
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
Canada
0
Listed Dossiers
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
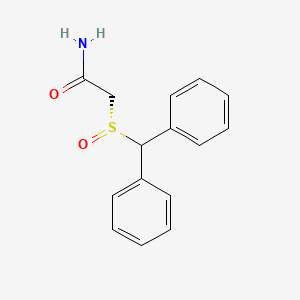

1. 2 Benzhydrylsulfinylacetamide
2. 2-((diphenylmethyl)sulfinyl)acetamide
3. 2-((r)-(diphenylmethyl)sulfinyl)acetamide
4. 2-(benzhydrylsulfinyl)acetamide
5. 2-benzhydrylsulfinylacetamide
6. Alertec
7. Benzhydrylsulfinylacetamide
8. Crl 40476
9. Crl-40476
10. Modafinil
11. Modiodal
12. Nuvigil
13. Provigil
14. R Modafinil
15. R-modafinil
16. Sparlon
1. 112111-43-0
2. Nuvigil
3. (r)-modafinil
4. (-)-modafinil
5. (r)-(-)-modafinil
6. Cep-10953
7. Crl 40982
8. Modafinil, (r)-
9. R-(-)-modafinil
10. 2-[(r)-(diphenylmethyl)sulfinyl]acetamide
11. (-)-(r)-modafinil
12. Cep 10953
13. Cep-10952
14. Crl-40982
15. V63xwa605i
16. Chembl1201192
17. Chebi:77590
18. Nsc-751850
19. Nsc-758711
20. (-)-2-((r)-(diphenylmethyl)sulfinyl)acetamide
21. Acetamide, 2-((diphenylmethyl)sulfinyl)-, (-)-
22. Armodafinil [inn]
23. (-)-2-[(r)-(diphenylmethyl)sulfinyl]acetamide
24. Armodafinil [usan:inn]
25. Armodafinilo
26. Armodafinilum
27. Unii-v63xwa605i
28. L-modafinil
29. (-) Modafinil
30. Nuvigil (tn)
31. Armodafinil (usan/inn)
32. Armodafinil [usan]
33. Armodafinil [vandf]
34. Armodafinil [mart.]
35. Schembl34489
36. Armodafinil [usp-rs]
37. Armodafinil [who-dd]
38. Zinc6156
39. Armodafinil, >=98% (hplc)
40. Armodafinil [orange Book]
41. Dtxsid90920667
42. 2-[(r)-benzhydrylsulfinyl]acetamide
43. Bdbm50336892
44. Akos030211019
45. At22562
46. Ccg-230228
47. Cs-0665
48. Db06413
49. Nsc 751850
50. Nsc 758711
51. 2-[(r)-diphenylmethanesulfinyl]acetamide
52. Hy-15201
53. (-)-2r-[(diphenylmethyl)sulfinyl]acetamide
54. S4645
55. D03215
56. Q418913
57. Acetamide, 2-((r)-(diphenylmethyl)sulfinyl)-
| Molecular Weight | 273.4 g/mol |
|---|---|
| Molecular Formula | C15H15NO2S |
| XLogP3 | 1.7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 5 |
| Exact Mass | 273.08234989 g/mol |
| Monoisotopic Mass | 273.08234989 g/mol |
| Topological Polar Surface Area | 79.4 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 302 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Armodafinil |
| PubMed Health | Armodafinil (By mouth) |
| Drug Classes | CNS Stimulant |
| Drug Label | NUVIGIL (armodafinil) is a wakefulness-promoting agent for oral administration. Armodafinil is the R-enantiomer of modafinil which is a mixture of the R- and S-enantiomers. The chemical name for armodafinil is 2-[(R)-(diphenylmethyl)sulfinyl]acetam... |
| Active Ingredient | Armodafinil |
| Dosage Form | Tablet |
| Route | oral; Oral |
| Strength | 2500mg; 200mg; 250mg; 100mg; 50mg; 150mg |
| Market Status | Tentative Approval; Prescription |
| Company | Watson Labs; Mylan Pharms; Teva Pharms Usa; Lupin |
| 2 of 4 | |
|---|---|
| Drug Name | Nuvigil |
| PubMed Health | Armodafinil (By mouth) |
| Drug Classes | CNS Stimulant |
| Drug Label | NUVIGIL (armodafinil) is a wakefulness-promoting agent for oral administration. Armodafinil is the R-enantiomer of modafinil which is a mixture of the R- and S-enantiomers. The chemical name for armodafinil is 2-[(R)-(diphenylmethyl)sulfinyl]acetam... |
| Active Ingredient | Armodafinil |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 250mg; 150mg; 50mg |
| Market Status | Prescription |
| Company | Cephalon |
| 3 of 4 | |
|---|---|
| Drug Name | Armodafinil |
| PubMed Health | Armodafinil (By mouth) |
| Drug Classes | CNS Stimulant |
| Drug Label | NUVIGIL (armodafinil) is a wakefulness-promoting agent for oral administration. Armodafinil is the R-enantiomer of modafinil which is a mixture of the R- and S-enantiomers. The chemical name for armodafinil is 2-[(R)-(diphenylmethyl)sulfinyl]acetam... |
| Active Ingredient | Armodafinil |
| Dosage Form | Tablet |
| Route | oral; Oral |
| Strength | 2500mg; 200mg; 250mg; 100mg; 50mg; 150mg |
| Market Status | Tentative Approval; Prescription |
| Company | Watson Labs; Mylan Pharms; Teva Pharms Usa; Lupin |
| 4 of 4 | |
|---|---|
| Drug Name | Nuvigil |
| PubMed Health | Armodafinil (By mouth) |
| Drug Classes | CNS Stimulant |
| Drug Label | NUVIGIL (armodafinil) is a wakefulness-promoting agent for oral administration. Armodafinil is the R-enantiomer of modafinil which is a mixture of the R- and S-enantiomers. The chemical name for armodafinil is 2-[(R)-(diphenylmethyl)sulfinyl]acetam... |
| Active Ingredient | Armodafinil |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 250mg; 150mg; 50mg |
| Market Status | Prescription |
| Company | Cephalon |
Investigated for use/treatment in sleep disorders, obstructive sleep apnea, schizophrenia and schizoaffective disorders, depression, and bipolar disorders.
FDA Label
Central Nervous System Stimulants
A loosely defined group of drugs that tend to increase behavioral alertness, agitation, or excitation. They work by a variety of mechanisms, but usually not by direct excitation of neurons. The many drugs that have such actions as side effects to their main therapeutic use are not included here. (See all compounds classified as Central Nervous System Stimulants.)
Cytochrome P-450 CYP3A Inducers
Drugs and compounds that induce the synthesis of CYTOCHROME P-450 CYP3A. (See all compounds classified as Cytochrome P-450 CYP3A Inducers.)
Wakefulness-Promoting Agents
A specific category of drugs that prevent sleepiness by specifically targeting sleep-mechanisms in the brain. They are used to treat DISORDERS OF EXCESSIVE SOMNOLENCE such as NARCOLEPSY. Note that this drug category does not include broadly-acting central nervous system stimulants such as AMPHETAMINES. (See all compounds classified as Wakefulness-Promoting Agents.)
N - Nervous system
N06 - Psychoanaleptics
N06B - Psychostimulants, agents used for adhd and nootropics
N06BA - Centrally acting sympathomimetics
N06BA13 - Armodafinil
Absorption
Tmax is 2 hours when fasted and can be delayed approximately 2-4 hours by food, potentially affecting the onset of action.
Volume of Distribution
Apparent volume of distribution: 42L.
Clearance
The oral clearance of armodafinil is approximately 33 mL/min.
In vitro and in vivo data show that armodafinil undergoes hydrolytic deamidation, S-oxidation, and aromatic ring hydroxylation, with subsequent glucuronide conjugation of the hydroxylated products. Amide hydrolysis is the single most prominent metabolic pathway, with sulfone formation by cytochrome P450 (CYP) 3A4/5 being next in importance. The other oxidative products are formed too slowly in vitro to enable identification of the enzyme(s) responsible. Only two metabolites reach appreciable concentrations in plasma (i.e., R-modafinil acid and modafinil sulfone). Data specific to armodafinil disposition are not available.
Terminal half-life is approximately 15 hours.
Nuvigil (armodafinil) is a single-isomer of modafini. The exact mechanism of action is unknown. Armodafinil belongs to a class of drugs known as eugeroics, which are stimulants that provide long-lasting mental arousal. Pharmacologically, armodafinil does not bind to or inhibit several receptors and enzymes potentially relevant for sleep/wake regulation. Armodafinil is not a direct- or indirect-acting dopamine receptor agonist. However, in vitro, both armodafinil and modafinil bind to the dopamine transporter and inhibit dopamine reuptake. [Medilexicon]
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
17
PharmaCompass offers a list of Armodafinil API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Armodafinil manufacturer or Armodafinil supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Armodafinil manufacturer or Armodafinil supplier.
A Cephalon brand of armodafinil manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Cephalon brand of armodafinil, including repackagers and relabelers. The FDA regulates Cephalon brand of armodafinil manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Cephalon brand of armodafinil API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Cephalon brand of armodafinil manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Cephalon brand of armodafinil supplier is an individual or a company that provides Cephalon brand of armodafinil active pharmaceutical ingredient (API) or Cephalon brand of armodafinil finished formulations upon request. The Cephalon brand of armodafinil suppliers may include Cephalon brand of armodafinil API manufacturers, exporters, distributors and traders.
click here to find a list of Cephalon brand of armodafinil suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Cephalon brand of armodafinil DMF (Drug Master File) is a document detailing the whole manufacturing process of Cephalon brand of armodafinil active pharmaceutical ingredient (API) in detail. Different forms of Cephalon brand of armodafinil DMFs exist exist since differing nations have different regulations, such as Cephalon brand of armodafinil USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Cephalon brand of armodafinil DMF submitted to regulatory agencies in the US is known as a USDMF. Cephalon brand of armodafinil USDMF includes data on Cephalon brand of armodafinil's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Cephalon brand of armodafinil USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Cephalon brand of armodafinil suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Cephalon brand of armodafinil Drug Master File in Korea (Cephalon brand of armodafinil KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Cephalon brand of armodafinil. The MFDS reviews the Cephalon brand of armodafinil KDMF as part of the drug registration process and uses the information provided in the Cephalon brand of armodafinil KDMF to evaluate the safety and efficacy of the drug.
After submitting a Cephalon brand of armodafinil KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Cephalon brand of armodafinil API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Cephalon brand of armodafinil suppliers with KDMF on PharmaCompass.
A Cephalon brand of armodafinil written confirmation (Cephalon brand of armodafinil WC) is an official document issued by a regulatory agency to a Cephalon brand of armodafinil manufacturer, verifying that the manufacturing facility of a Cephalon brand of armodafinil active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Cephalon brand of armodafinil APIs or Cephalon brand of armodafinil finished pharmaceutical products to another nation, regulatory agencies frequently require a Cephalon brand of armodafinil WC (written confirmation) as part of the regulatory process.
click here to find a list of Cephalon brand of armodafinil suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Cephalon brand of armodafinil as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Cephalon brand of armodafinil API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Cephalon brand of armodafinil as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Cephalon brand of armodafinil and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Cephalon brand of armodafinil NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Cephalon brand of armodafinil suppliers with NDC on PharmaCompass.
Cephalon brand of armodafinil Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Cephalon brand of armodafinil GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cephalon brand of armodafinil GMP manufacturer or Cephalon brand of armodafinil GMP API supplier for your needs.
A Cephalon brand of armodafinil CoA (Certificate of Analysis) is a formal document that attests to Cephalon brand of armodafinil's compliance with Cephalon brand of armodafinil specifications and serves as a tool for batch-level quality control.
Cephalon brand of armodafinil CoA mostly includes findings from lab analyses of a specific batch. For each Cephalon brand of armodafinil CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Cephalon brand of armodafinil may be tested according to a variety of international standards, such as European Pharmacopoeia (Cephalon brand of armodafinil EP), Cephalon brand of armodafinil JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Cephalon brand of armodafinil USP).

