
Synopsis
Synopsis
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
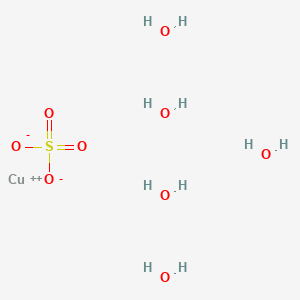

1. Blue Vitriol
2. Copper Sulfate
3. Cupric Sulfate
4. Sulfate, Copper
5. Sulfate, Cupric
6. Vitriol, Blue
1. Copper(ii) Sulfate Pentahydrate
2. 7758-99-8
3. Cupric Sulfate Pentahydrate
4. Blue Vitriol
5. Copper(2+) Sulfate Pentahydrate
6. Copper (ii) Sulfate Pentahydrate
7. Copper(ii) Sulfate, Pentahydrate
8. Copper;sulfate;pentahydrate
9. Copper Sulphate Pentahydrate
10. Cupric Sulfate [usp]
11. Copper(2+) Sulfate (1:1) Pentahydrate
12. Copper Sulfate, Pentahydrate
13. Mfcd00149681
14. Lrx7aj16dt
15. Calcanthite
16. Copper(ii) Sulphate Pentahydrate
17. Vitriol, Blue
18. Sulfuric Acid Copper(2+) Salt (1:1), Pentahydrate
19. Cupric Sulfate (usp)
20. Bluestone
21. Triangle
22. Vencedor
23. Blue Copperas
24. Blue Vicking
25. Salzburg Vitriol
26. Blue Copper As
27. Caswell No. 256
28. Kupfervitriol
29. Kupfervitriol [german]
30. Ccris 5556
31. Hsdb 2968
32. Kupfersulfat-pentahydrat [german]
33. Kupfersulfat-pentahydrat
34. Copper Sulfate (cuso4) Pentahydrate
35. Unii-lrx7aj16dt
36. Cuso4.5h2o
37. Epa Pesticide Chemical Code 024401
38. Sentry Aq Mardel Coppersafe
39. Cuso4(h2o)5
40. Cupric Sulfate (pentahydrate)
41. Sulfuric Acid, Copper(2+) Salt, Pentahydrate
42. Cuprum Sulphuricum
43. Sulfuric Acid, Copper(2+) Salt (1:1), Pentahydrate
44. Liquid Copper Sulfate
45. Cupric Sulfate (tn)
46. Copper Sulfate, Crystal
47. Copper Sulphate(5.h2o)
48. Cupric Sulphate Pentahydrate
49. Copper (as Cupric Sulfate)
50. Copper Sulfate-penta Hydrate
51. Copper Sulfate Fine Crystal
52. Cupric Sulfate [ii]
53. Copper(ii)sulfatepentahydrate
54. Copper(ii)sulfate Pentahydrate
55. Copper Sulfate [vandf]
56. Copper(ii)sulphate Pentahydrate
57. Copper(ii)sulphate-pentahydrate
58. Cupric Sulfate [vandf]
59. Copper(11) Sulfate Pentahydrate
60. Dtxsid9031066
61. Chebi:31440
62. Cuprum Sulphuricum [hpus]
63. Copper (2+) Sulfate Pentahydrate
64. Copper (ii) Sulphate Pentahydrate
65. Copper(2+) Sulfate, Pentahydrate
66. Copper(2+) Sulfate--water (1/5)
67. Cupric Sulfate [orange Book]
68. Copper Sulfate [ep Monograph]
69. Akos025243248
70. Cupric Sulfate [usp Monograph]
71. Cupric Sulfate Pentahydrate [mi]
72. Copper(2+) Sulphate Pentahydrate
73. Copper (as Cupric Sulfate) [vandf]
74. Copper Sulfate Pentahydrate [who-dd]
75. Ft-0624051
76. Copper(ii) Sulfate Pentahydrate, Acs Reagent
77. D03613
78. Copper(2+) Sulphate (1:1) Pentahydrate
79. Copper Sulfate Pentahydrate [ep Monograph]
80. Q6135414
81. Copper(ii) Sulfate Pentahydrate (99.999%-cu) Puratrem
82. Copper(ii) Sulfate Pentahydrate, Trace Metals Grade, 99.995%
83. Sulfuric Acid Copper(2+) Salt (1:1), Hydrate (1:5)
| Molecular Weight | 249.69 g/mol |
|---|---|
| Molecular Formula | CuH10O9S |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 0 |
| Exact Mass | 248.934150 g/mol |
| Monoisotopic Mass | 248.934150 g/mol |
| Topological Polar Surface Area | 93.6 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 62.2 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 7 |
MEDICATION (VET): AS PROPHYLACTIC &/OR THERAPEUTIC FOOT BATH IN FOOT ROT OF CATTLE AND SHEEP; AS FUNGICIDAL AGENT IN THE THERAPY OF RINGWORM & FUNGAL SYNDROMES OF SKIN; AS CAUSTIC IN DESTROYING FISTULOUS TRACTS, NECROTIC INTERDIGITAL MASSES IN FOOT ROT, PROUD FLESH, AND ULCERATIVE STOMATITIS; /HAS BEEN USED/ AS AN EMETIC IN CATS, DOGS, AND PIGS OR TO PPT POISONOUS PHORPHORUS CMPD SUCH AS ZINC PHOSPHIDE; AS HEMATINIC WITH IRON CMPD; AS ASTRINGENT IN ANTIDIARRHEALS PARTICULARLY FOR CALVES OR FEEDLOT CATTLE, TO CONTROL INTESTINAL MONILIASIS & INHIBIT ASCARID GROWTH IN POULTRY; CONTROL TRICHOMONIASIS & HEXAMITIASIS IN TURKEYS; & REFLEXLY CLOSE THE ESOPHAGEAL GROOVE IN SHEEP.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 136
IT MAY RARELY BE USED AS HEMATINIC IN CERTAIN FORMS OF NUTRITIONAL ANEMIAS, ESP IN CHILDREN.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 748
MEDICATION (VET): NUTRITIONAL FACTOR; IN COPPER DEFICIENCY OF RUMINANTS.
The Merck Index. 10th ed. Rahway, New Jersey: Merck Co., Inc., 1983., p. 379
IN UNITED KINGDOM ITS ANTIBACTERIAL EFFECT IS UTILIZED AS APPROVED FEED ADDITIVE FOR IMPROVING RATE OF GAIN & FEED EFFICIENCY OF SWINE (NOT APPROVED IN UNITED STATES BECAUSE OF ENVIRONMENTAL CONCERNS REGARDING BIOCONCENTRATION IN SOIL & SURFACE WATER).
Jones, L.M., et al. Veterinary Pharmacology & Therapeutics. 4th ed. Ames: Iowa State University Press, 1977., p. 885
MEDICATION (VET): IT IS ALSO USED IN TREATMENT OF PARASITIC GASTRITIS IN SHEEP.
Clarke, M. L., D. G. Harvey and D. J. Humphreys. Veterinary Toxicology. 2nd ed. London: Bailliere Tindall, 1981., p. 44
... Its routine use as an emetic is not recommended, because of the potential toxicity of improperly prepared soln and the hazards attending the use of large, corrosive doses.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 748
MAY CAUSE DRAMATIC INCR IN MORTALITY OF TURKEYS GIVEN BLACKHEAD CONTROL DRUGS CONTAINING ARSENIC & EXPOSED TO BLACKHEAD.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 136
CUPRIC ... SULFATE /AS EMETIC/ OFTEN IS EFFECTIVE, BUT POTENTIAL HEMOLYTIC & RENAL TOXICITY IS TOO GREAT TO RECOMMEND USE. ...
American Medical Association, AMA Department of Drugs. AMA Drug Evaluations. 4th ed. Chicago: American Medical Association, 1980., p. 1436
Antidotes
Agents counteracting or neutralizing the action of POISONS. (See all compounds classified as Antidotes.)
Emetics
Agents that cause vomiting. They may act directly on the gastrointestinal tract, bringing about emesis through local irritant effects, or indirectly, through their effects on the chemoreceptor trigger zone in the postremal area near the medulla. (See all compounds classified as Emetics.)
AFTER ADMIN COPPER(II) SULFATE, PENTAHYDRATE FOR 7 DAYS IP, COPPER ACCUMULATED IN MITOCHONDRIAL & LYSOSOMAL FRACTIONS OF RAT LIVER, MAINLY IN LYSOSOMES.
FELDMAN G ET AL; BIOL GASTRO-ENTEROL 5 (1): 37 (1972)
HUMAN RBC INCUBATED WITH COPPER SULFATE WERE TESTED FOR OSMOTIC FRAGILITY, DEFORMABILITY, & ELECTROPHORETIC PROPERTIES OF MEMBRANE PROTEINS. COPPER TREATMENT OXIDIZED THE -SH OF THE MEMBRANE PROTEINS TO FORM INTERMOLECULAR DISULFIDE BONDS, CAUSING A REDUCTION IN MEMBRANE FLEXIBILITY. /COPPER SULFATE/
NAKASHIMA K ET AL; BIOMED RES 1 (6): 548-551 (1981)
COPPER SULFATE INJECTED IP AT 2 MG COPPER/KG INTO VITAMIN E & SELENIUM DEFICIENT RATS CAUSED A 6 FOLD INCREASE IN THE FORMATION OF THE LIPID PEROXIDATION PRODUCT ETHANE, & CAUSED ACUTE MORTALITY IN 4/5 RATS. SELENIUM SUPPLEMENTATION OF THE DIET AT 0.5 PPM SELENIUM PREVENTED THE INCREASE IN ETHANE PRODUCTION CAUSED BY COPPER INJECTION & REDUCED MORTALITY TO 1/5 RATS. THE CORRELATION OF INCREASED PRODUCTION OF ETHANE WITH INCREASED MORTALITY SUGGESTED THAT LIPID PEROXIDATION MAY BE IMPORTANT IN THE INCREASED TOXICITY OF COPPER IN VITAMIN E & SELENIUM DEFICIENT RATS. /COPPER SULFATE/
PMID:7063501 DOUGHERTY JJ, HOEKSTRA WG; PROC SOC EXP BIOL MED 169 (2): 201-8 (1982)
Market Place

ABOUT THIS PAGE
83
PharmaCompass offers a list of Copper Sulphate Pentahydrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Copper Sulphate Pentahydrate manufacturer or Copper Sulphate Pentahydrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Copper Sulphate Pentahydrate manufacturer or Copper Sulphate Pentahydrate supplier.
A Copper Sulphate Pentahydrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Copper Sulphate Pentahydrate, including repackagers and relabelers. The FDA regulates Copper Sulphate Pentahydrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Copper Sulphate Pentahydrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Copper Sulphate Pentahydrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Copper Sulphate Pentahydrate supplier is an individual or a company that provides Copper Sulphate Pentahydrate active pharmaceutical ingredient (API) or Copper Sulphate Pentahydrate finished formulations upon request. The Copper Sulphate Pentahydrate suppliers may include Copper Sulphate Pentahydrate API manufacturers, exporters, distributors and traders.
click here to find a list of Copper Sulphate Pentahydrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Copper Sulphate Pentahydrate DMF (Drug Master File) is a document detailing the whole manufacturing process of Copper Sulphate Pentahydrate active pharmaceutical ingredient (API) in detail. Different forms of Copper Sulphate Pentahydrate DMFs exist exist since differing nations have different regulations, such as Copper Sulphate Pentahydrate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Copper Sulphate Pentahydrate DMF submitted to regulatory agencies in the US is known as a USDMF. Copper Sulphate Pentahydrate USDMF includes data on Copper Sulphate Pentahydrate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Copper Sulphate Pentahydrate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Copper Sulphate Pentahydrate suppliers with USDMF on PharmaCompass.
A Copper Sulphate Pentahydrate CEP of the European Pharmacopoeia monograph is often referred to as a Copper Sulphate Pentahydrate Certificate of Suitability (COS). The purpose of a Copper Sulphate Pentahydrate CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Copper Sulphate Pentahydrate EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Copper Sulphate Pentahydrate to their clients by showing that a Copper Sulphate Pentahydrate CEP has been issued for it. The manufacturer submits a Copper Sulphate Pentahydrate CEP (COS) as part of the market authorization procedure, and it takes on the role of a Copper Sulphate Pentahydrate CEP holder for the record. Additionally, the data presented in the Copper Sulphate Pentahydrate CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Copper Sulphate Pentahydrate DMF.
A Copper Sulphate Pentahydrate CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Copper Sulphate Pentahydrate CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Copper Sulphate Pentahydrate suppliers with CEP (COS) on PharmaCompass.
Copper Sulphate Pentahydrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Copper Sulphate Pentahydrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Copper Sulphate Pentahydrate GMP manufacturer or Copper Sulphate Pentahydrate GMP API supplier for your needs.
A Copper Sulphate Pentahydrate CoA (Certificate of Analysis) is a formal document that attests to Copper Sulphate Pentahydrate's compliance with Copper Sulphate Pentahydrate specifications and serves as a tool for batch-level quality control.
Copper Sulphate Pentahydrate CoA mostly includes findings from lab analyses of a specific batch. For each Copper Sulphate Pentahydrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Copper Sulphate Pentahydrate may be tested according to a variety of international standards, such as European Pharmacopoeia (Copper Sulphate Pentahydrate EP), Copper Sulphate Pentahydrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Copper Sulphate Pentahydrate USP).

