
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Canada
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
Regulatory FDF Prices
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
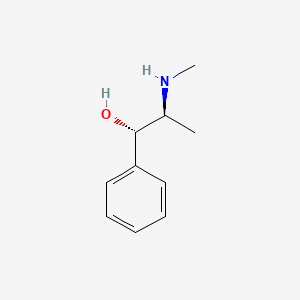

1. Ephedrine Threo Isomer
2. Isoephedrine
3. Pseudoephedrine Hcl
4. Pseudoephedrine Hydrochloride
5. Sudafed
6. Threo Isomer Of Ephedrine
1. (+)-pseudoephedrine
2. D-pseudoephedrine
3. Isoephedrine
4. Trans-ephedrine
5. D-isoephedrine
6. Psi-ephedrin
7. Sudafed
8. Psi-ephedrine
9. (+)-threo-ephedrine
10. D-psi-ephedrine
11. 90-82-4
12. L(+)-psi-ephedrine
13. L-(+)-pseudoephedrine
14. (+)-psi-ephedrine
15. Pseudoefedrina
16. Pseudoephedrinum
17. Besan
18. (+)-(1s,2s)-pseudoephedrine
19. (1s,2s)-2-(methylamino)-1-phenylpropan-1-ol
20. Pseudoephedrine D-form
21. (1s,2s)-pseudoephedrine
22. (1s,2s)-(+)-pseudoephedrine
23. Neodurasina
24. Acunaso
25. D-psi-2-methylamino-1-phenyl-1-propanol
26. Pseudoephedrine, (+)-
27. Isoephedrine, D-
28. Pseudoephedrine (inn)
29. 7cuc9ddi9f
30. Chebi:51209
31. Pseudoephedrine, L-(+)-
32. Alpha-(1-(methylamino)ethyl)benzyl Alcohol
33. Pseudoephedrine (d)
34. Benzenemethanol, Alpha-((1s)-1-(methylamino)ethyl)-, (alpha-s)-
35. Benzenemethanol, .alpha.-[(1s)-1-(methylamino)ethyl]-, (.alpha.s)-
36. Pseudoefedrina [inn-spanish]
37. Pseudoephedrinum [inn-latin]
38. (+) Threo-2-(methylamino)-1-phenyl-1-propanol
39. Pseudoephedrine [inn]
40. Pseudoephedrine [inn:ban]
41. Pseudoephedrine Ephedrine
42. Psi-ephedrine, (+)-
43. 2-(methylamino)-1-phenyl-1-propanol
44. Neodurasina (tn)
45. Acunaso (tn)
46. Pseudophedrine Sulphate
47. Hsdb 3177
48. Pseudoephedrine Polistirex
49. Einecs 202-018-6
50. Unii-7cuc9ddi9f
51. Benzenemethanol, .alpha.-((1s)-1-(methylamino)ethyl)-, (.alpha.s)-
52. (i)-ephedrin
53. Ephedrine-(racemic)
54. (+) Pseudoephedrine
55. D-pseudoephedrine Base
56. Spectrum_000878
57. Benzenemethanol, Alpha-(1-(methylamino)ethyl)-, (s-(r*,r*))-
58. Spectrum2_001303
59. Spectrum3_001771
60. Spectrum4_001162
61. Spectrum5_000650
62. Lopac-e-3250
63. (1s,2s) Pseudoephedrine
64. (1s,2s)-2-methylamino-1-phenylpropan-1-ol
65. (1s, 2s) Pseudoephedrine
66. Ec 202-018-6
67. Schembl4368
68. Chembl1590
69. Pseudoephedrine [mi]
70. (1s,2s)-2-(methylamino)-1-phenyl-propan-1-ol
71. Bspbio_003261
72. Kbiogr_001763
73. Kbioss_001358
74. Bidd:gt0817
75. Divk1c_000451
76. Pseudoephedrine [hsdb]
77. Spbio_001365
78. Pseudoephedrine [vandf]
79. Gtpl7286
80. Pseudoephedrine [mart.]
81. Benzenemethanol, .alpha.-(1-(methylamino)ethyl)-, (s-(r*,r*))-
82. Dtxsid0023537
83. Pseudoephedrine [who-dd]
84. Kbio1_000451
85. Kbio2_001358
86. Kbio2_003926
87. Kbio2_006494
88. Kbio3_002762
89. Zinc20259
90. (1s, 2s)-(+)-pseudoephedrine
91. Ninds_000451
92. Benzenemethanol, Alpha-((1s)-1-(methylamino)ethyl)-, (alphas)-
93. Pdsp1_001347
94. Akos025401512
95. Db00852
96. Idi1_000451
97. (1s,2s)-(+)-pseudoephedrine, 98%
98. Ncgc00015408-01
99. Ncgc00178180-01
100. Nci60_002955
101. Sbi-0051498.p003
102. 1-(s),2-(r)-erythro-(+)-ephedrine
103. C02765
104. D08449
105. 064p256
106. Q263958
107. Brd-k84175871-003-02-2
108. Ephedrine Hydrochloride Impurity B [ep Impurity]
109. (.alpha.s)-.alpha.-((1r)-1-(methylamino)ethyl)benzenemethanol
110. (s,s)-(+)-pseudoephedrine Solution, 1.0 Mg/ml In Methanol, Ampule Of 1 Ml, Certified Reference Material
| Molecular Weight | 165.23 g/mol |
|---|---|
| Molecular Formula | C10H15NO |
| XLogP3 | 0.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 3 |
| Exact Mass | 165.115364102 g/mol |
| Monoisotopic Mass | 165.115364102 g/mol |
| Topological Polar Surface Area | 32.3 Ų |
| Heavy Atom Count | 12 |
| Formal Charge | 0 |
| Complexity | 121 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Afrinol |
| Active Ingredient | Pseudoephedrine sulfate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | 120mg |
| Market Status | Over the Counter |
| Company | Schering Plough |
| 2 of 2 | |
|---|---|
| Drug Name | Afrinol |
| Active Ingredient | Pseudoephedrine sulfate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | 120mg |
| Market Status | Over the Counter |
| Company | Schering Plough |
The Combat Methamphetamine Epidemic Act of 2005 has been incorporated into the Patriot Act signed by President Bush on March 9, 2006. The act bans over-the-counter sales of cold medicines that contain the ingredient pseudoephedrine, which is commonly used to make methamphetamine. The sale of cold medicine containing pseudoephedrine is limited to behind the counter. The amount of pseudoephedrine that an individual can purchase each month is limited and individuals are required to present photo identification to purchase products containing pseudoephedrine. In addition, stores are required to keep personal information about purchasers for at least two years.
FDA, Center for Drug Evaluation and Research (CDER); Legal Requirements for the Sale and Purchase of Drug Products Containing Pseudoephedrine, Ephedrine, and Phenylpropanolamine (May 8, 2006). Available from, as of March 12, 2007: https://www.fda.gov/cder/news/methamphetamine.htm
Pseudoephedrine is indicated for temporary relief of congestion associated with acute coryza, acute eustachian salpingitis, serous otitis media with eustachian tube congestion, vasomotor rhinitis, and aerotitis (barotitis) media. Pseudoephedrine also be indicated as an adjunct to analgesics, antihistamines, antibiotics, antitussive, or expectorants for optimum results in allergic rhinitis, croup, acute and subacute sinusitis, acute otitis medica and acute tracheobronchitis. /Included in US product labeling/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 2452
Although pseudoephedrine has been used in the symptomatic treatment of bronchial asthma and reversible bronchospasm, the drug appear to be ineffective as a bronchodilator.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 1354
/This study assessed whether/ pseudoephedrine hydrochloride is effective in decreasing the risk for earache ... in chlidren traveling by air. ... Children aged 6 months to 6 years were included in this study. Pseudoephedrine hydrochloride (1 mg/kg body weight) or placebo was administered 30 to 60 minutes prior to departure on commercial air flights. Caregivers noted historical details and the degree of apparent ear pain, drowsiness, and excitability with ascent and descent. Ninety-one flights involving 50 children were studied, with ear pain being reported in 13 (14%) of flights. Ear pain was not associated with a history of air travel-associated ear pain, recent ear infection, or recent upper airway symptoms. Pseudoephedrine use was not associated with a decrease in ear pain during either ascent (4% with pseudoephedrine vs 5% with placebo; P approximately 1.00) or descent (12% with pseudoephedrine vs. 13% with placebo; P approximately 1.00). Pseudoephedrine use was, however, linked to drowsiness at takeoff (60% with pseudoephedrine vs. 27% with placebo; P = .003) but not at landing (P = .39). Treatment was not associated with excitability at takeoff (P = .09) or landing (P approximately 1.00). Ear pain is not uncommon in children traveling by commercial aircraft. The predeparture use of pseudoephedrine does not decrease the risk for in-flight ear pain in children but is associated with drowsiness. /Pseudoephedrine Hydrochloride/
PMID:10323625 Buchanan BJ et al; Arch Pediatr Adolesc Med 153 (5): 466-8 (1999)
For more Therapeutic Uses (Complete) data for PSEUDOEPHEDRINE (6 total), please visit the HSDB record page.
Maternal Medication usually Compatible with Breast-Feeding: Pseudoephedrine: Reported Sign or Symptom in Infant or Effect on Lactation: None. /from Table 6/
Report of the American Academy of Pediatrics Committee on Drugs in Pediatrics 93 (1): 141 (1994)
Pseudoephedrine may cause mild CNS stimulation, especially in patients who are hypersensitive to the effects of sympathomimetic drugs. Nervousness, excitability, restlessness, dizziness, weakness, and insomnia may occur. Headache and drowsiness also have been reported.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 1355
As with other sympathomimetic drugs, pseudoephedrine should not be used for self-medication in patients with hyperthyroidism, diabetes mellitus, hypertension, ischemic heart disease, or difficulty urinating secondary to prostatic hypertrophy without consulting a clinician. The drug should be used cautiously, and may be contraindicated, in patients who are hypersensitive to the effects of other sympathomimetic drugs. Pseudoephedrine is contraindicated in patients with severe hypertension, severe coronary artery disease, angle-closure glaucoma, or urinary retention.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 1355
Geriatric patients may be especially sensitive to the effects of sympathomimetic amines. Overdosage of sympathomimetic amines in patients older than 60 years of age may cause hallucinations, CNS depression, seizures, and death. Extended-release preparations containing pseudoephedrine therefore should not be administered to these patients until safety has been established by administration of a short-acting preparation.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 1355
For more Drug Warnings (Complete) data for PSEUDOEPHEDRINE (22 total), please visit the HSDB record page.
Pseudoephedrine is a sympathomimetic amine used for its decongestant activity.
Pseudoephedrine causes vasoconstriction which leads to a decongestant effect. It has a short duration of action unless formulated as an extended release product. Patients should be counselled regarding the risk of central nervous system stimulation.
Nasal Decongestants
Drugs designed to treat inflammation of the nasal passages, generally the result of an infection (more often than not the common cold) or an allergy related condition, e.g., hay fever. The inflammation involves swelling of the mucous membrane that lines the nasal passages and results in inordinate mucus production. The primary class of nasal decongestants are vasoconstrictor agents. (From PharmAssist, The Family Guide to Health and Medicine, 1993) (See all compounds classified as Nasal Decongestants.)
Bronchodilator Agents
Agents that cause an increase in the expansion of a bronchus or bronchial tubes. (See all compounds classified as Bronchodilator Agents.)
R01BA02
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
R - Respiratory system
R01 - Nasal preparations
R01B - Nasal decongestants for systemic use
R01BA - Sympathomimetics
R01BA02 - Pseudoephedrine
Absorption
A 240mg oral dose of pseudoephedrine reaches a Cmax of 246.310.5ng/mL fed and 272.513.4ng/mL fasted, with a Tmax of 6.601.38h fed and 11.870.72h fasted, with an AUC of 6862.0334.1ng\*h/mL fed and 7535.1333.0ng\*h/mL fasted.
Route of Elimination
55-75% of an oral dose is detected in the urine as unchanged pseudoephedrine.
Volume of Distribution
The apparent volume of distribution of pseudoephedrin is 2.6-3.3L/kg.
Clearance
A 60mg oral dose of pseudoephedrine has a clearance of 5.91.7mL/min/kg.
Elimination: Renal. About 55 to 75% of a dose is excreted unchange. The rate of excretion is accelerated in acidic urine.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 2452
Oral capsules containing 100 mg pseudoephedrine hydrochloride and different grades of sodium alginate were prepared and studied to determine drug dissolution, the penetration of dissolution medium into the capsules, and the bioavailability of the formulations in 16 healthy subjects (ages 19-24 yr) using a randomized crossover design. At pH 1.2, there was no difference in dissolution between the formulations. At pH 7.2, release rates correlated with the viscosity grade of sodium alginate. Bioavailability tests confirmed that capsules containing high viscosity grades of alginate led to slow release, with a time to reach peak concn of 6.5 hr, and no loss of amount of drug absorbed. It was concluded that controlled release pseudoephedrine capsules can be prepared with high viscosity grades of sodium alginate. /Pseudoephedrine hydrochloride/
Veski P et al; Int J Pharm 111: 171-79 (1994)
The release of pseudoephedrine hydrochloride from beadlets coated with different levels of Eudragit S-100 and Eudragit RS was studied. In vitro dissolution studies indicated that the coating levels types of resins and types of plasticizers in the film played a major role in determining drug release rate profiles. Drug release appeared to follow first order kinetics. A coating of 2% Eudragit S-100 eliminated the formation of soft lumps which are commonly seen with Eudragit RS coated beadlets. Eudragit S-100 coating also contributed to retarding drug release in dissolution media at pH 1.2-6.5. It was concluded that pseudoephedrine release from coated beadlets depended on coating levels types of resins and types of plasticizers used. /Pseudoephedrine hydrochloride/
Li SP et al; Drug Dev Ind Pharm 17 (12): 1655-83 (1991)
Only three drugs are commonly used as oral decongestants: phenylpropanolamine (PPA), pseudoephedrine (PDE), and phenylephrine (PE). They are all chiral drugs that exist as stereoisomers. It is possible that each enantiomer can reflect significant enantioselective differences with regard to both pharmacokinetic and pharmacodynamic effects. Both PPA and PDE are readily and completely absorbed, whereas PE, with a bioavailability of only approximately 38%, is subject to gut wall metabolism and is thought to be absorbed erratically. Peak concn are reached between 0.5 and 2 hr after admin. All three drugs are extensively distributed into extravascular sites (apparent volume of distribution between 2.6 and 5.0 L/kg). No protein binding data in humans are available. Whereas PPA and PDE are not substantially metabolized, PE undergoes extensive biotransformation in the gut wall and the liver. Elimination of PPA and PDE is predominantly renal, with urinary excretion being pH dependent. Half-lives are relatively short, approximately 2.5 hr for PE, 4 hr for PPA, and 6 hr for PDE. Elimination of PPA and PDE may be rapid in children, and the agents should be used with caution in patients with renal impairment. In addition, PPA incr caffeine plasma levels and decr theophylline clearance. Reduced metabolism of PE occurs with concurrent admin of monoamine oxidase inhibitors. No direct relationship between nasal decongestant effect and plasma concentration has been established.
Kanfer I et al; 13 (6 Pt 2): 116S-28S (1993)
For more Absorption, Distribution and Excretion (Complete) data for PSEUDOEPHEDRINE (13 total), please visit the HSDB record page.
Pseudoephedrine is <1% N-demethylated to an inactive metabolite. The majority of pseudoephedrine is eliminated unmetabolized in the urine.
Pseudoephedrine is incompletely metabolized (less than 1%) in the liver by N-demethylation to an inactive metabolite. The drug and its metabolite are excreted in urine; 55-96% of a dose is excreted unchanged.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 1356
D-PSEUDOEPHEDRINE YIELDS D-NORPSEUDOEPHEDRINE & L-PSEUDOEPHEDRINE YIELDS L-NORPSEUDOEPHEDRINE IN RABBIT: DANN, RE ET AL, EUR J PHARMAC, 16, 233 (1971). /FROM TABLE/
Goodwin, B.L. Handbook of Intermediary Metabolism of Aromatic Compounds. New York: Wiley, 1976., p. 43
Ephedrine (EPH), pseudoephedrine (PEPH), phenylpropanolamine (PPA), methylephedrine (MEPH) and cathine are sympathomimetic amines. ... Excretion studies of the ephedrine-related drugs have been performed to better understand the metabolic yields of ephedrines in urine. After consuming a single clinical dose of each of these drugs, urine samples from volunteers (n=3 for each drug) were subjected to tert-butyl-methyl-ether (TBME) extraction and trifluoroacetic acid (TFAA) derivatization before gas chromatography-mass spectrometry (GC-MS) analysis. Most ephedrines were excreted unchanged in urine, including EPH (40.9%), PEPH (72.2%), and PPA (59.3%). However, only a relatively small amount of MEPH (15.5%) was excreted unchanged in urine. In addition, a trace amount of PPA (1.6%) and cathine (0.7%) was found to be the metabolites of EPH and PEPH, respectively. Urinary EPH, PEPH, and PPA reached peaks at 2-6 hr and disappeared in urine at approximately 24-48 hr post-administration. ...
PMID:15885945 Tseng YL et al; Forensic Sci Int 157 (2-3): 149-55 (2006)
The mean elimination half life of pseudoephedrine is 6.0h.
In two sequential double-blind, parallel-group, single-dose studies, 21 children received either pseudoephedrine, 30 or 60 mg, or placebo, and 20 children received either phenylpropanolamine, 20 or 37.5 mg, or placebo. Before dosing and at intervals up to 7 hours after dosing, serum pseudoephedrine or phenylpropanolamine concentrations were measured, and pulse and blood pressure were recorded. In two children receiving each drug, these tests were also performed at 12 and 24 hours, and urine was collected from 0 to 12 and from 12 to 24 hours. In children, the mean (+/-SEM) terminal elimination half-life values for pseudoephedrine, 3.1 +/- 0.5 hours, and for phenylpropanolamine, 2.6 +/- 0.6 hours, were significantly shorter than those found by other investigators in adults. Pharmacokinetics were not dose dependent in the dose ranges studied.
PMID:8917241 Simons FE et al; J Pediatr 129 (5): 729-34 (1996)
Urinary pH can affect the elimination half-life of pseudoephedrine, prolonging it when alkaline (pH 8) and reducing it when acidic (pH 5). The elimination half-life of pseudoephedrine ranges from 3-6 or 9-16 hours when urinary pH is 5 or 8, respectively, while when urinary pH is 5.8, the elimination half-life of the drug ranges from 5-8 hours. In one study in children 6-12 years of age, the elimination half-life of pseudoephedrine averaged about 3 hours when urinary pH was 6.5.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 1356
Pseudoephedrine acts mainly as an agonist of alpha adrenergic receptors and less strongly as an agonist of beta adrenergic receptors. This agonism of adrenergic receptors produces vasoconstriction which is used as a decongestant and as a treatment of priapism. Pseudoephedrine is also an inhibitor of norepinephrine, dopamine, and serotonin transporters. The sympathomimetic effects of pseudoephedrine include an increase in mean arterial pressure, heart rate, and chronotropic response of the right atria. Pseudoephedrine is also a partial agonist of the anococcygeal muscle. Pseudoephedrine also inhibits NF-kappa-B, NFAT, and AP-1.
THESE AGENTS /BRONCHODILATORS/ ACT ON BETA-2 RECEPTORS TO RELAX BRONCHIAL SMOOTH MUSCLE & PERIPHERAL VASCULATURE, APPARENTLY BY STIMULATING PRODN OF CYCLIC ADENOSINE-3.5-MONOPHOSPHATE (CAMP)...
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 627
Pseudoephedrine acts on alpha-adrenergic receptors in the mucosa of the respiratory tract, producing vasoconstriction. The medication shrinks swollen nasal mucous membranes; reduces tissue hyperemia, edema, and nasal congestion; and increases nasal airway patency. Also, drainage of sinus secretions may be increased and obstructed eustachian ostia may be opened.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 2452
The pharmacological properties of the ephedrine derivative pseudoephedrine were investigated at the nuclear level. Following intraperitoneal injection of Sprague Dawley rats with pseudoephedrine, Fos induction was measured in various brain areas by Western blots and immunocytochemistry. Pseudoephedrine induced Fos-like immunoreactivity in the nucleus accumbens and striatum in a time and concentration-dependent manner with maximal effect at 60 mg/kg 2 hr after injection. Immunocytochemical studies confirmed that the majority of the signal was detectable in the nucleus accumbens and striatum. Pre-injection with the D1 dopamine receptor antagonist SCH23390 partially and completely blocked pseudoephedrine-induced Fos-like immunoreactivity in the striatum and nucleus accumbens, respectively, suggesting that the action of pseudoephedrine is mediated via dopamine release and results in the activation of D1 dopamine receptors. With the exception of the higher doses required, the actions of pseudoephedrine were similar to those previously described for the psychostimulant amphetamine.
PMID:10471092 Kumarnsit E et al; Neuropharmacology 38 (9): 1381-7 (1999)
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

ANALYTICAL

ABOUT THIS PAGE
42
PharmaCompass offers a list of Pseudoephedrine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Pseudoephedrine manufacturer or Pseudoephedrine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Pseudoephedrine manufacturer or Pseudoephedrine supplier.
A Decofed manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Decofed, including repackagers and relabelers. The FDA regulates Decofed manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Decofed API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Decofed manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Decofed supplier is an individual or a company that provides Decofed active pharmaceutical ingredient (API) or Decofed finished formulations upon request. The Decofed suppliers may include Decofed API manufacturers, exporters, distributors and traders.
click here to find a list of Decofed suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Decofed DMF (Drug Master File) is a document detailing the whole manufacturing process of Decofed active pharmaceutical ingredient (API) in detail. Different forms of Decofed DMFs exist exist since differing nations have different regulations, such as Decofed USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Decofed DMF submitted to regulatory agencies in the US is known as a USDMF. Decofed USDMF includes data on Decofed's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Decofed USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Decofed suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Decofed as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Decofed API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Decofed as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Decofed and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Decofed NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Decofed suppliers with NDC on PharmaCompass.
Decofed Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Decofed GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Decofed GMP manufacturer or Decofed GMP API supplier for your needs.
A Decofed CoA (Certificate of Analysis) is a formal document that attests to Decofed's compliance with Decofed specifications and serves as a tool for batch-level quality control.
Decofed CoA mostly includes findings from lab analyses of a specific batch. For each Decofed CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Decofed may be tested according to a variety of international standards, such as European Pharmacopoeia (Decofed EP), Decofed JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Decofed USP).

