
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz

1. (5beta)-3,7,12-trioxocholan-24-oic Acid
2. Atrocholin
3. Cholan Hmb
4. Cholan-hmb
5. Cholanhmb
6. Chologon
7. Decholin
8. Dehydrocholate
9. Dehydrocholate, Sodium
10. Dehydrocholic Acid, Lithium Salt
11. Dehydrocholic Acid, Magnesium Salt
12. Dehydrocholic Acid, Potassium Salt
13. Dehydrocholic Acid, Sodium Salt
14. Ketocholanic Acid
15. Sodium Dehydrocholate
16. Triketocholanic Acid
17. Trioxocholate
1. 81-23-2
2. Decholin
3. Dehystolin
4. Felacrinos
5. Sanocholen
6. Dilabil
7. Ketocholanic Acid
8. Chologon
9. Oxycholin
10. Procholon
11. Bilidren
12. Bilostat
13. Cholagon
14. Cholimed
15. Dehychol
16. Didrocolo
17. Drenobyl
18. Novocolin
19. Acolen
20. Dehycon
21. Didocol
22. Erebile
23. Hykolex
24. Triketocholanic Acid
25. Deidrocolico Vita
26. Biochol
27. Dehycol
28. Ketochol
29. Khologon
30. Dehydrocholate
31. Doxycholpotassium
32. Cholan Dh
33. Cholic Acid, Dehydro-
34. Dee-co
35. 3,7,12-trioxocholanic Acid
36. 3,7,12-triketocholanic Acid
37. 3,7,12-trioxo-5beta-cholanic Acid
38. Dehydrocholsaeure
39. 3,7,12-triketo-5beta-cholanoic Acid
40. Acide Dehydrocholique
41. 3,7,12-trioxo-5beta-cholan-24-oic Acid
42. Hydrochol
43. (4r)-4-[(5s,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-3,7,12-trioxo-1,2,4,5,6,8,9,11,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-17-yl]pentanoic Acid
44. (5beta)-3,7,12-trioxocholan-24-oic Acid
45. Nsc-8796
46. Cholan-24-oic Acid, 3,7,12-trioxo-, (5.beta.)-
47. Mls000069501
48. 81-23-2 (acid)
49. Chebi:31459
50. Cholan-24-oic Acid, 3,7,12-trioxo-, (5b)-
51. Nh5000009i
52. (r)-4-((5s,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-3,7,12-trioxohexadecahydro-1h-cyclopenta[a]phenanthren-17-yl)pentanoic Acid
53. Smr000058603
54. Dsstox_cid_2888
55. 3,7,12-trioxo-5.beta.-cholan-24-oic Acid
56. Dsstox_rid_76775
57. Dsstox_gsid_22888
58. Atrocholin
59. Cholepatin
60. Dehydrocholsaeure [german]
61. Acido Deidrocolico
62. Acido Deidrocolico [dcit]
63. Acido Dehidrocolico
64. Acido Dehidrocolico [spanish]
65. Acide Dehydrocholique [french]
66. Acidum Dehydrocholicum [latin]
67. Acidum Dehydrocholicum
68. Acido Dehidrocolico [inn-spanish]
69. Acide Dehydrocholique [inn-french]
70. Acidum Dehydrocholicum [inn-latin]
71. Nsc 8796
72. Einecs 201-335-7
73. Brn 3226734
74. Cholan-24-oic Acid, 3,7,12-trioxo-, (5beta)-
75. 5beta-cholanic Acid, 3,7,12-trioxo-
76. Dilahil
77. Dehydocholic Acid
78. 3,7,12-trioxo-5-beta-cholan-24-oic Acid
79. Unii-nh5000009i
80. Dehydro-ca
81. Dehydrocholate-acid
82. Ncgc00095777-01
83. Decholin (tn)
84. Prestwick_498
85. Cholan-24-oic Acid, 3,7,12-trioxo-, (5-beta)-
86. Mfcd00066410
87. Dehydrocholic Acid [usp:inn:ban:jan]
88. 3,7,12-trioxo-5beta-cholan-24-oicacid
89. Opera_id_1391
90. Prestwick3_000123
91. 3,12-trioxocholanic Acid
92. 5beta-cholan-24-oic Acid, 3,7,12-trioxo-
93. 3,12-triketocholanic Acid
94. 5-beta-cholan-24-oic Acid, 3,7,12-trioxo-
95. Schembl26391
96. Bspbio_000166
97. 4-10-00-03478 (beilstein Handbook Reference)
98. Mls001076501
99. Mls001424181
100. Bpbio1_000184
101. Chembl514446
102. Dehydrocholic Acid [mi]
103. Dehydrocholic Acid [inn]
104. Dehydrocholic Acid [jan]
105. Dtxsid2022888
106. Nsc8796
107. 3,7,12-trioxo-5-cholanic Acid
108. Dehydrocholic Acid [vandf]
109. Hms2052a11
110. Hms2095i08
111. Hms2235j07
112. Hms3712i08
113. Dehydrocholic Acid [mart.]
114. Dehydrocholic Acid [usp-rs]
115. Dehydrocholic Acid [who-dd]
116. Hy-b1393
117. Zinc3860869
118. 3,7,12-trioxo-24-cholanic Acid
119. Purified Dehydrocholic Acid (jp17)
120. Tox21_111519
121. Lmst04010106
122. Pdsp2_000076
123. S4562
124. 3,12-trioxo-5.beta.-cholanic Acid
125. Dehydrocholic Acid (jp17/usp/inn)
126. Dehydrocholic Acid, >=99.0% (t)
127. 3,12-triketo-5.beta.-cholanic Acid
128. Akos024284359
129. Tox21_111519_1
130. 3,12-triketo-5.beta.-cholanoic Acid
131. 5.beta.-cholanic Acid,7,12-trioxo-
132. Ccg-101103
133. Cs-4902
134. Db11622
135. Nc00353
136. 3,7,12-trioxo-5.beta.-cholanic Acid
137. 3,7,12-trioxocholan-24-oic Acid #
138. 3,7,12-triketo-5.beta.-cholanic Acid
139. Ncgc00024025-03
140. Ncgc00024025-05
141. 3,7,12-triketo-5.beta.-cholanoic Acid
142. 4-((1s,2s,7s,11s,10r,14r,15r)-2,15-dimethyl-5,9,16-trioxotetracyclo[8.7.0.0<2, 7>.0<11,15>]heptadec-14-yl)pentanoic Acid
143. Dehydrocholic Acid [usp Monograph]
144. 5.beta.-cholanic Acid, 3,7,12-trioxo-
145. 3,12-trioxo-5.beta.-cholan-24-oic Acid
146. 5.beta.-cholan-24-oic Acid,7,12-trioxo-
147. D0042
148. D01693
149. D94651
150. 3,7,12-tri-keto-5.beta.-cholan-24-oic Acid
151. 5.beta.-cholan-24-oic Acid, 3,7,12-trioxo-
152. Cholan-24-oic Acid,7,12-trioxo-, (5.beta.)-
153. Q903387
154. Sr-01000003022
155. Sr-01000003022-3
156. W-104212
157. Brd-k90976994-001-02-1
158. 3,7,12-trioxo-5beta-cholanic Acid 5beta-cholanic Acid-3,5,12-trione
159. Dehydrocholic Acid, United States Pharmacopeia (usp) Reference Standard
160. (r)-4-((5s,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-3,7,12-trioxohexadecahydro-1h-cyclopenta[a]phenanthren-17-yl)pentanoicacid
| Molecular Weight | 402.5 g/mol |
|---|---|
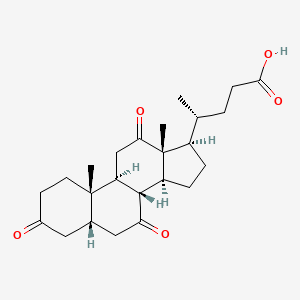
| Molecular Formula | C24H34O5 |
| XLogP3 | 2.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Exact Mass | 402.24062418 g/mol |
| Monoisotopic Mass | 402.24062418 g/mol |
| Topological Polar Surface Area | 88.5 Ų |
| Heavy Atom Count | 29 |
| Formal Charge | 0 |
| Complexity | 756 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 8 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
No approved therapeutic indications.
Following infusion of dehydrocholic acid (DHCA) in rats, the secretions of all the endogenous biliary bile acids were decreased within 30-60 minutes of infusion. Phospholipid secretion as well as cholesterol levels were also declined. The bile flow was increased after administration of dehydrocholic acid.
Cholagogues and Choleretics
Gastrointestinal agents that stimulate the flow of bile into the duodenum (cholagogues) or stimulate the production of bile by the liver (choleretic). (See all compounds classified as Cholagogues and Choleretics.)
Absorption
The duodenal experiment indicates that dehydrocholic acid is absorbed from the proximal small intestine.
Route of Elimination
Administered dehydrocholic acid is excreted rapidly in bile as glycine- and taurine-conjugated bile acids.
Volume of Distribution
No pharmacokinetic data available.
Clearance
No pharmacokinetic data available.
The major site of metabolism is proposed to be the liver. The major metabolite accounting for 70% of total detectable metabolites is dihydroxymonoketo bile acid (3,7-dihydroxy-12-keto-5-cholanoic acid). About 20% of metabolites is monohydroxydiketoacid (3-hydroxy-7,12-keto-5-cholanoic acid) and about 10% is cholic acid.
No pharmacokinetic data available.
It is proposed that dehydrocholic acid induces choleresis, which is associated with biliary lipid secretion and reduced secretion of endogenous and/or exogenous biliary components. Dehydrocholic acid may decrease bile phospholipid secretion due to a lack of micelle formation by dehydrocholic acid-produced bile. A study suggests that due to enhanced permeability of tight junctions in the canalicular membranes, dehydrocholic acid facilitates direct exchange between bile and plasma.
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
96
PharmaCompass offers a list of Dehydrocholic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Dehydrocholic Acid manufacturer or Dehydrocholic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Dehydrocholic Acid manufacturer or Dehydrocholic Acid supplier.
A Dehydrocholic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Dehydrocholic Acid, including repackagers and relabelers. The FDA regulates Dehydrocholic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Dehydrocholic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Dehydrocholic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Dehydrocholic Acid supplier is an individual or a company that provides Dehydrocholic Acid active pharmaceutical ingredient (API) or Dehydrocholic Acid finished formulations upon request. The Dehydrocholic Acid suppliers may include Dehydrocholic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Dehydrocholic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
Dehydrocholic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Dehydrocholic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Dehydrocholic Acid GMP manufacturer or Dehydrocholic Acid GMP API supplier for your needs.
A Dehydrocholic Acid CoA (Certificate of Analysis) is a formal document that attests to Dehydrocholic Acid's compliance with Dehydrocholic Acid specifications and serves as a tool for batch-level quality control.
Dehydrocholic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Dehydrocholic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Dehydrocholic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Dehydrocholic Acid EP), Dehydrocholic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Dehydrocholic Acid USP).

