
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
Europe
0
Canada
0
Australia
0
South Africa
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
Regulatory FDF Prices
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
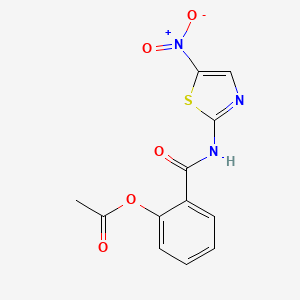

1. 2-(acetolyloxy)-n-(5-nitro-2-thiazolyl)benzamide
2. Alinia
3. Colufase
4. Cryptaz
5. Daxon
6. Heliton
7. Ntz
8. Taenitaz
1. 55981-09-4
2. Alinia
3. 2-((5-nitrothiazol-2-yl)carbamoyl)phenyl Acetate
4. Nitazoxamide
5. Daxon
6. Nitazoxanidum [inn-latin]
7. Nitazoxanida [inn-spanish]
8. 2-[(5-nitro-1,3-thiazol-2-yl)carbamoyl]phenyl Acetate
9. Benzamide, 2-(acetyloxy)-n-(5-nitro-2-thiazolyl)-
10. [2-[(5-nitro-1,3-thiazol-2-yl)carbamoyl]phenyl] Acetate
11. 2-(acetolyloxy)-n-(5-nitro-2-thiazolyl)benzamide
12. Nsc 697855
13. Nsc697855
14. Ntz
15. 2-acetyloxy-n-[(5-nitro-2-thiazolyl)]benzamide
16. Nsc-697855
17. Nsc-760057
18. N-(5-nitrothiazol-2-yl)salicylamide Acetate Ester
19. Ph 5776
20. Ph-5776
21. Nitazoxanide (alinia, Annita)
22. Soa12p041n
23. Nitazoxanida
24. Cryptaz
25. [2-[(5-nitrothiazol-2-yl)carbamoyl]phenyl] Acetate
26. Ncgc00090774-01
27. Nitazoxanid
28. Nitazoxanidum
29. Colufase
30. Heliton
31. Dsstox_cid_13757
32. Dsstox_rid_79095
33. Dsstox_gsid_33757
34. Phavic-1
35. Smr000466367
36. Alinia (tn)
37. Azt + Nitazoxanide
38. Cas-55981-09-4
39. Einecs 259-931-8
40. Nitazoxanide (usan/inn)
41. (2-((5-nitro-2-thiazolyl)carbamoyl)phenyl)acetat
42. Brn 1225475
43. Unii-soa12p041n
44. N-(5-nitro-2-thiazolyl)salicylamide Acetate (ester)
45. Nitrazoxanide
46. Pacovanton
47. Dexidex
48. Kidonax
49. Nitazox
50. Paramix
51. Nitax
52. Toza
53. Nitazoxanide [usan:inn:ban]
54. Alinia(tm)
55. Mfcd00416599
56. O-(n-(5-nitrothiazol-2-yl)carbamoyl)phenyl Acetate
57. Nitazoxanide- Bio-x
58. Salicylamide, N-(5-nitro-2-thiazolyl)-, Acetate (ester)
59. Cpd000466367
60. Nitazoxanide [mi]
61. Nitazoxanide [inn]
62. Ncimech_000843
63. Nitazoxanide [usan]
64. Chembl1401
65. Nitazoxanide [vandf]
66. 2-(acetyloxy)-n-(5-nitro-2-thiazolyl)benzamide
67. Oprea1_263587
68. Schembl40981
69. Nitazoxanide [mart.]
70. Mls000759492
71. Mls001424074
72. Mls006010127
73. Nitazoxanide [usp-rs]
74. Nitazoxanide [who-dd]
75. Dtxsid5033757
76. Chebi:94807
77. Nitazoxanide [green Book]
78. Nitazoxanide, >=98% (hplc)
79. Ntz;nsc 697855
80. Hms2051l04
81. Hms3393l04
82. Hms3655m11
83. Hms3715f10
84. Nitazoxanide [orange Book]
85. Pharmakon1600-01503843
86. Bcp13918
87. Hy-b0217
88. Zinc3956788
89. Tox21_111018
90. Tox21_201226
91. Bdbm50075050
92. Ccg-35851
93. Mmv688991
94. Nsc760057
95. Stk395664
96. Akos015915393
97. Tox21_111018_1
98. Ac-1302
99. Db00507
100. Ks-1160
101. Nc00246
102. Ncgc00090774-02
103. Ncgc00090774-03
104. Ncgc00090774-04
105. Ncgc00090774-05
106. Ncgc00258778-01
107. Bn164151
108. Nci60_034935
109. Ft-0601547
110. N1031
111. Nitrazoxanide 100 Microg/ml In Acetonitrile
112. S1627
113. Sw197626-2
114. 2-(5-nitrothiazol-2-ylcarbamoyl)phenyl Acetate
115. D02486
116. Ab00639988-07
117. Ab00639988-09
118. Ab00639988_10
119. Ab00639988_11
120. 2-(acetyloxy)-n-(5-nitro-2-thiazolyl)-benzamide
121. 981n094
122. A830877
123. Sr-01000759418
124. Q-201475
125. Q2943789
126. Sr-01000759418-9
127. Salicylamide, N-(5-nitro-2-thiazolyl)- Acetate (ester)
128. Z1514087129
129. Nitazoxanide, United States Pharmacopeia (usp) Reference Standard
130. Acetic Acid [2-[[(5-nitro-2-thiazolyl)amino]-oxomethyl]phenyl] Ester
131. Zox
| Molecular Weight | 307.28 g/mol |
|---|---|
| Molecular Formula | C12H9N3O5S |
| XLogP3 | 2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 4 |
| Exact Mass | 307.02629157 g/mol |
| Monoisotopic Mass | 307.02629157 g/mol |
| Topological Polar Surface Area | 142 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 428 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Alinia |
| PubMed Health | Nitazoxanide (By mouth) |
| Drug Classes | Antiprotozoal |
| Drug Label | Alinia Tablets and Alinia for Oral Suspension contain the active ingredient, nitazoxanide, a synthetic antiprotozoal agent for oral administration. Nitazoxanide is a light yellow crystalline powder. It is poorly soluble in ethanol and practically ins... |
| Active Ingredient | Nitazoxanide |
| Dosage Form | Tablet; For suspension |
| Route | oral; Oral |
| Strength | 100mg/5ml; 500mg |
| Market Status | Prescription |
| Company | Romark |
| 2 of 2 | |
|---|---|
| Drug Name | Alinia |
| PubMed Health | Nitazoxanide (By mouth) |
| Drug Classes | Antiprotozoal |
| Drug Label | Alinia Tablets and Alinia for Oral Suspension contain the active ingredient, nitazoxanide, a synthetic antiprotozoal agent for oral administration. Nitazoxanide is a light yellow crystalline powder. It is poorly soluble in ethanol and practically ins... |
| Active Ingredient | Nitazoxanide |
| Dosage Form | Tablet; For suspension |
| Route | oral; Oral |
| Strength | 100mg/5ml; 500mg |
| Market Status | Prescription |
| Company | Romark |
For the treatment of diarrhea in adults and children caused by the protozoa Giardia lamblia, and for the treatment of diarrhea in children caused by the protozoan, Cryptosporidium parvum. Nitazoxanide has not been shown to be superior to placebo medication for the management of diarrhea caused by Cryptosporidium parvum in patients with HIV/immunodeficiency.
FDA Label
The general effect of this medication is the prevention of microbe activity through disruption of important energy pathways for survival and proliferation. Nitazoxanide exhibits antiprotozoal activity by interfering with the pyruvate ferredoxin/flavodoxin oxidoreductase dependent electron transfer reaction, an essential reaction need for anaerobic energy metabolism of various microorganisms. Sporozoites of Cryptosporidium parvum and trophozoites of Giardia lamblia are therefore inhibited, relieving symptoms of diahrrea. Interference with the PFOR enzyme-dependent electron transfer reaction may only be one of the many pathways by which nitazoxanide exhibits antiprotozoal activity.
Antiparasitic Agents
Drugs used to treat or prevent parasitic infections. (See all compounds classified as Antiparasitic Agents.)
P - Antiparasitic products, insecticides and repellents
P01 - Antiprotozoals
P01A - Agents against amoebiasis and other protozoal diseases
P01AX - Other agents against amoebiasis and other protozoal diseases
P01AX11 - Nitazoxanide
Absorption
The relative bioavailability of the suspension compared to the tablet was 70%. When administered with food the AUC and Cmax increased by two-fold and 50%, respectively, for the tablet and 45 to 50% and ≤ 10%, respectively, for the oral suspension.
Route of Elimination
Tizoxanide is excreted in the urine, bile and feces, and tizoxanide glucuronide is excreted in urine and bile. Approximately 2/3 of the oral dose of nitazoxanide is excreted in the faeces and 1/3 in the urine.
Clearance
Nitazoxanide is cleared in the urine and feces. The metabolite, tizoxanide, is also found in the urine, plasma, and breastmilk. The drug is not found unchanged in the urine.
The active metabolite of this drug is tizoxanide (desacetyl-nitazoxanide). The initial reaction in the metabolic pathway of Nitazoxanide is hydrolysis to tizoxanide, followed by conjugation, primarily by glucuronidation to tizoxanide glucuronide. The oral suspension bioavailability of this drug is not equivalent to that of the oral tablets. Compared to the to the tablet, the bioavailability of the suspension was 70%. When administered with food, the AUCt of tizoxanide and tizoxanide glucuronide in plasma is increased to almost two-fold and the maximum concentration is increased by almost 50% compared to when ingested without food. When the oral suspension was ingested with food, the AUC of tizoxanide and tizoxanide glucuronide increased by approximately 50% and the Cmax increased by less than 10%.
7.3h
The most widely accepted mechanism of NTZ is believed to be the disruption of the energy metabolism in anaerobic microbes by inhibition of the pyruvate: ferredoxin/flavodoxin oxidoreductase (PFOR) cycle. In parasitic-protozoa, Nitazoxanide also induces lesions in the cell membranes and depolarizes the mitochondrial membrane while inhibiting quinone oxidoreductase NQO1, nitroreductase-1 and protein disulphide isomerase enzymes. In addition, this drug also inhibits the glutathione-S-transferase (a major detoxifying enzyme) and modulates the Avr-14 gene, encoding for the alpha-type subunit of glutamate-gated chloride ion channel present in nematodes. Aside from its well understood non-competitive inhibition of the PFOR in anaerobic bacteria, NTZ also demonstrates various other antibacterial mechanisms. It inhibits pyruvate dehydrogenase in E Coli, disrupts the membrane potential and pH homeostasis in the Mycobacterium tuberculosis, suppresses the chaperone/usher (CU) pathway of the gram-negative bacteria, and stimulates host macrophage autophagy in tuberculosis patients. NTZ also suppresses viral replication by inhibiting the maturation of the viral hemagglutinin and the viral transcription factor immediate early 2 (IE2) as well as by activating the eukaryotic translation initiation factor 2 (an antiviral intracellular protein). Lastly, NTZ exhibits an inhibitory effect on tumor cell progression by altering drug detoxification (glutathione-S-transferase P1), unfolded protein response, autophagy, anti-cytokines activity, and c-Myc inhibition.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
32
PharmaCompass offers a list of Nitazoxanide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Nitazoxanide manufacturer or Nitazoxanide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Nitazoxanide manufacturer or Nitazoxanide supplier.
A Celectan manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Celectan, including repackagers and relabelers. The FDA regulates Celectan manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Celectan API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Celectan manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Celectan supplier is an individual or a company that provides Celectan active pharmaceutical ingredient (API) or Celectan finished formulations upon request. The Celectan suppliers may include Celectan API manufacturers, exporters, distributors and traders.
click here to find a list of Celectan suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Celectan DMF (Drug Master File) is a document detailing the whole manufacturing process of Celectan active pharmaceutical ingredient (API) in detail. Different forms of Celectan DMFs exist exist since differing nations have different regulations, such as Celectan USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Celectan DMF submitted to regulatory agencies in the US is known as a USDMF. Celectan USDMF includes data on Celectan's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Celectan USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Celectan suppliers with USDMF on PharmaCompass.
A Celectan written confirmation (Celectan WC) is an official document issued by a regulatory agency to a Celectan manufacturer, verifying that the manufacturing facility of a Celectan active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Celectan APIs or Celectan finished pharmaceutical products to another nation, regulatory agencies frequently require a Celectan WC (written confirmation) as part of the regulatory process.
click here to find a list of Celectan suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Celectan as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Celectan API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Celectan as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Celectan and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Celectan NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Celectan suppliers with NDC on PharmaCompass.
Celectan Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Celectan GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Celectan GMP manufacturer or Celectan GMP API supplier for your needs.
A Celectan CoA (Certificate of Analysis) is a formal document that attests to Celectan's compliance with Celectan specifications and serves as a tool for batch-level quality control.
Celectan CoA mostly includes findings from lab analyses of a specific batch. For each Celectan CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Celectan may be tested according to a variety of international standards, such as European Pharmacopoeia (Celectan EP), Celectan JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Celectan USP).

