
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Australia
0
Listed Dossiers
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
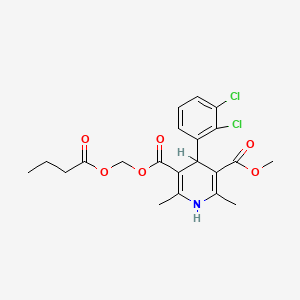

1. Butyroxymethyl Methyl 4-(2',3'-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate
2. Cleviprex
1. 167221-71-8
2. Clevidipine Butyrate
3. Cleviprex
4. Clevelox
5. Clevidepine
6. Clevidepine Butyrate
7. 166432-28-6
8. 3,5-pyridinedicarboxylic Acid, 4-(2,3-dichlorophenyl)-1,4-dihydro-2,6-dimethyl-, 3-methyl 5-[(1-oxobutoxy)methyl] Ester
9. Methyl (1-oxobutoxy)methyl 4-(2,3-dichlorophenyl)-1,4-dihydro-2,6-dime Thyl-3,5-pyridinedicarboxylate
10. 19o2gp3b7q
11. 3-((butyryloxy)methyl) 5-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate
12. 5-o-(butanoyloxymethyl) 3-o-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate
13. Methyl (1-oxobutoxy)methyl 4-(2,3-dichlorophenyl)-1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate
14. Rac-clevidipine
15. H324/38
16. 3,5-pyridinedicarboxylic Acid, 4-(2,3-dichlorophenyl)-1,4-dihydro-2,6-dimethyl-, Methyl (1-oxobutoxy)methyl Ester
17. Methyl 5-{[(butanoyloxy)methoxy]carbonyl}-4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3-carboxylate
18. H-324/38
19. (butanoyloxy)methyl Methyl (4rs)-4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate
20. Sr-01000945183
21. Clevidipine [usan:inn]
22. Unii-19o2gp3b7q
23. Cleviprex (tn)
24. 3,5-pyridinedicarboxylic Acid, 4-(2,3-dichlorophenyl)-1,4-dihydro-2,6-dimethyl-, 3-methyl 5-((1-oxobutoxy)methyl) Ester
25. 3-[(butyryloxy)methyl] 5-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate
26. Clevidipine [inn]
27. H 324/38
28. Cleviprex (clevidipine)
29. Clevidipine [mi]
30. Clevidipine (usan/inn)
31. Clevidipine [usan]
32. Clevidepine [vandf]
33. Clevidipine [vandf]
34. Clevidipine [mart.]
35. Dsstox_cid_31450
36. Dsstox_rid_97336
37. Dsstox_gsid_57661
38. Clevidipine [who-dd]
39. Schembl115522
40. Gtpl7468
41. Clevidipine Butyrate;clevidipine
42. Chembl1237132
43. Dtxsid6057661
44. Clevidipine [orange Book]
45. Chebi:135738
46. Hms3604h21
47. Hms3651g10
48. Hms3884l03
49. Amy22141
50. Bcp22676
51. Bcp22687
52. Ex-a2898
53. Clevidepine Butyrate [vandf]
54. Clevidipine Butyrate [vandf]
55. Tox21_113923
56. Bdbm50088387
57. Mfcd00940070
58. S2080
59. Akos015896325
60. Ccg-269294
61. Cs-1427
62. Db04920
63. Ncgc00262928-01
64. 4-(2,3-dichlorophenyl)-1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylic Acid 3-methyl 5-[(1-oxobutoxy)methyl] Ester
65. Ac-24370
66. As-19935
67. Butyroxymethyl Methyl 4-(2',3'-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate
68. Clevidipine Butyrate [orange Book]
69. Hy-17436
70. Db-064636
71. C3503
72. Cas-167221-71-8
73. Ft-0659562
74. Ft-0688416
75. Sw220087-1
76. D08892
77. Ab01566888_01
78. 221c718
79. A810832
80. J-520103
81. Q5132338
82. Sr-01000945183-1
83. Sr-01000945183-2
84. Z1691545244
85. 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylic Aicd 3-[(butyryloxy)methyl] 5-methyl Ester
86. Butyroxymethyl Methyl 4-(2',3'-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylat
87. O3-(butanoyloxymethyl) O5-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate
| Molecular Weight | 456.3 g/mol |
|---|---|
| Molecular Formula | C21H23Cl2NO6 |
| XLogP3 | 4.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 10 |
| Exact Mass | 455.0902428 g/mol |
| Monoisotopic Mass | 455.0902428 g/mol |
| Topological Polar Surface Area | 90.9 Ų |
| Heavy Atom Count | 30 |
| Formal Charge | 0 |
| Complexity | 748 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Cleviprex |
| PubMed Health | Clevidipine (Injection) |
| Drug Classes | Antihypertensive |
| Drug Label | Cleviprex is a sterile, milky-white emulsion containing 0.5 mg/mL of clevidipine suitable for intravenous administration. Clevidipine is a dihydropyridine calcium channel blocker. Chemically, the active substance, clevidipine, is butyroxymethyl methy... |
| Active Ingredient | Clevidipine |
| Dosage Form | Emulsion |
| Route | Intravenous |
| Strength | 125mg/250ml (0.5mg/ml); 25mg/50ml (0.5mg/ml); 50mg/100ml (0.5mg/ml) |
| Market Status | Prescription |
| Company | Medicines |
| 2 of 2 | |
|---|---|
| Drug Name | Cleviprex |
| PubMed Health | Clevidipine (Injection) |
| Drug Classes | Antihypertensive |
| Drug Label | Cleviprex is a sterile, milky-white emulsion containing 0.5 mg/mL of clevidipine suitable for intravenous administration. Clevidipine is a dihydropyridine calcium channel blocker. Chemically, the active substance, clevidipine, is butyroxymethyl methy... |
| Active Ingredient | Clevidipine |
| Dosage Form | Emulsion |
| Route | Intravenous |
| Strength | 125mg/250ml (0.5mg/ml); 25mg/50ml (0.5mg/ml); 50mg/100ml (0.5mg/ml) |
| Market Status | Prescription |
| Company | Medicines |
For the reduction of blood pressure when when oral antihypertensive therapy is not feasible or not desirable.
FDA Label
Treatment of hypertensive disease
Clevidipine belongs to a well-known class of drugs called dihydropyridine calcium channel antagonists. Clevidpine is the first third generation intravenous dihydropyridine calcium channel blocker. In vitro studies demonstrated that clevidipine acts by selectively relaxing the smooth muscle cells that line small arteries, resulting in arterial dilation, widening of the artery opening, and without reducing central venous pressure or reducing cardiac output.
Calcium Channel Blockers
A class of drugs that act by selective inhibition of calcium influx through cellular membranes. (See all compounds classified as Calcium Channel Blockers.)
C08CA16
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
C - Cardiovascular system
C08 - Calcium channel blockers
C08C - Selective calcium channel blockers with mainly vascular effects
C08CA - Dihydropyridine derivatives
C08CA16 - Clevidipine
Route of Elimination
urine 63-74%, feces 7-22%
Clevidipine is rapidly hydrolyzed to inactive metabolites by esterases in arterial blood.
1 minute
Possibly by deforming the channel, inhibiting ion-control gating mechanisms, and/or interfering with the release of calcium from the sarcoplasmic reticulum, clevidipine inhibits the influx of extracellular calcium across both the myocardial and vascular smooth muscle cell membranes. The resultant inhibition of the contractile processes of the myocardial smooth muscle cells leads to dilation of the coronary and systemic arteries and improved oxygen delivery to the myocardial tissue.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
75
PharmaCompass offers a list of Clevidipine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Clevidipine manufacturer or Clevidipine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Clevidipine manufacturer or Clevidipine supplier.
A Clevidipine Butyrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Clevidipine Butyrate, including repackagers and relabelers. The FDA regulates Clevidipine Butyrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Clevidipine Butyrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Clevidipine Butyrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Clevidipine Butyrate supplier is an individual or a company that provides Clevidipine Butyrate active pharmaceutical ingredient (API) or Clevidipine Butyrate finished formulations upon request. The Clevidipine Butyrate suppliers may include Clevidipine Butyrate API manufacturers, exporters, distributors and traders.
click here to find a list of Clevidipine Butyrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Clevidipine Butyrate DMF (Drug Master File) is a document detailing the whole manufacturing process of Clevidipine Butyrate active pharmaceutical ingredient (API) in detail. Different forms of Clevidipine Butyrate DMFs exist exist since differing nations have different regulations, such as Clevidipine Butyrate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Clevidipine Butyrate DMF submitted to regulatory agencies in the US is known as a USDMF. Clevidipine Butyrate USDMF includes data on Clevidipine Butyrate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Clevidipine Butyrate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Clevidipine Butyrate suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Clevidipine Butyrate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Clevidipine Butyrate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Clevidipine Butyrate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Clevidipine Butyrate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Clevidipine Butyrate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Clevidipine Butyrate suppliers with NDC on PharmaCompass.
Clevidipine Butyrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Clevidipine Butyrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Clevidipine Butyrate GMP manufacturer or Clevidipine Butyrate GMP API supplier for your needs.
A Clevidipine Butyrate CoA (Certificate of Analysis) is a formal document that attests to Clevidipine Butyrate's compliance with Clevidipine Butyrate specifications and serves as a tool for batch-level quality control.
Clevidipine Butyrate CoA mostly includes findings from lab analyses of a specific batch. For each Clevidipine Butyrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Clevidipine Butyrate may be tested according to a variety of international standards, such as European Pharmacopoeia (Clevidipine Butyrate EP), Clevidipine Butyrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Clevidipine Butyrate USP).

