMS, & THEIR EFFECTS ARE ADDITIVE. POSSIBLE ADVANTAGES OF THEIR COMBINED USE ARE BEING EXPLORED. /PROSTAGLANDINS/
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 871
Market Place

ABOUT THIS PAGE
75
PharmaCompass offers a list of Dinoprost API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Dinoprost manufacturer or Dinoprost supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Dinoprost manufacturer or Dinoprost supplier.
A Dinoprost manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Dinoprost, including repackagers and relabelers. The FDA regulates Dinoprost manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Dinoprost API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Dinoprost manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Dinoprost supplier is an individual or a company that provides Dinoprost active pharmaceutical ingredient (API) or Dinoprost finished formulations upon request. The Dinoprost suppliers may include Dinoprost API manufacturers, exporters, distributors and traders.
click here to find a list of Dinoprost suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Dinoprost Drug Master File in Japan (Dinoprost JDMF) empowers Dinoprost API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Dinoprost JDMF during the approval evaluation for pharmaceutical products. At the time of Dinoprost JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Dinoprost suppliers with JDMF on PharmaCompass.
Dinoprost Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Dinoprost GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Dinoprost GMP manufacturer or Dinoprost GMP API supplier for your needs.
A Dinoprost CoA (Certificate of Analysis) is a formal document that attests to Dinoprost's compliance with Dinoprost specifications and serves as a tool for batch-level quality control.
Dinoprost CoA mostly includes findings from lab analyses of a specific batch. For each Dinoprost CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Dinoprost may be tested according to a variety of international standards, such as European Pharmacopoeia (Dinoprost EP), Dinoprost JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Dinoprost USP).

