
Synopsis
Synopsis
0
CEP/COS
0
VMF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
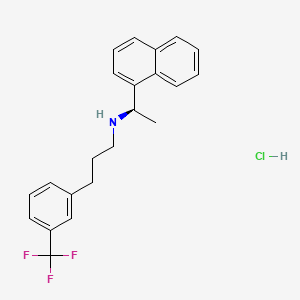

1. Alpha-methyl-n-(3-(3-(trifluoromethyl)phenyl)propyl)-1-naphthalenemethanamine, (alphar)-hydrochloride
2. Amg 073
3. Amg073
4. Cinacalcet
5. Krn 1493
6. Sensipar
1. 364782-34-3
2. Cinacalcet Hcl
3. Sensipar
4. Mimpara
5. Amg073 Hcl
6. Regpara
7. (r)-n-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)propan-1-amine Hydrochloride
8. Cinacalcet, Hcl
9. Cinacalcet Hydrochloride [usan]
10. Cinacalcet (hydrochloride)
11. Amg-073 Hcl
12. Amg073 Hydrochloride
13. Chebi:48391
14. 1k860wsg25
15. Parareg
16. N-[(1r)-1-naphthalen-1-ylethyl]-3-[3-(trifluoromethyl)phenyl]propan-1-amine;hydrochloride
17. Amg-073 Hcl (cinacalcet Hydrochloride)
18. 364782-34-3 (hcl)
19. [(1r)-1-(naphthalen-1-yl)ethyl]({3-[3-(trifluoromethyl)phenyl]propyl})amine Hydrochloride
20. Dsstox_cid_26792
21. Dsstox_rid_81909
22. N-((1r)-1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)propan-1-amine Hydrochloride
23. N-[(1r)-1-(1-naphthyl)ethyl]-3-[3-(trifluoromethyl)phenyl]propan-1-amine Hydrochloride
24. Dsstox_gsid_46792
25. (r)-n-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoro-methyl)phenyl)propan-1-amine Hydrochloride
26. Smr002530058
27. Cas-364782-34-3
28. Krn 1493
29. Ncgc00181002-01
30. Amg 073 Hcl
31. Unii-1k860wsg25
32. Sensipar (tn)
33. Mimpara (tn)
34. Cnc-hcl
35. Cinacelcet Hydrochloride
36. Sensipar(tm)
37. Amg-073 Hydrochloride
38. Amg-073.hcl
39. Mls004774045
40. Mls006010213
41. Chembl1200776
42. Dtxsid3046792
43. Bcpp000409
44. Bcp02533
45. Nps-1493
46. Tox21_112654
47. Cinacalcet Hydrochloride (jan/usan)
48. Cinacalcet Hydrochloride [mi]
49. Hy-70037a
50. Mfcd08067750
51. S1260
52. Cinacalcet Hydrochloride [jan]
53. Akos005146514
54. Akos015969126
55. Tox21_112654_1
56. Ac-1799
57. Am90312
58. Bcp9000286
59. Ccg-268579
60. Cs-0288
61. Gs-4171
62. Cinacalcet Hydrochloride [mart.]
63. Cinacalcet Hydrochloride [usp-rs]
64. Cinacalcet Hydrochloride [who-dd]
65. Ncgc00181002-04
66. 1-naphthalenemethanamine, Alpha-methyl-n-(3-(3-(trifluoromethyl)phenyl)propyl)-, (alphar)-, Hydrochloride
67. Sw219246-1
68. Cinacalcet Hydrochloride [orange Book]
69. D03505
70. 782c343
71. J-520045
72. Q27121179
73. Z1741977003
74. (r)-n-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)propan-1-amine Hcl
75. (r)-n-(3-(3-(trifluoromethyl)phenyl)propyl)-1- -(1-napthyl)ethylamine Hydrochloride
76. (alphar)-alpha-methyl-n-[3-[3-(trifluoromethyl)phenyl)propyl]-1-napthalenemethanamine Hydrochloride
77. 1-naphthalenemethanamine, .alpha.-methyl-n-(3-(3-(trifluoromethyl)phenyl)propyl)-, (.alpha.r)-, Hydrochloride
78. 1-naphthalenemethanamine, Alpha-methyl-n-[3-[3-(trifluoromethyl)phenyl]propyl]-, Hydrochloride (1:1), (alphar)-
79. 1-naphthalenemethanamine,a-methyl-n-[3-[3-(trifluoromethyl)phenyl]propyl]-, Hydrochloride, (ar)-
| Molecular Weight | 393.9 g/mol |
|---|---|
| Molecular Formula | C22H23ClF3N |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Exact Mass | 393.1471119 g/mol |
| Monoisotopic Mass | 393.1471119 g/mol |
| Topological Polar Surface Area | 12 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 422 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 4 | |
|---|---|
| Drug Name | Cinacalcet hydrochloride |
| PubMed Health | Cinacalcet (By mouth) |
| Drug Classes | Calcium Regulator |
| Drug Label | Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C22H22F3NHCl with a molecular weight of 393.9 g/mol (hydrochloride salt... |
| Active Ingredient | Cinacalcet hydrochloride |
| Dosage Form | Tablet |
| Route | oral |
| Strength | 60mg; 30mg; 90mg |
| Market Status | Tentative Approval |
| Company | Teva Pharms; Barr |
| 2 of 4 | |
|---|---|
| Drug Name | Sensipar |
| Drug Label | Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C22H22F3NHCl with a molecular weight of 393.9 g/mol (hydrochloride salt... |
| Active Ingredient | Cinacalcet hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 90mg base; eq 30mg base; eq 60mg base |
| Market Status | Prescription |
| Company | Amgen |
| 3 of 4 | |
|---|---|
| Drug Name | Cinacalcet hydrochloride |
| PubMed Health | Cinacalcet (By mouth) |
| Drug Classes | Calcium Regulator |
| Drug Label | Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C22H22F3NHCl with a molecular weight of 393.9 g/mol (hydrochloride salt... |
| Active Ingredient | Cinacalcet hydrochloride |
| Dosage Form | Tablet |
| Route | oral |
| Strength | 60mg; 30mg; 90mg |
| Market Status | Tentative Approval |
| Company | Teva Pharms; Barr |
| 4 of 4 | |
|---|---|
| Drug Name | Sensipar |
| Drug Label | Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C22H22F3NHCl with a molecular weight of 393.9 g/mol (hydrochloride salt... |
| Active Ingredient | Cinacalcet hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 90mg base; eq 30mg base; eq 60mg base |
| Market Status | Prescription |
| Company | Amgen |
* Secondary hyperparathyroidism :
Adults
Treatment of secondary hyperparathyroidism (HPT) in adult patients with end-stage renal disease (ESRD) on maintenance dialysis therapy.
Paediatric population
Treatment of secondary hyperparathyroidism (HPT) in children aged 3 years and older with end-stage renal disease (ESRD) on maintenance dialysis therapy in whom secondary HPT is not adequately controlled with standard of care therapy (see section 4. 4).
Cinacalcet Accordpharma may be used as part of a therapeutic regimen including phosphate binders and/or Vitamin D sterols, as appropriate (see section 5. 1).
* Parathyroid carcinoma and primary hyperparathyroidism in adults:
Reduction of hypercalcaemia in adult patients with:
- parathyroid carcinoma.
- primary HPT for whom parathyroidectomy would be indicated on the basis of serum calcium levels (as defined by relevant treatment guidelines), but in whom parathyroidectomy is not clinically appropriate or is contraindicated.
Treatment of secondary hyperparathyroidism (HPT) in patients with end-stage renal disease (ESRD) on maintenance dialysis therapy.
Cinacalcet Mylan may be used as part of a therapeutic regimen including phosphate binders and/or vitamin D sterols, as appropriate.
Reduction of hypercalcaemia in patients with:
- parathyroid carcinoma
- primary HPT for whom parathyroidectomy
would be indicated on the basis of serum calcium levels (as defined by relevant treatment guidelines), but in whom parathyroidectomy is not clinically appropriate or is contraindicated.
* Secondary hyperparathyroidism :
Adults
Treatment of secondary hyperparathyroidism (HPT) in adult patients with end stage renal disease (ESRD) on maintenance dialysis therapy.
Paediatric population
Treatment of secondary hyperparathyroidism (HPT) in children aged 3 years and older with end stage renal disease (ESRD) on maintenance dialysis therapy in whom secondary HPT is not adequately controlled with standard of care therapy.
Mimpara may be used as part of a therapeutic regimen including phosphate binders and/or Vitamin D sterols, as appropriate.
Parathyroid carcinoma and primary hyperparathyroidism in adults.
Reduction of hypercalcaemia in adult patients with:
- parathyroid carcinoma;
- primary HPT for whom parathyroidectomy would be indicated on the basis of serum calcium levels (as defined by relevant treatment guidelines), but in whom parathyroidectomy is not clinically appropriate or is contraindicated.
Treatment of secondary hyperparathyroidism (HPT) in patients with end-stage renal disease (ESRD) on maintenance dialysis therapy.
Mimpara may be used as part of a therapeutic regimen including phosphate binders and/or Vitamin D sterols, as appropriate (see section 5. 1).
Reduction of hypercalcaemia in patients with:
-parathyroid carcinoma.
- primary HPT for whom parathyroidectomy would be indicated on the basis of serum calcium
levels (as defined by relevant treatment guidelines), but in whom parathyroidectomy is not clinically appropriate or is contraindicated.
Treatment of parathyroid carcinoma, Treatment of primary hyperparathyroidism , Treatment of secondary hyperparathyroidism in patients with end-stage renal disease
Calcimimetic Agents
Small organic molecules that act as allosteric activators of the calcium sensing receptor (CaSR) in the PARATHYROID GLANDS and other tissues. They lower the threshold for CaSR activation by extracellular calcium ions and diminish PARATHYROID HORMONE (PTH) release from parathyroid cells. (See all compounds classified as Calcimimetic Agents.)
Calcium-Regulating Hormones and Agents
Hormones and molecules with calcium-regulating hormone-like actions that modulate OSTEOLYSIS and other extra-skeletal activities to maintain calcium homeostasis. (See all compounds classified as Calcium-Regulating Hormones and Agents.)
H05BX01
H05BX01
H05BX01
H05BX01
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
15
PharmaCompass offers a list of Cinacalcet Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cinacalcet Hydrochloride manufacturer or Cinacalcet Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Cinacalcet Hydrochloride manufacturer or Cinacalcet Hydrochloride supplier.
A Cinacalcet Hydrochloride manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Cinacalcet Hydrochloride, including repackagers and relabelers. The FDA regulates Cinacalcet Hydrochloride manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Cinacalcet Hydrochloride API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Cinacalcet Hydrochloride manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Cinacalcet Hydrochloride supplier is an individual or a company that provides Cinacalcet Hydrochloride active pharmaceutical ingredient (API) or Cinacalcet Hydrochloride finished formulations upon request. The Cinacalcet Hydrochloride suppliers may include Cinacalcet Hydrochloride API manufacturers, exporters, distributors and traders.
click here to find a list of Cinacalcet Hydrochloride suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Cinacalcet Hydrochloride DMF (Drug Master File) is a document detailing the whole manufacturing process of Cinacalcet Hydrochloride active pharmaceutical ingredient (API) in detail. Different forms of Cinacalcet Hydrochloride DMFs exist exist since differing nations have different regulations, such as Cinacalcet Hydrochloride USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Cinacalcet Hydrochloride DMF submitted to regulatory agencies in the US is known as a USDMF. Cinacalcet Hydrochloride USDMF includes data on Cinacalcet Hydrochloride's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Cinacalcet Hydrochloride USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Cinacalcet Hydrochloride suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Cinacalcet Hydrochloride Drug Master File in Japan (Cinacalcet Hydrochloride JDMF) empowers Cinacalcet Hydrochloride API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Cinacalcet Hydrochloride JDMF during the approval evaluation for pharmaceutical products. At the time of Cinacalcet Hydrochloride JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Cinacalcet Hydrochloride suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Cinacalcet Hydrochloride Drug Master File in Korea (Cinacalcet Hydrochloride KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Cinacalcet Hydrochloride. The MFDS reviews the Cinacalcet Hydrochloride KDMF as part of the drug registration process and uses the information provided in the Cinacalcet Hydrochloride KDMF to evaluate the safety and efficacy of the drug.
After submitting a Cinacalcet Hydrochloride KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Cinacalcet Hydrochloride API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Cinacalcet Hydrochloride suppliers with KDMF on PharmaCompass.
A Cinacalcet Hydrochloride written confirmation (Cinacalcet Hydrochloride WC) is an official document issued by a regulatory agency to a Cinacalcet Hydrochloride manufacturer, verifying that the manufacturing facility of a Cinacalcet Hydrochloride active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Cinacalcet Hydrochloride APIs or Cinacalcet Hydrochloride finished pharmaceutical products to another nation, regulatory agencies frequently require a Cinacalcet Hydrochloride WC (written confirmation) as part of the regulatory process.
click here to find a list of Cinacalcet Hydrochloride suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Cinacalcet Hydrochloride as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Cinacalcet Hydrochloride API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Cinacalcet Hydrochloride as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Cinacalcet Hydrochloride and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Cinacalcet Hydrochloride NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Cinacalcet Hydrochloride suppliers with NDC on PharmaCompass.
Cinacalcet Hydrochloride Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Cinacalcet Hydrochloride GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cinacalcet Hydrochloride GMP manufacturer or Cinacalcet Hydrochloride GMP API supplier for your needs.
A Cinacalcet Hydrochloride CoA (Certificate of Analysis) is a formal document that attests to Cinacalcet Hydrochloride's compliance with Cinacalcet Hydrochloride specifications and serves as a tool for batch-level quality control.
Cinacalcet Hydrochloride CoA mostly includes findings from lab analyses of a specific batch. For each Cinacalcet Hydrochloride CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Cinacalcet Hydrochloride may be tested according to a variety of international standards, such as European Pharmacopoeia (Cinacalcet Hydrochloride EP), Cinacalcet Hydrochloride JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Cinacalcet Hydrochloride USP).

