
Synopsis
Synopsis
0
USDMF
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
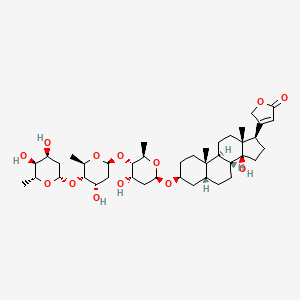

1. Awd, Digitoxin
2. Brger, Digitoxin
3. Coramedan
4. Didier, Digitoxin
5. Digimed
6. Digimerck
7. Digitaline Nativelle
8. Digitoxin Awd
9. Digitoxin Brger
10. Digitoxin Didier
11. Digitoxin Philo
12. Digitoxin-philo
13. Digophton
14. Nativelle, Digitaline
1. 71-63-6
2. Digitoxoside
3. Crystodigin
4. Digitoxinum
5. Unidigin
6. Digitophyllin
7. Cristapurat
8. Digitoksin
9. Purpurid
10. Glucodigin
11. Lanatoxin
12. Crystodigin (tn)
13. Digitalin, Crystalline
14. Digitoxin, Usp
15. Tradigal
16. Digitalis
17. Cardidigin
18. Carditoxin
19. Cardigin
20. Coramedan
21. Digimerck
22. Acedoxin
23. Digipural
24. Digicor
25. Digitrin
26. 1339-93-1
27. E90nzp2l9u
28. Mls000069787
29. Digitaline
30. Chebi:28544
31. Nsc-7529
32. 3-[(3s,5r,8r,9s,10s,13r,14s,17r)-3-[(2r,4s,5s,6r)-5-[(2s,4s,5s,6r)-5-[(2s,4s,5s,6r)-4,5-dihydroxy-6-methyloxan-2-yl]oxy-4-hydroxy-6-methyloxan-2-yl]oxy-4-hydroxy-6-methyloxan-2-yl]oxy-14-hydroxy-10,13-dimethyl-1,2,3,4,5,6,7,8,9,11,12,15,16,17-tetradecahydrocyclopenta[a]phenanthren-17-yl]-2h-furan-5-one
33. Asthenthilo
34. Digitossina
35. Digitoxosidum
36. Carditalin
37. Digitoksim
38. Digitoxin 100 Microg/ml In Acetonitrile
39. Digitoxina
40. Digitoxine
41. Smr000058529
42. Monodigitoxoside
43. Mono-digitoxid
44. Mono-glycocard
45. Dsstox_cid_2933
46. Dsstox_rid_76793
47. Dsstox_gsid_22933
48. Digitossina [dcit]
49. Nsc 7529
50. Mono-digitoxid [german]
51. Digitoxine [inn-french]
52. Digitoxinum [inn-latin]
53. Tri-digitoxoside [german]
54. Digitoxina [inn-spanish]
55. Digitoxigenin-tridigitoxosid
56. Cas-71-63-6
57. 3beta-[2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1-4)-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1-4)-2,6-dideoxy-beta-d-ribo-hexopyranosyloxy]-14-hydroxy-5beta-card-20(22)-enolide
58. Caswell No. 349
59. Digitoxigenin-tridigitoxosid [german]
60. Unii-e90nzp2l9u
61. Nsc7529
62. Ccris 7116
63. Hsdb 215
64. Digitoxin [usp:inn:ban:jan]
65. Digitoxin Solution
66. Ncgc00159428-02
67. 3-[(3s,5r,8r,9s,10s,13r,14s,17r)-3-[(2r,4s,5s,6r)-5-[(2s,4s,5s,6r)-5-[(2s,4s,5s,6r)-4,5-dihydroxy-6-methyl-tetrahydropyran-2-yl]oxy-4-hydroxy-6-methyl-tetrahydropyran-2-yl]oxy-4-hydroxy-6-methyl-tetrahydropyran-2-yl]oxy-14-hydroxy-10,13-dimethyl-1,2,3,4,5,6,7,8,9,11,12,15,16,17-tetradecahydrocyclopenta[a]phenanthren-17-yl]-2h-furan-5-one
68. 3-[(3s,5r,8r,9s,10s,13r,14s,17r)-3-[(2r,4s,5s,6r)-5-[(2s,4s,5s,6r)-5-[(2s,4s,5s,6r)-4,5-dihydroxy-6-methyloxan-2-yl]oxy-4-hydroxy-6-methyloxan-2-yl]oxy-4-hydroxy-6-methyloxan-2-yl]oxy-14-hydroxy-10,13
69. Einecs 200-760-5
70. Epa Pesticide Chemical Code 097002
71. Brn 0076678
72. Digitoxin For Lc Assay
73. Digitoxin [inn]
74. Digitoxin [jan]
75. Digitoxin [mi]
76. Digitoxin [hsdb]
77. Digitoxin [vandf]
78. Digitoxinum [hpus]
79. Digitoxin [usp-rs]
80. Digitoxin [who-dd]
81. Digitoxin [who-ip]
82. Digitoxin (jan/usp/inn)
83. Schembl20940
84. Chembl254219
85. Cid_441207
86. Gtpl6782
87. Megxp0_001901
88. Digitoxin [orange Book]
89. Digitoxin [ep Monograph]
90. Digitoxin [usp Impurity]
91. Dtxsid0022933
92. Regid_for_cid_441207
93. Acon0_000319
94. Acon1_000610
95. Bdbm46356
96. Digitoxin [usp Monograph]
97. Digitoxinum [who-ip Latin]
98. Card-20(22)-enolide, 3-((o-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl-(1->4)-o-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl)oxy)-14-hydroxy-, (3.beta.,5.beta.)-
99. Hy-b1357
100. Tox21_111659
101. Tox21_302269
102. Lmst01120018
103. Zinc95862733
104. Akos015915863
105. Digitoxin, >=92% (hplc), Powder
106. Tox21_111659_1
107. Ccg-208538
108. Cs-5502
109. Db01396
110. Smp1_000096
111. Digoxin Impurity A [ep Impurity]
112. Ncgc00094652-06
113. Ncgc00142623-02
114. Ncgc00142623-03
115. Ncgc00142623-04
116. Ncgc00142623-05
117. Ncgc00255265-01
118. (3beta,5beta)-3-{[2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-beta-d-ribo-hexopyranosyl]oxy}-14-hydroxycard-20(22)-enolide
119. As-75726
120. Card-20(22)-enolide, 3-((o-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1-4)-o-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1-4)-2,6-dideoxy-beta-d-ribo-hexopyranosyl)oxy)-14-hydroxy-, (3beta,5beta)-
121. Sbi-0051348.p003
122. D0542
123. C06955
124. D00297
125. D89676
126. Ncgc00142623-05_c41h64o13_crystodigin
127. 071d636
128. Q423890
129. Q-100791
130. Sr-01000721879-2
131. Brd-k63668566-001-01-7
132. A572a148-ec30-47d2-a32a-c9d8a93f8efc
133. Digitoxin, British Pharmacopoeia (bp) Reference Standard
134. Z1558290128
135. Digitoxin, European Pharmacopoeia (ep) Reference Standard
136. Digitoxin, United States Pharmacopeia (usp) Reference Standard
137. (3.beta.,5.beta.)-3-((o-2,6-dideoxy-.beta.-d-ribo-hexapyranosyl-(1->4)-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl)oxy)-14-hydroxycard-20(22)-enolide
138. (3.beta.,5.beta.)-3-((o-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl-(1->4)-o-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl)oxy)-14-hydroxycard-20(22)-enolide
139. 3-[(3s,5r,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-3-[(2r,4s,5s,6r)-6-methyl-5-[(2s,4s,5s,6r)-6-methyl-5-[(2s,4s,5s,6r)-6-methyl-4,5-bis(oxidanyl)oxan-2-yl]oxy-4-oxidanyl-oxan-2-yl]oxy-4-oxidanyl-oxan-2-yl]oxy-14-oxidanyl-1,2,3,4,5,6,7,8,9,11,12,15,16,17-tetradecahydrocyclopenta[a]phenanthren-17-yl]-2h-furan-5-one
140. 3.beta.-((2,6-dideoxy-.beta.-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl)oxy)-14-hydroxy-5.beta.-card-20(22)-enolide
141. 3beta-[2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-beta-d-ribo-hexopyranosyloxy]-14-hydroxy-5beta-card-20(22)-enolide
142. 4-((1s,2s,5s,11s,7r,10r,14r,15r)-5-{5-[5-((2s,4s,5s,6r)-4,5-dihydroxy-6-methyl (2h-3,4,5,6-tetrahydropyran-2-yloxy))(4s,5s,2r,6r)-4-hydroxy-6-methyl(2h-3,4,5 ,6-tetrahydropyran-2-yloxy)](4s,5s,2r,6r)-4-hydroxy-6-methyl(2h-3,4,5,6-tetrah Ydropyran-2-yloxy)
143. 4-((3s,5r,8r,9s,10s,13r,14s,17r)-3-(((2r,4s,5s,6r)-5-(((2s,4s,5s,6r)-5-(((2s,4s,5s,6r)-4,5-dihydroxy-6-methyltetrahydro-2h-pyran-2-yl)oxy)-4-hydroxy-6-methyltetrahydro-2h-pyran-2-yl)oxy)-4-hydroxy-6-methyltetrahydro-2h-pyran-2-yl)oxy)-14-hydroxy-10,13-dimethylhexadecahydro-1h-cyclopenta[a]phenanthren-17-yl)furan-2(5h)-one
144. 4-[(1s,2s,5s,7r,10r,11s,14r,15r)-5-{[(2r,4s,5s,6r)-5-{[(2s,4s,5s,6r)-5-{[(2s,4s,5s,6r)-4,5-dihydroxy-6-methyloxan-2-yl]oxy}-4-hydroxy-6-methyloxan-2-yl]oxy}-4-hydroxy-6-methyloxan-2-yl]oxy}-11-hydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-yl]-2,5-dihydrofuran-2-one
145. 4-[(3s,5r,8r,9s,10s,13r,14s,17r)-3-[(2r,4s,5s,6r)-5-[(2s,4s,5s,6r)-5-[(2s,4s,5s,6r)-4,5-dihydroxy-6-methyloxan-2-yl]oxy-4-hydroxy-6-methyloxan-2-yl]oxy-4-hydroxy-6-methyloxan-2-yl]oxy-14-hydroxy-10,13-dimethyl-1,2,3,4,5,6,7,8,9,11,12,15,16,17-tetradecahydrocyclopenta[a]phenanthren-17-yl]-5h-furan-2-one
146. Card-20(22)-enolide, 3-((o-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl-(1-4)-o-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl-(1-4)-2,6-dideoxy-.beta.-d-ribo-hexopyranosyl)oxy)-14-hydroxy-, (3beta,5beta)-
147. Card-20(22)-enolide, 3-((o-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1->4)-o-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1->4)-2,6-dideoxy-beta-d-ribo-hexopyranosyl)oxy)-14-hydroxy-, (3beta,5beta)-
148. Card-20(22)-enolide, 3-((o-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1-4)-o-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1-4)-2,6-dideoxy-beta-d-ribo-hexopyranosyl)oxy)-14-hydroxy, (3beta,5beta)-
149. Card-20(22)-enolide, 3-((o-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1.fwdarw.4)-o-2,6-dideoxy-beta-d-ribo-hexopyranosyl-(1.fwdarw.4)-2,6-dideoxy-beta-d-ribo-hexopyranosyl)oxy)-14-hydroxy-, (3beta,5beta)-
| Molecular Weight | 764.9 g/mol |
|---|---|
| Molecular Formula | C41H64O13 |
| XLogP3 | 2.3 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 7 |
| Exact Mass | 764.43469209 g/mol |
| Monoisotopic Mass | 764.43469209 g/mol |
| Topological Polar Surface Area | 183 Ų |
| Heavy Atom Count | 54 |
| Formal Charge | 0 |
| Complexity | 1410 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 20 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Arrhythmia Agents; Cardiotonic Agents; Enzyme Inhibitors
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
MEDICATION (VET): cardiac tonic
Budavari, S. (ed.). The Merck Index - Encyclopedia of Chemicals, Drugs and Biologicals. Rahway, NJ: Merck and Co., Inc., 1989., p. 498
Cardiac glycosides are used principally in the prophylactic management and treatment of heart failure and to control the ventricular rate in patients with atrial fibrillation. ... Digoxin is the most commonly used cardiac glycoside, principally because it may be administered by various routes, has an intermediate duration of action, and has been extensively studied in patients with or without renal insufficiency. Some clinicians believe that digitoxin is the cardiac glycoside of choice in patients with renal failure because elimination half life in unchanged in these patients; however, digitoxin is no longer commercially available in the US. ...
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018, p. 1812
Cardiac glycosides are used, usually in conjunction with other agents, in the management of symptomatic congestive heart failure associated with left ventricular systolic dysfunction. /Cardiac glycosides/
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018, p. 1812
For more Therapeutic Uses (Complete) data for Digitoxin (14 total), please visit the HSDB record page.
Cardiac glycosides should be used with caution in patients with severe pulmonary disease, hypoxia, myxedema, acute myocardial infarction, severe heart failure, acute myocarditis (including rheumatic carditis) or an otherwise damaged myocardium, since the likelihood of cardiac glycoside-induced arrhythmias is increased in these patients. The possibility that use of cardiac lycosides in some patient with acute myocardial infarction may result in an undesirable increase in oxygen demand and associated ischemia should be considered. In patients with rheumatic carditis, dosage should be low initially and increased gradually until a beneficial effect is obtained or, if improvement does not occur in these patients, the drug should be discontinued. Cardiac glycosides should be used with caution in patient with chronic constrictive pericarditis since these patients may respond unfavorably. Cardiac glycosides should be administered with extreme caution in patients with acute glomerulonephritis and congestive heart failure; if the drugs are necessary, total daily dosage must be reduced and given in divided doses with constant ECG monitoring. These patients should be treated concomitantly with diuretics and hypotensive agents and the glycoside should be discontinued as soon as possible. Cardiac glycosides should also be used with extreme caution, if at all, in patients with idiopathic hypertrophic subaortic stenosis because increased obstruction to left ventricular outflow may result. Patients with certain ejection fraction (e.g., restrictive cardiomyopathy, constrictive pericarditis, amyloid heart disease, acute cor pulmonale) may be particularly susceptible to the toxicity of cardiac glycosides. /Cardiac glycosides/
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018, p. 1814
Cardiac glycosides should be given iv with caution in hypertensive patients, since iv administration of these drugs may increase blood pressure transiently. /Cardiac glycosides/
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018, p. 1814
Safe use of cardiac glycosides during pregnancy has not been established. Although the drugs have been used in pregnant women without apparent harm to the mother or fetus, one neonatal death has been reported, allegedly due to digitoxin (no longer commercially available in the US) overdosage in utero.
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018, p. 1814
Cardiac glycosides should not be administered to patients with substantial sinus or atrioventricular (VA) block, unless the conduction block has been addressed with a permanent pacemaker. The drugs should be used cautiously with other drugs that can depress sinus or AV nodal function. /Cardiac glycosides/
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018, p. 1814
For more Drug Warnings (Complete) data for Digitoxin (24 total), please visit the HSDB record page.
For the treatment and management of congestive cardiac insufficiency, arrhythmias and heart failure.
Digitoxin is a cardiac glycoside sometimes used in place of DIGOXIN. It has a longer half-life than digoxin; toxic effects, which are similar to those of digoxin, are longer lasting (From Martindale, The Extra Pharmacopoeia, 30th ed, p665). It is eliminated hepatically making it useful in patients with poor or erratic kidney function, although it is now rarely used in practice. Digitoxin lacks the strength of evidence that digoxin has in the management of heart failure.
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Anti-Arrhythmia Agents
Agents used for the treatment or prevention of cardiac arrhythmias. They may affect the polarization-repolarization phase of the action potential, its excitability or refractoriness, or impulse conduction or membrane responsiveness within cardiac fibers. Anti-arrhythmia agents are often classed into four main groups according to their mechanism of action: sodium channel blockade, beta-adrenergic blockade, repolarization prolongation, or calcium channel blockade. (See all compounds classified as Anti-Arrhythmia Agents.)
Cardiotonic Agents
Agents that have a strengthening effect on the heart or that can increase cardiac output. They may be CARDIAC GLYCOSIDES; SYMPATHOMIMETICS; or other drugs. They are used after MYOCARDIAL INFARCT; CARDIAC SURGICAL PROCEDURES; in SHOCK; or in congestive heart failure (HEART FAILURE). (See all compounds classified as Cardiotonic Agents.)
C - Cardiovascular system
C01 - Cardiac therapy
C01A - Cardiac glycosides
C01AA - Digitalis glycosides
C01AA04 - Digitoxin
Postmortem digitoxin levels in the choroid-retina and vitreous humor of patients who had undergone digitoxin therapy (therapeutic group) and in one case of suicidal digitoxin poisoning were measured and compared with levels in femoral vein blood, myocardium, kidney and liver. The results were interpreted in light of the medical history of each patient. The digitoxin level in the choroid-retina of the single case of suicidal poisoning was far higher than the choroid-retinal levels in the therapeutic group. In the latter, variation in choroid-retinal levels was comparable to that in the other tissues. In cases where the choroid-retina of the right and left eyes were examined, digitoxin levels in both eyes were essentially equal. There was no indication of significant changes in choroid-retinal levels due to postmortem diffusion of digitoxin into the vitreous body. Based on these results, determination of digitoxin levels in the choroid-retina could contribute to improving postmortem diagnosis of lethal digitoxin poisoning.
PMID:1419876 Ritz S et al; Int J Legal Med 105 (3): 155-9 (1992)
In cats ... 100% of digitoxin is absorbed in 80-100 min following duodenal admin ... digitalis glycosides are transported by blood ... bound to ... albumin, and in part free. ... Tissue distribution is not primarily to heart ... /highest concentration/ ... in excretory organs (liver, bile, intestinal tract, kidney) ... .
Jones, L.M., et al. Veterinary Pharmacology & Therapeutics. 4th ed. Ames: Iowa State University Press, 1977., p. 509
It is not predictably absorbed from gut of dogs... .
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 173
...Prolonged biological half-life of digitoxin and its metabolites appears to depend on recirculation of free drug molecules after biliary excretion as glucuronide and sulfate conjugate.
Testa, B. and P. Jenner. Drug Metabolism: Chemical & Biochemical Aspects. New York: Marcel Dekker, Inc., 1976., p. 449
For more Absorption, Distribution and Excretion (Complete) data for Digitoxin (14 total), please visit the HSDB record page.
Hepatic.
Eliminated by hepatic degradation ...to inactive genins ...Stepwise hydrolysis of 3 molecules of digitoxose converts glycoside to aglycone digitoxigenin, which is ...converted to inactive epidigitoxigenin. Because enterohepatic recirculation occurs, approx 25% of metabolic end products appear in stool.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 674
Studies with various tissues of guinea pig showed that liver, kidney, and adrenal tissues converted digitoxin to digoxin.
Casarett, L.J., and J. Doull. Toxicology: The Basic Science of Poisons. New York: MacMillan Publishing Co., 1975., p. 119
Digitoxin in bile of rats was excreted largely in form of glucuronide of digitoxigenin monodigitoxoside.
PMID:20296 Richards LG et al; Drug Metab Dispos 5 (5): 469-73 (1977)
Cardiac glycosides undergo varying degrees of hepatic metabolism, enterohepatic circulation, and renal filtration and reabsorption depending on their polarity and lipid solubility. ... Less polar glycosides such as digitoxin are metabolized extensively before they are excreted. Metabolism includes stepwise cleavage of the sugar molecules, hydroxylation, epimerization, and formation of glucuronide and sulfate conjugates. /Cardiac glycosides/
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018, p. 1819
For more Metabolism/Metabolites (Complete) data for Digitoxin (6 total), please visit the HSDB record page.
The digitoxin half-life in elderly patients in the eight and ninth decade was more prolonged (mean +/- SD: 25 +/- 9 days) than in younger people (6.7 +/- 1.7). These elderly patients accumulated digitoxin even on a dose of 0.05 mg/ day. The symptoms of digitoxin intoxication disappeared on discontinuation of medication. When digitoxin is used in the treatment for heart failure in the very elderly patients, one should be aware of the possibility of digitoxin intoxication, even on a low dose.
PMID:16296683 Bohmer T1, Roseth A; Age Ageing 27 (2): 222-4 (1998)
After administration of 0.6 mg digitoxin mean serum digitoxin half-life of 4.3 days and 8.1 days respectively observed in cholecystectomized heart patients and control subjects.
PMID:1120397 Storstein L; Clin Pharmacol Ther 17 (3): 313-20 (1975)
The elimination half-life of digitoxin is usually 5-7 days, but may range from 4-14 days. The elimination half-life of digitoxin is generally unchanged in patients with renal failure. In patients with biliary fistulas, plasma half-life is decreased by about 50%. Variability among patients in the degree of enterohepatic recycling of digitoxin may account for part of the variability in plasma half-life in some patients. The elimination half-life of digitoxin is prolonged in hypothyroid patients and decreased in hyperthyroid patients.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 93. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1993 (Plus Supplements, 1993)., p. 893
Digitoxin inhibits the Na-K-ATPase membrane pump, resulting in an increase in intracellular sodium and calcium concentrations. Increased intracellular concentrations of calcium may promote activation of contractile proteins (e.g., actin, myosin). Digitoxin also acts on the electrical activity of the heart, increasing the slope of phase 4 depolarization, shortening the action potential duration, and decreasing the maximal diastolic potential.
AIMS: Recent studies suggest that proarrhythmic effects of cardiac glycosides (CGs) on cardiomyocyte Ca(2+) handling involve generation of reactive oxygen species (ROS). However, the specific pathway(s) of ROS production and the subsequent downstream molecular events that mediate CG-dependent arrhythmogenesis remain to be defined. METHODS AND RESULTS: We examined the effects of digitoxin (DGT) on Ca(2+) handling and ROS production in cardiomyocytes using a combination of pharmacological approaches and genetic mouse models. Myocytes isolated from mice deficient in NADPH oxidase type 2 (NOX2KO) and mice transgenically overexpressing mitochondrial superoxide dismutase displayed markedly increased tolerance to the proarrhythmic action of DGT as manifested by the inhibition of DGT-dependent ROS and spontaneous Ca(2+) waves (SCW). Additionally, DGT-induced mitochondrial membrane potential depolarization was abolished in NOX2KO cells. DGT-dependent ROS was suppressed by the inhibition of PI3K, PKC, and the mitochondrial KATP channel, suggesting roles for these proteins, respectively, in activation of NOX2 and in mitochondrial ROS generation. Western blot analysis revealed increased levels of oxidized CaMKII in WT but not in NOX2KO hearts treated with DGT. The DGT-induced increase in SCW frequency was abolished in myocytes isolated from mice in which the Ser 2814 CaMKII phosphorylation site on RyR2 is constitutively inactivated. CONCLUSION: These results suggest that the arrhythmogenic adverse effects of CGs on Ca(2+) handling involve PI3K- and PKC-mediated stimulation of NOX2 and subsequent NOX2-dependent ROS release from the mitochondria; mitochondria-derived ROS then activate CaMKII with consequent phosphorylation of RyR2 at Ser 2814.
PMID:24104877 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3868350 Ho HT et al; Cardiovasc Res 101 (1):165-74 (2014)
Pro-inflammatory processes initiated in the endothelium represent a crucial step in the pathogenesis of inflammatory cardiovascular disease, such as atherosclerosis. Recent observations pointed to potential anti-inflammatory properties of the cardiac glycoside digitoxin. Therefore, the present study investigated potential anti-inflammatory and vasoprotective properties of digitoxin as well as the underlying signaling pathways affected in endothelial cells (EC). Digitoxin, employing therapeutical concentrations used in patients (3-30 nM), potently inhibited the IL-1beta-induced expression of MCP-1 and VCAM-1 in EC and the capacity of corresponding cell culture supernatants on monocyte migration as well as monocyte adhesion to endothelial monolayers, respectively. Furthermore, digitoxin prevented the IL-1beta-induced activation of p44/42-MAPK and NF-kappaB without affecting activation of JNK and p38-MAPK. Inhibition of NF-kappaB signaling but not p44/42-MAPK mimicked the observed inhibitory effects of digitoxin on MCP-1 expression and monocyte migration. Moreover, digitoxin inhibited NF-kappaB signaling at the level of TAK-1/IKK. Additionally, digitoxin prevented TNF-alpha-induced apoptosis in EC accompanied by activation of Akt. Blockade of PI-3-kinase, activator of Akt, prevented the anti-apoptotic properties of digitoxin and impaired its inhibitory action on NF-kappaB signaling and MCP-1 expression. Finally, digitoxin activated endothelial NO-synthase, which was blocked by inhibition of PI-3-kinase, Ca(2+)/Calmodulin-dependent-proteinkinase-II and chelation of intracellular calcium. Digitoxin elicits anti-inflammatory and vasoprotective properties by blocking NF-kappaB and activating PI-3-kinase/Akt signaling as well as Ca(2+)/Calmodulin-dependent-proteinkinase-II in EC. These observations indicate a potential therapeutical application of digitoxin in the treatment of cardiovascular diseases, such as atherosclerosis.
PMID:19446813 Jagielska J et al; Atherosclerosis 206 (2): 390-6 (2009)
Cardiac glycosides have been used in the treatment of arrhythmias for more than 200 years. Two-pore-domain (K2P) potassium channels regulate cardiac action potential repolarization. Recently, K2P3.1 [tandem of P domains in a weak inward rectifying K+ channel (TWIK)-related acid-sensitive K+ channel (TASK)-1] has been implicated in atrial fibrillation pathophysiology and was suggested as an atrial-selective antiarrhythmic drug target. We hypothesized that blockade of cardiac K2P channels contributes to the mechanism of action of digitoxin and digoxin. All functional human K2P channels were screened for interactions with cardiac glycosides. Human K2P channel subunits were expressed in Xenopus laevis oocytes, and voltage clamp electrophysiology was used to record K+ currents. Digitoxin significantly inhibited K2P3.1 and K2P16.1 channels. By contrast, digoxin displayed isolated inhibitory effects on K2P3.1. K2P3.1 outward currents were reduced by 80% (digitoxin, 1 Hz) and 78% (digoxin, 1 Hz). Digitoxin inhibited K2P3.1 currents with an IC50 value of 7.4 uM. Outward rectification properties of the channel were not affected. Mutagenesis studies revealed that amino acid residues located at the cytoplasmic site of the K2P3.1 channel pore form parts of a molecular binding site for cardiac glycosides. In conclusion, cardiac glycosides target human K2P channels. The antiarrhythmic significance of repolarizing atrial K2P3.1 current block by digoxin and digitoxin requires validation in translational and clinical studies.
PMID:29643254 Schmidt C et al; J Pharmacol Exp Ther 365 (3): 614-623 (2018)
Cardiac glycosides inhibit the activity of sodium-potassium-activated adenosine triphosphatase (Na+-K+-ATPase), an enzyme required for active transport of sodium across myocardial cell membranes. Inhibition of this enzyme in cardiac cells results in an increase in the contractile state of the heart and it was believed that benefits of cardiac glycosides in heart failure were mainly associated with inotropic action. However, it has been suggested that benefits of cardiac glycosides may be in part related to enzyme inhibition in noncardiac tissues. Inhibition of Na+-K+-ATPase in vagal afferents acts to sensitize cardiac baroreceptors which may in turn decrease sympathetic outflow from the CNS. In addition, by inhibiting Na+-K+-ATPase in the kidney, cardiac glycosides decrease the renal tubular reabsorption of sodium; the resulting increase in the delivery of sodium to the distal tubules leads to the suppression of renin secretion from the kidneys. These observations led to the hypothesis that cardiac glycosides act in heart failure principally by attenuating the activation of the neurohormonal system, rather than by a positive inotropic action. Toxic doses of cardiac glycosides cause efflux of potassium from the myocardium and concurrent influx of sodium. Toxicity results in part from loss of intracellular potassium associated with inhibition of Na+-K+-ATPase. With therapeutic doses, augmentation of calcium influx to the contractile proteins with resultant enhancement of excitation-contraction coupling is involved in the positive inotropic action of cardiac glycosides; the role of Na+-K+-ATPase in this effect is controversial. /Cardiac glycosides/
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018, p. 1818
Low concentrations of cardiac glycosides including ouabain, digoxin, and digitoxin block cancer cell growth without affecting Na+,K+-ATPase activity, but the mechanism underlying this anti-cancer effect is not fully understood. Volume-regulated anion channel (VRAC) plays an important role in cell death signaling pathway in addition to its fundamental role in the cell volume maintenance. Here, we report cardiac glycosides-induced signaling pathway mediated by the crosstalk between Na+,K+-ATPase and VRAC in human cancer cells. Submicromolar concentrations of ouabain enhanced VRAC currents concomitantly with a deceleration of cancer cell proliferation. The effects of ouabain were abrogated by a specific inhibitor of VRAC (DCPIB) and knockdown of an essential component of VRAC (LRRC8A), and they were also attenuated by the disruption of membrane microdomains or the inhibition of NADPH oxidase. Digoxin and digitoxin also showed anti-proliferative effects in cancer cells at their therapeutic concentration ranges, and these effects were blocked by DCPIB. In membrane microdomains of cancer cells, LRRC8A was found to be co-immunoprecipitated with Na+,K+-ATPase a1-isoform. These ouabain-induced effects were not observed in non-cancer cells. Therefore, cardiac glycosides were considered to interact with Na+,K+-ATPase to stimulate the production of reactive oxygen species, and they also apparently activated VRAC within membrane microdomains, thus producing anti-proliferative effects.
PMID:30251696 Fujii T et al; Biochim Biophys Acta Mol Basis Dis 1864 (11): 3792-3804 (2018)
Global Sales Information

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
57
PharmaCompass offers a list of Digitoxin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Digitoxin manufacturer or Digitoxin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Digitoxin manufacturer or Digitoxin supplier.
A Digitoxin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Digitoxin, including repackagers and relabelers. The FDA regulates Digitoxin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Digitoxin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Digitoxin supplier is an individual or a company that provides Digitoxin active pharmaceutical ingredient (API) or Digitoxin finished formulations upon request. The Digitoxin suppliers may include Digitoxin API manufacturers, exporters, distributors and traders.
click here to find a list of Digitoxin suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Digitoxin CEP of the European Pharmacopoeia monograph is often referred to as a Digitoxin Certificate of Suitability (COS). The purpose of a Digitoxin CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Digitoxin EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Digitoxin to their clients by showing that a Digitoxin CEP has been issued for it. The manufacturer submits a Digitoxin CEP (COS) as part of the market authorization procedure, and it takes on the role of a Digitoxin CEP holder for the record. Additionally, the data presented in the Digitoxin CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Digitoxin DMF.
A Digitoxin CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Digitoxin CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Digitoxin suppliers with CEP (COS) on PharmaCompass.
Digitoxin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Digitoxin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Digitoxin GMP manufacturer or Digitoxin GMP API supplier for your needs.
A Digitoxin CoA (Certificate of Analysis) is a formal document that attests to Digitoxin's compliance with Digitoxin specifications and serves as a tool for batch-level quality control.
Digitoxin CoA mostly includes findings from lab analyses of a specific batch. For each Digitoxin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Digitoxin may be tested according to a variety of international standards, such as European Pharmacopoeia (Digitoxin EP), Digitoxin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Digitoxin USP).

