
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
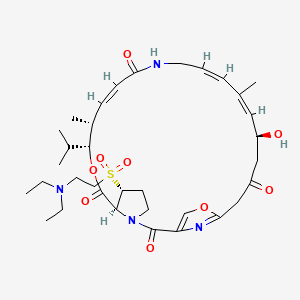

1. 26-(2-diethylaminoethyl)sulfonylpristamycin Iib
2. Rp 54476
3. Rp-54476
1. Dalfopristine
2. Dalfopristina
3. Dalfopristinum
4. Rp 54476
5. 112362-50-2
6. Rp-54476
7. R9m4fje48e
8. 5-(2-diethylamino-ethanesulfonyl)-21-hydroxy-10-isopropyl-11,19-dimethyl-9,26-dioxa-3,15,28-triaza-tricyclo[23.2.1.00,255]octacosa-1(27),12,17,19,25(28)-pentaene-2,8,14,23-tetraone
9. Rp54476
10. Dalfopristine [inn-french]
11. Dalfopristinum [inn-latin]
12. Dalfopristina [inn-spanish]
13. (3r,4r,5e,10e,12e,14s,26r,26as)-26-((2-(diethylamino)ethyl)sulfonyl)-8,9,14,15,24,25,26,26a-octahydro-14-hydroxy-3-isopropyl-4,12-dimethyl-3h-21,18-nitrilo-1h,22h-pyrrolo(2,1-c)(1,8,4,19)dioxadiazacyclotetracosine-1,7,16,22(4h,17h)-tetrone
14. Virginiamycin M1, 26-((2-(diethylamino)ethyl)sulfonyl)-26,27-dihydro-, (26r,27s)-
15. Unii-r9m4fje48e
16. Dalfopristin [usan:inn:ban]
17. Hsdb 8068
18. Dol
19. Dalfopristin [mi]
20. Dalfopristin [inn]
21. Dalfopristin [jan]
22. Dalfopristin [usan]
23. Dalfopristin [vandf]
24. Schembl40993
25. Dalfopristin [who-dd]
26. Chebi:4309
27. Chembl1200937
28. Gtpl10797
29. Dtxsid10869549
30. Dalfopristin [orange Book]
31. (6r,7s,10r,11r,12e,17e,19e,21s)-6-[2-(diethylamino)ethylsulfonyl]-21-hydroxy-11,19-dimethyl-10-propan-2-yl-9,26-dioxa-3,15,28-triazatricyclo[23.2.1.03,7]octacosa-1(27),12,17,19,25(28)-pentaene-2,8,14,23-tetrone
32. Zinc3917540
33. Synercid Component Dalfopristin
34. Db01764
35. Dalfopristin Component Of Synercid
36. Ncgc00508820-02
37. Q5210829
38. 2-diethylaminoethylsulfonyl-hydroxy-isopropyl-dimethyl-[?]tetrone
39. (3r,4r,5e,10e,12e,14s,26r,26as)-26-{[2-(diethylamino)ethyl]sulfonyl}-14-hydroxy-4,12-dimethyl-3-(1-methylethyl)-8,9,14,15,24,25,26,26a-octahydro-1h,3h,22h-21,18-(azeno)pyrrolo[2,1-c][1,8,4,19]dioxadiazacyclotetracosine-1,7,16,22(4h,17h)-tetrone
40. (3r,4r,5e,10e,12e,14s,26r,26as)-26-{[2-(diethylamino)ethyl]sulfonyl}-14-hydroxy-4,12-dimethyl-3-(propan-2-yl)-8,9,14,15,24,25,26,26a-octahydro-1h,3h,22h-21,18-(azeno)pyrrolo[2,1-c][1,8,4,19]dioxadiazacyclotetracosine-1,7,16,22(4h,17h)-tetrone
| Molecular Weight | 690.8 g/mol |
|---|---|
| Molecular Formula | C34H50N4O9S |
| XLogP3 | 2.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 7 |
| Exact Mass | 690.32985036 g/mol |
| Monoisotopic Mass | 690.32985036 g/mol |
| Topological Polar Surface Area | 185 Ų |
| Heavy Atom Count | 48 |
| Formal Charge | 0 |
| Complexity | 1340 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 3 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Bacterial Agents
National Library of Medicine's Medical Subject Headings online file (MeSH, 2012)
Quinupristin and dalfopristin is used IV in adults for the treatment of serious or life-threatening infections caused by susceptible strains of vancomycin-resistant Enterococcus faecium (VREF), including infections associated with VREF bacteremia. Quinupristin and dalfopristin became commercially available in the US for this indication under the principles and procedures of FDA's accelerated review process that allows approval based on analysis of surrogate markers of response (i.e., clearance of bacteremia), rather than clinical end points such as cure of infection or survival. Controlled clinical studies are underway to confirm the validity of this surrogate marker. /Included in US product labeling/
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012, p. 491
Quinupristin and dalfopristin is used IV for the treatment of complicated skin and skin structure infections caused by Staphylococcus aureus (methicillin-susceptible strains) or Streptococcus pyogenes (group A beta-hemolytic streptococci). /Included in US product labeling/
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012, p. 491
The semi-synthetic streptogramin quinupristin/dalfopristin antibiotic exerts potent bactericidal activity against Staphylococcus aureus. /The researchers/ investigated whether, like other bactericidal antibiotics used at subinhibitory concentrations, quinupristin/dalfopristin enhances release of toxins by Gram-positive cocci. The activity of quinupristin/dalfopristin on exotoxin release by S. aureus was investigated by 2D SDS-PAGE combined with MALDI-TOF/MS analysis and by western blotting. /The researchers/ show that quinupristin/dalfopristin at subinhibitory concentrations reduces the release of S. aureus factors that induce tumour necrosis factor secretion in macrophages. Furthermore, quinupristin/dalfopristin but not linezolid attenuated S. aureus-mediated killing of infected host cells. When added to S. aureus cultures at different stages of bacterial growth, quinupristin/dalfopristin reduced in a dose-dependent manner the release of specific virulence factors (e.g. autolysin, protein A, alpha- and beta-haemolysins, lipases). In contrast, other presumably non-toxic exoproteins remained unchanged. The results of the present study suggest that subinhibitory quinupristin/dalfopristin inhibits virulence factor release by S. aureus, which might be especially helpful for the treatment of S. aureus infections, where both bactericidal as well as anti-toxin activity may be advantageous.
PMID:16895938 Koszczol C et al; J Antimicrob Chemother 58 (3): 564-74 (2006)
Adverse venous effects (e.g., thrombophlebitis, pain) may occur; therefore, flush infusion lines with 5% dextrose injection following completion of peripheral infusions with quinupristin and dalfopristin. Do not flush with sodium chloride injection or heparin solutions because of possible incompatibilities. Recommended measures for moderate-to-severe reactions include increasing the infusion volume, changing infusion sites, or establishing central venous access. Concomitant hydrocortisone or diphenhydramine did not alleviate adverse venous effects during clinical studies.
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012, p. 491
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
US Natl Inst Health; DailyMed. Current Medication Information for SYNERCID (quinupristin and dalfopristin) injection, powder, lyophilized, for solution (November 2010). Available from, as of July 5, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=28f698df-b584-4455-2e94-6efc41313960
Because Clostridium difficile-associated diarrhea and colitis has been reported with quinupristin and dalfopristin, ranging in severity from mild to life-threatening, it should be considered in the differential diagnosis of patients who develop diarrhea during or following therapy with the drug.
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012, p. 491
To determine whether myalgias/arthralgias occurring in cancer patients who receive quinupristin/dalfopristin are associated with biliary tract dysfunction, 56 patients with vancomycin-resistant enterococcal infections who were treated with quinupristin/dalfopristin 7.5 mg/kg every 8 hr for a mean duration of 12 days (range 2-52 days) /were studied/. Liver function tests, including a test for alkaline phosphatase, were performed before, during and after the end of therapy. All patients were followed for 1 month after completion of therapy. Thirty-eight (68%) of the 56 patients responded. Myalgias/arthralgias were the leading adverse events occurring in 20 (36%) of the patients. Patients with myalgias/arthralgias had significantly higher levels of alkaline phosphatase (mean 318.7 IU/L) during the mid-term therapy cycle compared with patients without any joint or muscular pain (mean 216.3 IU/L, P = 0.05). In addition, 3/18 (16.6%) patients with myalgias/arthralgias had more than five-fold the normal levels of alkaline phosphatase, which did not occur in any of the other patients who did not develop myalgias/arthralgias (P = 0.04). All myalgias/arthralgias resolved after the discontinuation of quinupristin/dalfopristin. By univariate analysis, other factors associated with myalgias/arthralgias were relapse of hematological malignancy (P = 0.01), receiving tacrolimus within 1 month prior to treatment (P = 0.04) and receiving methotrexate during antimicrobial therapy (P = 0.05). Myalgias/arthralgias occur frequently in cancer patients receiving quinupristin/dalfopristin and may be associated with biliary tract dysfunction, as measured by alkaline phosphatase or other factors that could lead to intra-hepatic cholestasis, such as relapse of haematological malignancy or treatment with tacrolimus or methotrexate.
PMID:15128724 Raad I et al; J Antimicrob Chemother 53 (6): 1105-8 (2004)
For more Drug Warnings (Complete) data for Dalfopristin (13 total), please visit the HSDB record page.
For the treatment of bacterial infections (usually in combination with quinupristin).
FDA Label
Dalfopristin is a streptogramin antibiotic, derived from pristinamycin IIA.
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
Quinupristin and dalfopristin is distributed into milk in rats ... .
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012, p. 492
The pharmacokinetics of quinupristin/dalfopristin have been studied in rats, monkeys and humans following intravenous infusion of radiolabelled and unlabelled drug. In rats and monkeys quinupristin and dalfopristin undergo rapid elimination from the blood and wide tissue distribution. Nevertheless, they do not penetrate the central nervous system or cross the placenta to any significant degree and they do not appear to be subject to significant body retention following cessation of administration. The blood elimination half-life of quinupristin was approximately 0.6 hr in rats and 0.5 hr in monkeys, and that of dalfopristin was approximately 0.6 hr and 0.2 hr, respectively. Both compounds are primarily eliminated through the bile into the faeces; quinupristin is mainly excreted unchanged whereas dalfopristin is extensively metabolized beforehand. The metabolites include the microbiologically active pristinamycin PIIA for dalfopristin and the microbiologically active glutathione- and cysteine-conjugated derivatives for quinupristin. Quinupristin and dalfopristin appear to be handled in a similar manner by humans. Following intravenous administration both compounds are rapidly cleared from the blood with elimination half-lives of approximately 1 hr for quinupristin and 0.4-0.5 hr for dalfopristin. The pharmacokinetic profile of quinupristin is dose-independent and so is that of dalfopristin and RP 12536 when considered together. Extravascular diffusion of quinupristin/dalfopristin has been assessed in human non-inflammatory interstitial fluid.
PMID:9511077 Bergeron M, Montay G; J Antimicrob Chemother 39 (Suppl A): 129-38 (1997)
Fecal excretion constitutes the main elimination route for both parent drugs and their metabolites (75 to 77% of dose). Urinary excretion accounts for approximately 15% of the quinupristin and 19% of the dalfopristin dose. Preclinical data in rats have demonstrated that approximately 80% of the dose is excreted in the bile and suggest that in man, biliary excretion is probably the principal route for fecal elimination.
US Natl Inst Health; DailyMed. Current Medication Information for SYNERCID (quinupristin and dalfopristin) injection, powder, lyophilized, for solution (November 2010). Available from, as of July 5, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=28f698df-b584-4455-2e94-6efc41313960
Converted to an active non-conjugated metabolite by hydrolysis.
Quinupristin and dalfopristin are converted to several major active metabolites: 2 conjugated (with glutathione and cysteine) metabolites for quinupristin and one nonconjugated (formed by hydrolysis) metabolite for dalfopristin, which also act synergistically with the complementary parent drug. This conversion occurs in vitro by nonenzymatic reactions independent of cytochrome P-450 (CYP) and glutathione transferase enzymes.
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012, p. 492
The elimination half-life is approximately 0.70 hours.
The elimination half-life of quinupristin and dalfopristin is approximately 0.85 and 0.70 hours, respectively.
US Natl Inst Health; DailyMed. Current Medication Information for SYNERCID (quinupristin and dalfopristin) injection, powder, lyophilized, for solution (November 2010). Available from, as of July 5, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=28f698df-b584-4455-2e94-6efc41313960
The pharmacokinetics of quinupristin/dalfopristin have been studied in rats, monkeys and humans following intravenous infusion of radiolabelled and unlabelled drug. ... The blood elimination half-life of quinupristin was approximately 0.6 hr in rats and 0.5 hr in monkeys, and that of dalfopristin was approximately 0.6 hr and 0.2 hr, respectively. ... Following intravenous administration both compounds are rapidly cleared from the blood with elimination half-lives of approximately 1 hr for quinupristin and 0.4-0.5 hr for dalfopristin.
PMID:9511077 Bergeron M, Montay G; J Antimicrob Chemother 39 (Suppl A): 129-38 (1997)
The site of action of dalfopristin is the bacterial ribosome. Dalfopristin has been shown to inhibit the early phase of protein synthesis.
The site of action of quinupristin and dalfopristin is the bacterial ribosome. Dalfopristin has been shown to inhibit the early phase of protein synthesis while quinupristin inhibits the late phase of protein synthesis. Synercid is bactericidal against isolates of methicillin-susceptible and methicillin-resistant staphylococci. The mode of action of Synercid differs from that of other classes of antibacterial agents such as beta-lactams, aminoglycosides, glycopeptides, quinolones, macrolides, lincosamides and tetracyclines. Therefore, there is no cross resistance between Synercid and these agents when tested by the minimum inhibitory concentration (MIC) method.
US Natl Inst Health; DailyMed. Current Medication Information for SYNERCID (quinupristin and dalfopristin) injection, powder, lyophilized, for solution (November 2010). Available from, as of July 5, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=28f698df-b584-4455-2e94-6efc41313960
The unique mechanism of action for quinupristin and dalfopristin is inhibition of the late (peptide chain elongation inhibition) and early (peptidyl transferase inhibition and resultant conformational changes) phases of protein synthesis, respectively, by binding at different sites on the 50S subunit of the bacterial ribosome. Antagonism of beta-lactams, aminoglycosides, glycopeptides, quinolones, macrolides, lincosamides, or tetracyclines has not occurred in vitro.
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012, p. 492
The bacterial ribosome is a primary target of several classes of antibiotics. Investigation of the structure of the ribosomal subunits in complex with different antibiotics can reveal the mode of inhibition of ribosomal protein synthesis. Analysis of the interactions between antibiotics and the ribosome permits investigation of the specific effect of modifications leading to antimicrobial resistances. Streptogramins are unique among the ribosome-targeting antibiotics because they consist of two components, streptogramins A and B, which act synergistically. Each compound alone exhibits a weak bacteriostatic activity, whereas the combination can act bactericidal. The streptogramins A display a prolonged activity that even persists after removal of the drug. However, the mode of activity of the streptogramins has not yet been fully elucidated, despite a plethora of biochemical and structural data. RESULTS: The investigation of the crystal structure of the 50S ribosomal subunit from Deinococcus radiodurans in complex with the clinically relevant streptogramins quinupristin and dalfopristin reveals their unique inhibitory mechanism. Quinupristin, a streptogramin B compound, binds in the ribosomal exit tunnel in a similar manner and position as the macrolides, suggesting a similar inhibitory mechanism, namely blockage of the ribosomal tunnel. Dalfopristin, the corresponding streptogramin A compound, binds close to quinupristin directly within the peptidyl transferase centre affecting both A- and P-site occupation by tRNA molecules. The crystal structure indicates that the synergistic effect derives from direct interaction between both compounds and shared contacts with a single nucleotide, A2062. Upon binding of the streptogramins, the peptidyl transferase centre undergoes a significant conformational transition, which leads to a stable, non-productive orientation of the universally conserved U2585. Mutations of this rRNA base are known to yield dominant lethal phenotypes. It seems, therefore, plausible to conclude that the conformational change within the peptidyl transferase centre is mainly responsible for the bactericidal activity of the streptogramins and the post-antibiotic inhibition of protein synthesis.
PMID:15059283 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC400760 Harms JM et al; BMC Biol 1 (April): 2-4 (2004)
Market Place

ABOUT THIS PAGE
98
PharmaCompass offers a list of Dalfopristin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Dalfopristin manufacturer or Dalfopristin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Dalfopristin manufacturer or Dalfopristin supplier.
A Dalfopristin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Dalfopristin, including repackagers and relabelers. The FDA regulates Dalfopristin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Dalfopristin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Dalfopristin supplier is an individual or a company that provides Dalfopristin active pharmaceutical ingredient (API) or Dalfopristin finished formulations upon request. The Dalfopristin suppliers may include Dalfopristin API manufacturers, exporters, distributors and traders.
Dalfopristin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Dalfopristin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Dalfopristin GMP manufacturer or Dalfopristin GMP API supplier for your needs.
A Dalfopristin CoA (Certificate of Analysis) is a formal document that attests to Dalfopristin's compliance with Dalfopristin specifications and serves as a tool for batch-level quality control.
Dalfopristin CoA mostly includes findings from lab analyses of a specific batch. For each Dalfopristin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Dalfopristin may be tested according to a variety of international standards, such as European Pharmacopoeia (Dalfopristin EP), Dalfopristin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Dalfopristin USP).

