
Synopsis
Synopsis
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
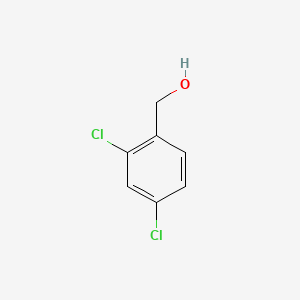

1. Dichlorobenzyl Alcohol
1. 1777-82-8
2. (2,4-dichlorophenyl)methanol
3. Dybenal
4. 2,4-dichlorobenzenemethanol
5. Rapidosept
6. Benzenemethanol, 2,4-dichloro-
7. Myacide Sp
8. Benzyl Alcohol, 2,4-dichloro-
9. Chebi:48220
10. Mfcd00004606
11. Nsc-15635
12. 1nkx3648j9
13. Ncgc00181337-01
14. Dsstox_cid_21362
15. Dsstox_rid_79699
16. Dsstox_gsid_41362
17. Cas-1777-82-8
18. 2,4-dichlorobenzylalcohol
19. Einecs 217-210-5
20. Nsc 15635
21. 2,4-dichlorobenzoyl Alcohol
22. Brn 1448652
23. Unii-1nkx3648j9
24. Ai3-20619
25. Schembl41323
26. Oxiconazole Related Compound D
27. Chembl3184437
28. Dtxsid9041362
29. (2,4-dichlorophenyl)methanol #
30. Dalc2-h_000039
31. Zinc157458
32. 2,4-dichlorobenzyl Alcohol, 99%
33. Dichlorobenzyl Alcohol [ii]
34. Dichlorobenzyl Alcohol [mi]
35. Nsc15635
36. Tox21_112796
37. Tox21_300895
38. Bdbm50498198
39. Dichlorobenzyl Alcohol [inci]
40. S6067
41. Stl480884
42. Dichlorobenzyl Alcohol [vandf]
43. Akos000248902
44. Dichlorobenzyl Alcohol [mart.]
45. Tox21_112796_1
46. Am81457
47. Db13269
48. Dichlorobenzyl Alcohol [who-dd]
49. Hy-w039454
50. Ps-5315
51. Ncgc00181337-02
52. Ncgc00181337-03
53. Ncgc00254799-01
54. Ac-10679
55. Sy015811
56. Db-015914
57. Cs-0097299
58. D1837
59. Ft-0610050
60. Ft-0674330
61. 77d828
62. Oxiconazole Related Compound D [usp-rs]
63. 2,4-dichlorobenzyl Alcohol [ep Monograph]
64. Q209202
65. Sr-01000944729
66. Sr-01000944729-1
67. W-107827
68. (2,4-dichlorophenyl)methanol;2,4-dichlorobenzyl Alcohol
69. Z111479786
70. 2,4-dichlorobenzyl Alcohol, European Pharmacopoeia (ep) Reference Standard
| Molecular Weight | 177.02 g/mol |
|---|---|
| Molecular Formula | C7H6Cl2O |
| XLogP3 | 2.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Exact Mass | 175.9795702 g/mol |
| Monoisotopic Mass | 175.9795702 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 108 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Dichlorobenzyl alcohol in combination with [DB13908] is available in over-the-counter products used for symptomatic relief of acute sore throat and postoperative sore throat.
In vitro studies with the combination of dichlorobenzyl alcohol and amylmetacresol have shown a virucidal against a number of viruses associated with the common cold which is observed by a reduction in the viral load. In clinical trials, administration of dichlorobenzyl alcohol lozenges has been shown to generate a reduced throat soreness and to provide pain relief and relief from difficulty in swallowing 5 minutes after administration. This effect can last for even 2 hours. The relief effect was shown to reach a steady-state after 45 minutes.
R - Respiratory system
R02 - Throat preparations
R02A - Throat preparations
R02AA - Antiseptics
R02AA03 - Dichlorobenzyl alcohol
Absorption
Dichlorobenzyl alcohol is released almost immediately from its formulation and reaches peak concentration after 3-4 minutes. The concentration in saliva after 120 minutes represents about 50% of the administered dose.
Route of Elimination
In preclinical trials, dermal administration of dichlorobenzyl alcohol results in renal elimination of 90% of the administered dose. After metabolism, dichlorobenzyl alcohol is excreted in the urine.
Volume of Distribution
This pharmacokinetic property has not been fully studied.
Clearance
This pharmacokinetic property has not been fully studied.
Dichlorobenzyl alcohol is metabolized in the liver to form hippuric acid.
This pharmacokinetic property has not been fully studied.
The use of dichlorobenzyl alcohol has been related to its antibacterial, antiviral and local anesthetic properties. The local anesthetic action of dichlorobenzyl alcohol is thought to be due to a reduced sodium channel blockade. The antiseptic mechanism of action of dichlorobenzyl alcohol is not fully understood but it is thought to be related to a denaturation of external proteins and rearrangement of the tertiary structure proteins.
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
36
PharmaCompass offers a list of 2,4-Dichlorobenzyl Alcohol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 2,4-Dichlorobenzyl Alcohol manufacturer or 2,4-Dichlorobenzyl Alcohol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred 2,4-Dichlorobenzyl Alcohol manufacturer or 2,4-Dichlorobenzyl Alcohol supplier.
A Dichlorobenzyl Alcohol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Dichlorobenzyl Alcohol, including repackagers and relabelers. The FDA regulates Dichlorobenzyl Alcohol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Dichlorobenzyl Alcohol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Dichlorobenzyl Alcohol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Dichlorobenzyl Alcohol supplier is an individual or a company that provides Dichlorobenzyl Alcohol active pharmaceutical ingredient (API) or Dichlorobenzyl Alcohol finished formulations upon request. The Dichlorobenzyl Alcohol suppliers may include Dichlorobenzyl Alcohol API manufacturers, exporters, distributors and traders.
click here to find a list of Dichlorobenzyl Alcohol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Dichlorobenzyl Alcohol DMF (Drug Master File) is a document detailing the whole manufacturing process of Dichlorobenzyl Alcohol active pharmaceutical ingredient (API) in detail. Different forms of Dichlorobenzyl Alcohol DMFs exist exist since differing nations have different regulations, such as Dichlorobenzyl Alcohol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Dichlorobenzyl Alcohol DMF submitted to regulatory agencies in the US is known as a USDMF. Dichlorobenzyl Alcohol USDMF includes data on Dichlorobenzyl Alcohol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Dichlorobenzyl Alcohol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Dichlorobenzyl Alcohol suppliers with USDMF on PharmaCompass.
A Dichlorobenzyl Alcohol CEP of the European Pharmacopoeia monograph is often referred to as a Dichlorobenzyl Alcohol Certificate of Suitability (COS). The purpose of a Dichlorobenzyl Alcohol CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Dichlorobenzyl Alcohol EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Dichlorobenzyl Alcohol to their clients by showing that a Dichlorobenzyl Alcohol CEP has been issued for it. The manufacturer submits a Dichlorobenzyl Alcohol CEP (COS) as part of the market authorization procedure, and it takes on the role of a Dichlorobenzyl Alcohol CEP holder for the record. Additionally, the data presented in the Dichlorobenzyl Alcohol CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Dichlorobenzyl Alcohol DMF.
A Dichlorobenzyl Alcohol CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Dichlorobenzyl Alcohol CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Dichlorobenzyl Alcohol suppliers with CEP (COS) on PharmaCompass.
A Dichlorobenzyl Alcohol written confirmation (Dichlorobenzyl Alcohol WC) is an official document issued by a regulatory agency to a Dichlorobenzyl Alcohol manufacturer, verifying that the manufacturing facility of a Dichlorobenzyl Alcohol active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Dichlorobenzyl Alcohol APIs or Dichlorobenzyl Alcohol finished pharmaceutical products to another nation, regulatory agencies frequently require a Dichlorobenzyl Alcohol WC (written confirmation) as part of the regulatory process.
click here to find a list of Dichlorobenzyl Alcohol suppliers with Written Confirmation (WC) on PharmaCompass.
Dichlorobenzyl Alcohol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Dichlorobenzyl Alcohol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Dichlorobenzyl Alcohol GMP manufacturer or Dichlorobenzyl Alcohol GMP API supplier for your needs.
A Dichlorobenzyl Alcohol CoA (Certificate of Analysis) is a formal document that attests to Dichlorobenzyl Alcohol's compliance with Dichlorobenzyl Alcohol specifications and serves as a tool for batch-level quality control.
Dichlorobenzyl Alcohol CoA mostly includes findings from lab analyses of a specific batch. For each Dichlorobenzyl Alcohol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Dichlorobenzyl Alcohol may be tested according to a variety of international standards, such as European Pharmacopoeia (Dichlorobenzyl Alcohol EP), Dichlorobenzyl Alcohol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Dichlorobenzyl Alcohol USP).

