
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
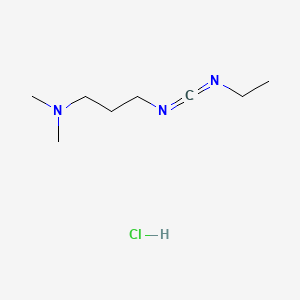

1. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride
1. 25952-53-8
2. Edc Hydrochloride
3. Edci
4. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride
5. Edap
6. Edc.hcl
7. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride
8. 7084-11-9
9. Edc-hcl Crosslinker
10. Wsc Hcl
11. N1-((ethylimino)methylene)-n3,n3-dimethylpropane-1,3-diamine Hydrochloride
12. Edac Hydrochloride
13. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hcl
14. Edac, Hydrochloride
15. N-ethyl-n'-(3-dimethylaminopropyl)carbodiimide Hydrochloride
16. Edcl
17. Edac-hcl
18. Edci Hcl
19. Wscd Hcl
20. Carbodiimide, Hydrochloride
21. Edac.hcl
22. Edc-hcl
23. Edci.hcl
24. Wsci.hcl
25. 3-(3-dimethylaminopropyl)-1-ethylcarbodiimide Hydrochloride
26. Mfcd00012503
27. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Monohydrochloride
28. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimidehydrochloride
29. 19w5tl0wj4
30. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hydrochloride
31. 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide Hydrochloride
32. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydrochloride
33. N-[3-(dimethylamino)propyl]-n'-ethylcarbodiimide Hydrochloride
34. ({[3-(dimethylamino)propyl]imino}methylidene)(ethyl)amine Hydrochloride
35. Ethyldimethylaminopropyl Carbodiimide Hydrochloride
36. N'-(ethylcarbonimidoyl)-n,n-dimethylpropane-1,3-diamine Monohydrochloride
37. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide, Hcl
38. 1,3-propanediamine, N'-(ethylcarbonimidoyl)-n,n-dimethyl-, Monohydrochloride
39. N1-((ethylimino)methylene)-n3,n3-dimethylpropane-1,3-diamine Xhydrochloride
40. Nsc-97064
41. (3-(dimethylamino)propyl)ethyl Carbodiimide Hydrochloride
42. N-(3-dimethylaminopropyl)-n-ethylcarbodiimide Hydrochloride
43. 1-ethyl-3-(3-dimethylaminopropyl) Carbodiimide Hydrochloride
44. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Monohydrochloride
45. Carbodiimide, (3-(dimethylamino)propyl)ethyl-, Monohydrochloride
46. N-(3-(dimethylamino)propyl)-n'-ethylcarbodiimide Hydrochloride
47. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride(chunks Or Pellets)
48. 1,3-propanediamine, N3-(ethylcarbonimidoyl)-n1,n1-dimethyl-, Hydrochloride (1:1)
49. 1-ethyl-3-(3-dimethylaminopropyl) Carbodiimide Hcl
50. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hcl
51. Wsc
52. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide Hcl
53. (3-dimethylamino-propyl)-ethyl-carbodiimide Hydrochloride
54. [3-(dimethylamino)propyl]ethyl Carbodiimide Hydrochloride
55. 1-ethyl-(3-dimethylaminopropyl)carbodiimide Hydrochloride
56. Ethyl-n'-(3-dimethylaminopropyl)carbodiimide Hydrochloride
57. N-ethyl-n'(3-dimethylaminopropyl) Carbodiimide Hcl
58. 1-(3-(dimethylamino)propyl)-3-ethylcarbodiimide Hydrochloride
59. 1-(3-dimethylaminopropyl)-3-ethyl-carbodiimide Hydrochloride
60. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide (hydrochloride)
61. 1-ethyl-3-(3-dimethylamino Propyl)carbodiimide Hydrochloride
62. 1-ethyl-3-[3-(dimethylamino)propyl]carbodiimide Hydrochloride
63. 3-[3-(dimethylamino)propyl]-1-ethylcarbodiimide Hydrochloride
64. N'-(3-dimethylaminopropyl)-n-ethylcarbodiimide Hydrochloride
65. N-ethyl-n-(3-dimethylaminopropyl) Carbodiimide Hydrochloride
66. 1-[3-(dimethylamino) Propyl]-3-ethylcarbodiimide Hydrochloride
67. N-ethyl-n'-[3-(dimethylamino)propyl]carbodiimide Hydrochloride
68. Edac Hcl
69. Edc (reagent)
70. Nsc97064
71. N-ethyl-n'-(dimethylaminopropyl)-carbodiimide Hcl
72. N-ethyl-n'-(3-dimethylaminopropyl)-carbodiimide Hcl
73. Water Soluble Carbodiimide
74. 1-(3-(dimethylamino)propyl)-3-ethylcarbodiimide Hcl
75. 1-[3-dimethylaminopropyl]-3-ethylcarbodiimide Hydrochloride
76. 1-ethyl-3-(3-dimethylaminopropyl]carbodiimide Hydrochloride
77. 1-ethyl-3-[3-dimethylaminopropyl]carbodiimide Hydrochloride
78. N-(dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride
79. N-[dimethylaminopropyl]-n'-ethylcarbodiimide Hydrochloride
80. N-ethyl-n-(3-dimethylaminopropyl)carbodiimide Hydrochloride
81. Unii-19w5tl0wj4
82. 1-(3(dimethylamino)propyl)-3-ethylcarbodiimide Hydrochloride
83. 1-(3-(dimethylamino) Propyl)-3-ethylcarbodiimide Hydrochloride
84. 1-(3-(dimethylamino)propyl)-3-ethyl Carbodiimide Hydrochloride
85. 1-(3-(dimethylamino)propyl)-3-ethyl-carbodiimide Hydrochloride
86. 1-(3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydrochloride
87. 1-(3-dimethylaminopropyl)-3-ethyl Carbodiimide Hydrochloride
88. 1-[3(dimethylamino)propyl]-3-ethylcarbodiimide Hydrochloride
89. 1-[3-(dimethyl-amino)propyl)-3-ethylcarbodiimide Hydrochloride
90. 1-[3-(dimethyl-amino)propyl]-3-ethylcarbodiimide Hydrochloride
91. 1-[3-(dimethylamino)propyl)-3-ethylcarbodiimide Hydrochloride
92. 1-[3-(dimethylamino)propyl)-3-ethylcarbodimide Hydrochloride
93. 1-[3-(dimethylamino)propyl]-3-ethyl Carbodiimide Hydrochloride
94. 1-[3-(dimethylamino)propyl]-3-ethyl-carbodiimide Hydrochloride
95. 1-[3-(dimethylamino)propyl]-3-ethylcarbodimide Hydrochloride
96. 1-[3-(dimethylamino)propyl}-3-ethylcarbodiimide Hydrochloride
97. 1-[3-[dimethylamino]propyl]-3-ethylcarbodiimide Hydrochloride
98. 1-[3-dimethylaminopropyl]-3-ethyl Carbodiimide Hydrochloride
99. 1-{3-(dimethylamino) Propyl}-3-ethylcarbodiimide Hydrochloride
100. 1-{3-(dimethylamino)propyl}-3-ethylcarbodiimide Hydrochloride
101. 1-ethyl-3-(3-(dimethylamino)propyl)-carbodiimide Hydrochloride
102. 1-ethyl-3-(3-(dimethylamino)propyl)carbodiimide Hydrochloride
103. 1-ethyl-3-[3-(dimethylamino)propyl]-carbodiimide Hydrochloride
104. N-(3-(dimethylamino)propyl)-n'ethylcarbodiimide Hydrochloride
105. N-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hcl; (edc Hcl)
106. N-[3-(dimethylamino)propyl]-n'ethylcarbodiimide Hydrochloride
107. N-[3-dimethylaminopropyl]-n'-ethylcarbodiimide Hydrochloride
108. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide Monohydrochloride
109. 1-ethyl-3-(3-(n,n-dimethylamino)propyl)-carbodiimide Hydrochloride
110. 1-ethyl-3-[3-(n,n-dimethylamino)propyl]-carbodiimide Hydrochloride
111. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride 25952-53-8
112. 3-(3-(dimethylamino)propyl)-1-ethylcarbodiimide Hydrochloride
113. Wsc Hydrochloride
114. Edci Hydrochloride
115. Wsci Hydrochloride
116. Edc - Hcl
117. Einecs 247-361-2
118. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide, Hydrochloride
119. Nsc 97064
120. Wscdi
121. Edc.hcl Crosslinker
122. Edac,hcl
123. Wsc.hcl
124. Schembl2761
125. Carbodiimide, Monohydrochloride
126. Edc Hydrochloride [mi]
127. Chembl1968770
128. Dtxsid50880076
129. Wln: 2ncn3n1&1 &gh
130. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Monohydrochloride
131. Act02727
132. Bcp15512
133. Bcp30024
134. Carbodiimide, (3-dimethylaminopropyl)ethyl-, Hydrochloride
135. Hy-d0178
136. (3-{[(ethylimino)methylidene]amino}propyl)dimethylamine Hydrochloride
137. 3-(ethyliminomethylideneamino)-n,n-dimethylpropan-1-amine Hydrochloride
138. 3-(ethyliminomethylideneamino)-n,n-dimethylpropan-1-amine,hydrochloride
139. 3-(((ethylimino)methylene)amino)-n,n-dimethylpropan-1-amine Hydrochloride
140. Akos005146190
141. Am83824
142. Cs-w008839
143. Gs-3161
144. Pid:816617
145. S12393
146. Bp-20545
147. Sy246358
148. Db-010822
149. 1-benzyl-5-oxopyrrolidine-3-carboxylic?acid
150. D1601
151. Ft-0607744
152. Ft-0647967
153. D70025
154. M02891
155. Z-1027
156. (3-dimethylaminopropyl)-3-ethylcarbodiimide Hcl
157. 1-(3-dimethylaminopropyl)3-ethylcarbodiimide Hcl
158. 1-ethyl-(3-dimethylaminopropyl) Carbodiimide Hcl
159. 1-ethyl-(3-dimethylaminopropyl)carbodiimide Hcl
160. 1-ethyl-3(3-dimethylaminopropyl)carbodiimide.hcl
161. Dimethylaminopropylethylcarbodiimide Hydrochloride
162. Ethyl Dimethylaminopropylcarbodimide Hydrochloride
163. Ethyldimethylaminopropylcarbodiimide Hydrochloride
164. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide-hcl
165. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide.hcl
166. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide-hcl
167. Ethyl Dimethylaminopropyl Carbodiimide Hydrochloride
168. Ethyl(dimethylaminopropyl)carbodiimide Hydrochloride
169. Ethyl-dimethylaminopropyl-carbodiimide Hydrochloride
170. N-(3-dimethylaminopropyl)-n'-ethylcarbodimide Hcl
171. N-(3-dimethylaminopropyl)-n'-ethylcarbodimide-hcl
172. N-ethyldimethylaminopropylcarbodiimide Hydrochloride
173. 1-(3- Dimethylaminopropyl)-3-ethylcarbodiimide Hcl
174. 1-(3-dimethylaminopropyl) -3-ethylcarbodiimide-hcl
175. 1-(3-dimethylaminopropyl)-3-ethyl Carbodiimide Hcl
176. 1-(3-dimethylaminopropyl)-3-ethyl-carbodiimide Hcl
177. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hcl
178. 1-ethyl-3-(3-dimethylaminopropyl) Carbodiimide-hcl
179. 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide Hcl
180. 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide-hcl
181. Dimethylaminopropyl-3-ethylcarbodiimide Hydrochloride
182. J-670019
183. J-801018
184. N-(3-dimethylamino-propyl)-n'-ethylcarbodiimide Hcl
185. N-(3-dimethylaminopropyl)-n'-ethyl Carbodiimide Hcl
186. N-(3-dimethylaminopropyl)-n'-ethyl-carbodiimide Hcl
187. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hcl
188. (3-dimethylaminopropyl)-ethylcarbodiimide Hydrochloride
189. (3-dimethylaminopropyl)ethyl-carbodiimid Hydrochloride
190. (3-dimethylaminopropyl)ethyl-carbodiimide Hydrochloride
191. (3-dimethylaminopropyl)ethylcarbodiimide Hydrochloride
192. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide-hcl
193. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide.hcl
194. 1-ethyl(dimethylaminopropyl)carbodiimide Hydrochloride
195. 1-ethyl-(3-dimethylaminopropyl)carbodiimide Hcl Salt
196. 1-ethyl-(3-dimethylaminopropyl)carbodimidehydrochloride
197. 1-ethyl-(dimethylaminopropyl)carbodiimide Hydrochloride
198. 1-ethyl-3-(3'-dimethylaminopropyl) Carbodiimide Hcl
199. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide, Hcl
200. 1-ethyl-3-dimethylaminopropylcarbodiimide Hydrochloride
201. 3-dimethylaminopropyl 3-ethylcarbodiimide Hydrochloride
202. 3-dimethylaminopropyl Ethyl Carbodiimide Hydrochloride
203. 3-dimethylaminopropyl-3-ethylcarbodiimide Hydrochloride
204. 3-dimethylaminopropyl-n-ethylcarbodiimide Hydrochloride
205. Ethyl-(3-dimethylaminopropyl)carbodiimide Hydrochloride
206. Ethyldimethylaminopropylcarbodiimide Hydrochloride Salt
207. Q55444948
208. (3-(dimethylamino)propyl)ethylcarbodimide Hydrochloride
209. (3-dimethylamino-propyl)-ethyl Carbodiimide Hydrochloride
210. (3-dimethylamino-propyl)-ethyl-carbodiimid Hydrochloride
211. (3-dimethylamino-propyl)-ethyl-carbodiimide.hydrochloride
212. (3-dimethylamino-propyl)-ethylcarbodiimide Hydrochloride
213. (3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
214. (3-dimethylaminopropyl)-ethyl-carbodiimide Hydrochloride
215. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hcl Salt
216. 1-(3-dimethylaminopropyl)3-ethylcarbodimide Hydrochloride
217. 1-(3-dimethylaminopropyl)3ethylcarbodiimide Hydrochloride
218. 1-(3dimethylaminopropyl)-3ethylcarbodiimide Hydrochloride
219. 1-(dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
220. 1-ethyl-(3-dimethylaminopropyl)carbodiimidehydrochloride
221. 1-ethyl-(3dimethylaminopropyl)-carbodiimide Hydrochloride
222. 1-ethyl-3(3-dimethylaminopropyl)carbodiimidehydrochloride
223. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hcl Salt
224. 1-ethyl-3-(dimethylaminopropyl)-carbodimide Hydrochloride
225. 1-ethyl-3-(dimethylaminopropyl)carbodiimide Hydrochloride
226. 1-ethyl-3-(dimethylaminopropyl)carbodimide Hydrochloride
227. 1-ethyl-3-dimethylaminopropyl Carbodiimide Hydrochloride
228. 1-ethyl-3-dimethylaminopropyl-carbodiimide Hydrochloride
229. 1-ethyl3-(dimethylaminopropyl)carbodiimide Hydrochloride
230. 3-(dimethylamino)propyl-/v-ethylcarbodimide Hydrochloride
231. 3-(dimethylamino)propyl-3-ethylcarbodiimide Hydrochloride
232. 3-(dimethylamino)propyl-n'-ethylcarbodimide Hydrochloride
233. 3-(dimethylamino)propyl-n-ethylcarbodimide Hydrochloride
234. 3-dimethylaminopropyl-n'-ethylcarbodiimide Hydrochloride
235. Ethyl-3(3-dimethylaminopropyl)carbodiimide Hydrochloride
236. Ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride
237. F0001-0862
238. N-(3-dimethylaminopropyl)-ethylcarbodiimide Hydrochloride
239. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hcl Salt
240. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimidhydrochlorid
241. N-dimethylaminopropyl-n'-ethylcarbodiimide Hydrochloride
242. N-ethyl-n'-(3-dimethylaminopropyl)-carbodiimide . Hcl
243. N-ethyl-n'-dimethylaminopropyl-carbodiimide Hydrochloride
244. N-ethyl-n'-dimethylaminopropylcarbodiimide Hydrochloride
245. (3-dimethylamino-propyl)-ethyl-carbodiimide, Hydrochloride
246. (3-dimethylaminopropyl)-3-ethyl Carbodiimide Hydrochloride
247. [3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydrochloride
248. 1(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
249. 1-(3 Dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
250. 1-(3-dimehtylaminopropyl)-3-ethylcarbodiimide Hydrochloride
251. 1-(3-dimethlaminopropyl) -3-ethylcarbodiimide Hydrochloride
252. 1-(3-dimethlaminopropyl)-3-ethylcarbodiimide Hydrochloride
253. 1-(3-dimethyaminopropyl)-3-ethylcarbodiimide Hydrochloride
254. 1-(3-dimethylaminopropy)-3-ethyl-carbodiimide Hydrochloride
255. 1-(3-dimethylaminopropyi)-3-ethylcarbodiimide Hydrochloride
256. 1-(3-dimethylaminopropyl) 3-ethylcarbodiimide Hydrochloride
257. 1-(3-dimethylaminopropyl)-3-ethyl Carbodimide Hydrochloride
258. 1-(3-dimethylaminopropyl)-3-ethyl-carbodiimid Hydrochloride
259. 1-(3-dimethylaminopropyl)-3-ethyl-carbodiimidhydrochlorid
260. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimid Hydrochloride
261. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydochloride
262. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrocloride
263. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide-hydrochloride
264. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide.hydrochloride
265. 1-(3-dimethylaminopropyl)-3-ethylcarbodimide Hydrochloride
266. 1-(3-dimethylaminopropyl)-3ethyl Carbodiimide Hydrochloride
267. 1-(3-dimethylaminopropyl)-3ethylcarbodiimide Hydrochloride
268. 1-(3-dimethylaminopropyl)3-ethyl Carbodiimide Hydrochloride
269. 1-(3-dimethylaminopropyl)3-ethyl-carbodiimide Hydrochloride
270. 1-(3-dimethylaminopropyl)3-ethylcarbodiimide Hydrochloride
271. 1-(3-dimethylaminoproyl)-3-ethylcarbodiimide Hydrochloride
272. 1-(3dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
273. 1-(dimethylaminopropyl)-3-(ethyl)carbodiimide Hydrochloride
274. 1-ethy1-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride
275. 1-ethyl-(3-dimethylamino Propyl) Carbodiimide Hydrochloride
276. 1-ethyl-(3-dimethylamino Propyl)carbodiimide Hydrochloride
277. 1-ethyl-(3-dimethylamino-propyl) Carbodiimide Hydrochloride
278. 1-ethyl-(3-dimethylamino-propyl)carbodiimide Hydrochloride
279. 1-ethyl-(3-dimethylaminopropyl) Carbodiimide Hydrochloride
280. 1-ethyl-(3-dimethylaminopropyl)-carbodiimide Hydrochloride
281. 1-ethyl-3 (3-dimethylaminopropyl)carbodiimide Hydrochloride
282. 1-ethyl-3(3-dimethylaminopropyl) Carbodiimide Hydrochloride
283. 1-ethyl-3(3-dimethylaminopropyl)carbodiimide Hydrochloride
284. 1-ethyl-3-(3 Dimethylaminopropyl)carbodiimide Hydrochloride
285. 1-ethyl-3-(3-dimethlaminopropyl) Carbodiimide Hydrochloride
286. 1-ethyl-3-(3-dimethyaminopropyl)carbodiimide Hydrochloride
287. 1-ethyl-3-(3-dimethylamino-propyl)carbodiimidehydrochloride
288. 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimidehydrochloride
289. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimid Hydrochloride
290. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrocloride
291. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide-hydrochloride
292. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide.hydrochloride
293. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimidehydrochloride
294. 1-ethyl-3-(3-dimethylaminopropyl)carbodimide Hydrochloride
295. 1-ethyl-3-(3dimethylaminopropyl) Carbodiimide Hydrochloride
296. 1-ethyl-3-(3dimethylaminopropyl)-carbodiimide Hydrochloride
297. 1-ethyl-3-(3dimethylaminopropyl)carbodiimide Hydro-chloride
298. 1-ethyl-3-(3dimethylaminopropyl)carbodiimide Hydrochloride
299. 1-ethyl-3-(dimethylaminopropyl)-carbodiimide Hydrochloride
300. 1ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride
301. 3-(3-dimethylaminopropyl)-1-ethylcarbodimide Hydrochloride
302. 3-(dimethylamino) Propyl-3-ethylcarbodiimide Hydrochloride
303. 3-(dimethylamino)propyl-3-ethyl Carbodiimide Hydrochloride
304. 3-ethyl-1-(3-dimethylaminopropyl)carbodiimide Hydrochloride
305. Ethyl-(n',n'-dimethylamino)propyl-carbodimide-hydrochloride
306. Ethyl-3-(3' Dimethylaminopropyl)carbodiimide Hydrochloride
307. Ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hydrochloride
308. Ethyl-3-(3-dimethylaminopropyl) Carbodiimide Hydrochloride
309. L-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride
310. N'-(3-dimethylaminopropyl)-n-ethylcarbodiimid Hydrochloride
311. N'-(3-dimethylaminopropyl)-n-ethylcarbodiimide-hydrochloride
312. N'-(3-dimethylaminopropyl)-n-ethylcarbodiimide.hydrochloride
313. N'-(3-dimethylaminopropyl)-n-ethylcarbodimide Hydrochloride
314. N(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride
315. N- [dimethylaminopropyl]-n'-ethylcarbodiimide Hydrochloride
316. N-(3-dimehtylaminopropyl)-n'-ethylcarbodiimide Hydrochloride
317. N-(3-dimethyaminopropyl)-n'-ethylcarbodiimide Hydrochloride
318. N-(3-dimethylaminopropy)-n'-ethylcarbodiimide Hydrochloride
319. N-(3-dimethylaminopropyl)-n -ethylcarbodiimide Hydrochloride
320. N-(3-dimethylaminopropyl)-n Ethylcarbodiimide Hydrochloride
321. N-(3-dimethylaminopropyl)-n'-ethyl-carbodiimid-hydrochlorid
322. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimid Hydrochloride
323. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide-hydrochloride
324. N-(3-dimethylaminopropyl)-n'-ethylcarbodimide Hydrochloride
325. N-(3-dimethylaminopropyl)-n'ethylcarbodiimide Hydrochloride
326. N-(3-dimethylaminopropyl)-n-ethylcarbodimide Hydrochloride
327. N-(3dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride
328. N-[dimethylaminopropyl]-n'-ethyl-carbodiimide Hydrochloride
329. N-ethyl-n'-(3-dimethylaminopropyl)carbodiimide-hydrochloride
330. N-ethyl-n'-(3-dimethylaminopropyl)carbodiimidehydrochloride
331. N-ethyl-n'-(3-dimethylaminopropyl)carbodimide Hydrochloride
332. N-ethyl-n'-(dimethylaminopropyl) Carbodiimide Hydrochloride
333. N-ethyl-n'-(dimethylaminopropyl)-carbodiimide Hydrochloride
334. N-ethyl-n'-(dimethylaminopropyl)carbodiimide Hydrochloride
335. N-ethyl-n-(3-dimethylaminopropyl)carbodimide Hydrochloride
336. (1-(3-dimethylaminopropyl)-3-ethylcarbodiimide).hydrochloride
337. (3-dimethylamino-propyl)-ethyl-carbodiimide Hydrogen Chloride
338. 1 -(3-dimethylaminopropyl)-3-ethyl-carbodiimide Hydrochloride
339. 1 -(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
340. 1 -ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hydrochloride
341. 1, N'-(ethylcarbonimidoyl)-n,n-dimethyl-, Monohydrochloride
342. 1- (3-dimethylaminopropyl) -3-ethylcarbodiimide Hydrochloride
343. 1- (3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
344. 1- [3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydrochloride
345. 1- Ethyl-3-[3-dimethylaminopropyl]carbodiimide Hydrochloride
346. 1-(-3-dimethylamino-propyl)-3-ethylcarbodiimide Hydrochloride
347. 1-(-3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
348. 1-(3 -dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
349. 1-(3 Dimethylaminopropyl)-3-ethyl Carbodiimide Hydrochloride
350. 1-(3'-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
351. 1-(3(dimethylamino) Propyl)-3-ethylcarbodiimide Hydrochloride
352. 1-(3- Dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
353. 1-(3-di-methylaminopropyl)-3-ethylcarbodiimide Hydrochloride
354. 1-(3-dimethylamino Propyl)-3-ethyl Carbodiimide Hydrochloride
355. 1-(3-dimethylamino Propyl)-3-ethylcarbodiimide Hydrochloride
356. 1-(3-dimethylamino-propy)-3-ethyl-carbodiimide Hydrochloride
357. 1-(3-dimethylamino-propyl) -3-ethylcarbodiimide Hydrochloride
358. 1-(3-dimethylamino-propyl)-3 -ethylcarbodiimide Hydrochloride
359. 1-(3-dimethylamino-propyl)-3-ethyl-carbodiimide Hydrochloride
360. 1-(3-dimethylamino-propyl)-3-ethylcarbodiimide Hydrochloride
361. 1-(3-dimethylamino-propyl)-3-ethylcarbodiimide, Hydrochloride
362. 1-(3-dimethylaminopropyl) -3-ethylcarbodiimide Hydrochloride
363. 1-(3-dimethylaminopropyl) 3-ethyl Carbodiimide Hydrochloride
364. 1-(3-dimethylaminopropyl)- 3-ethylcarbodiimide Hydrochloride
365. 1-(3-dimethylaminopropyl)-3 -ethylcarbodiimide Hydrochloride
366. 1-(3-dimethylaminopropyl)-3- Ethylcarbodiimide Hydrochloride
367. 1-(3-dimethylaminopropyl)-3-ethylcarbo-diimide Hydrochloride
368. 1-(3-dimethylaminopropyl)-3-ethylcarbodi-imide Hydrochloride
369. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydro-chloride
370. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide, Hydrochloride
371. 1-(3-dimethylaminopropyl)3-ethylcarbodiimide Hydrochloride Salt
372. 1-(3-dimethylarninopropyl)-3-ethylcarbodiimide Hydrochloride
373. 1-(n,n-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride
374. 1-[3-(dimethlamino)propyl]-3-ethylcarbodiimide Hydrochloride
375. 1-[3-(dimethyamino)propyl]-3-ethylcarbodiimide Hydrochloride
376. 1-[3-(dimethyl-amino)propyl]-3ethylcarbodiimide Hydrochloride
377. 1-[3-(dimethylamino) Propyl]-3-ethylcarbodimide Hydrochloride
378. 1-[3-(dimethylamino)-propyl]-3-ethylcarbodiimide Hydrochloride
379. 1-[3-(dimethylamino)-propyl]-3-ethylcarbodimide Hydrochloride
380. 1-[3-(dimethylamino)propyl]-3 -ethylcarbodiimide Hydrochloride
381. 1-[3-(dimethylamino)propyl]-3-ethylcarbo-diimide Hydrochloride
382. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydro-chloride
383. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydrocloride
384. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide, Hydrochloride
385. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide-hydrochloride
386. 1-[3-(dimethylamino)propyl]3-ethylcarbodiimide Hydrochloride
387. 1-ethyl -3-(3-dimethylaminopropyl) Carbodiimide Hydrochloride
388. 1-ethyl-3(3'-dimethylaminopropyl) Carbodiimide Hydrochloride
389. 1-ethyl-3- (3-dimethylaminopropyl ) Carbodiimide Hydrochloride
390. 1-ethyl-3- (3-dimethylaminopropyl)carbodiimide Hydrochloride
391. 1-ethyl-3-(3 '-dimethylaminopropyl)carbodiimide Hydrochloride
392. 1-ethyl-3-(3 Dimethylaminopropyl) Carbodiimide Hydrochloride
393. 1-ethyl-3-(3'- Dimethylaminopropyl)carbodiimide Hydrochloride
394. 1-ethyl-3-(3'-dim Ethylaminopropyl)carbodiimide Hydrochloride
395. 1-ethyl-3-(3'-dimethyl Aminopropyl)-carbodiimide.hydrochloride
396. 1-ethyl-3-(3'-dimethylamino-propyl)carbodiimide Hydrochloride
397. 1-ethyl-3-(3'-dimethylaminopropyl) Carbodiimide Hydrochloride
398. 1-ethyl-3-(3'-dimethylaminopropyl)-carbodiimide Hydrochloride
399. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide, Hydrochloride
400. 1-ethyl-3-(3- Dimethylaminopropyl)carbodiimide Hydrochloride
401. 1-ethyl-3-(3-di-methylaminopropyl)carbodiimide Hydrochloride
402. 1-ethyl-3-(3-dim Ethylaminopropyl)carbodiimide Hydrochloride
403. 1-ethyl-3-(3-dimethyl-aminopropyl)-carbodiimide Hydrochloride
404. 1-ethyl-3-(3-dimethylamino Propyl) Carbodiimide Hydrochloride
405. 1-ethyl-3-(3-dimethylamino Propyl)-carbodiimide.hydrochloride
406. 1-ethyl-3-(3-dimethylamino- Propyl)carbodiimide Hydrochloride
407. 1-ethyl-3-(3-dimethylamino-propyl) Carbodiimide Hydrochloride
408. 1-ethyl-3-(3-dimethylamino-propyl)carbodiimide Hydro-chloride
409. 1-ethyl-3-(3-dimethylamino-propyl)carbodiimide Hydrochloride
410. 1-ethyl-3-(3-dimethylamino-propyl)carbodiimide, Hydrochloride
411. 1-ethyl-3-(3-dimethylaminoprop Yl) Carbodiimide Hydrochloride
412. 1-ethyl-3-(3-dimethylaminoprop-1-yl)carbodiimide Hydrochloride
413. 1-ethyl-3-(3-dimethylaminopropyl )carbodiimide Hydrochloride
414. 1-ethyl-3-(3-dimethylaminopropyl) -carbodiimide Hydrochloride
415. 1-ethyl-3-(3-dimethylaminopropyl)- Carbodiimide Hydrochloride
416. 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide-hydrochloride
417. 1-ethyl-3-(3-dimethylaminopropyl)carbodi-imide Hydrochloride
418. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydro-chloride
419. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloric Acid
420. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride-
421. 1-ethyl-3-(n,n-dimethylaminopropyl) Carbodiimide Hydrochloride
422. 1-ethyl-3-(n,n-dimethylaminopropyl)carbodiimide Hydrochloride
423. 1-ethyl-3-[(3-(dimethylamino)propyl]carbodiimide Hydrochloride
424. 1-ethyl-3-[3'-(dimethylamino)propyl]carbodiimide Hydrochloride
425. 1-ethyl-3-[3-(dimethylamino) Propyl]carbodiimide Hydrochloride
426. 1-ethyl-3-[3-(dimethylamino)-propyl]carbodiimide Hydrochloride
427. 1-ethyl-3-[3-(dimethylamino)proply]carbodiimide Hydrochloride
428. 1-ethyl-3-[3-(dimethylamino)propyl] Carbodiimide Hydrochloride
429. 1-ethyl-3-[3-(dimethylamino)propyl]-carbodiimidehydrochloride
430. 1-ethyl-3-[3-(dimethylamino)propyl]carbodiimide.hydrochloride
431. 1-ethyl-n'-(3-dimethylaminopropyl)-carbodiimide Hydrochloride
432. 1[3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydrochloride
433. 3-(3-dimethylamino-propyl)-1-ethylcarbodiimide Hydrochloride
434. 3-(3-dimethylaminopropyl)-1-ethyl-carbodiimide Hydrochloride
435. 3-(3-dimethylaminopropyl)-alpha-ethylcarbodiimide Hydrochloride
436. 3-(cyano-ethyl-amino)propyl-dimethyl-azanium Chloride
437. 3-ethyl-1-(3-dimethylaminopropyl)-carbodiimide Hydrochloride
438. 3-ethyl-1-[3-(dimethylamino)propyl]carbodiimide Hydrochloride
439. Carbodiimide, (3-dimethylaminopropyl)ethyl-, Monohydrochloride
440. Ethyl-(n',n'-dimethyl Amino)propylcarbodiimide Hydrochloride
441. Hydrochloric Acid 1-(dimethylaminopropyl)-3-ethylcarbodiimide
442. Hydrochloric Acid 1-ethyl-(3-dimethylaminopropyl)carbodiimide
443. Hydrochloric Acid 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide
444. L-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hydrochloride
445. L-ethyl-3-(3-dimethylaminopropyl) Carbodiimide Hydrochloride
446. N'-(3 Dimethylaminopropyl)-n-ethyl-carbodiimide Hydrochloride
447. N- (3-dimethylaminopropyl)-n'-ethyl-carbodiimide Hydrochloride
448. N-(-3-dimethylaminopropyl)-n'-ethyl-carbodiimide-hydrochloride
449. N-(-3-dimethylaminopropyl)-n'-ethylcarbodiimid Hydrochloride
450. N-(3- Dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride
451. N-(3-dim Ethylaminopropyl)-n-ethylcarbodiimide Hydrochloride
452. N-(3-dimethylamino-propyl)-n'-ethyl Carbodiimide Hydrochloride
453. N-(3-dimethylamino-propyl)-n'-ethyl-carbodiimide Hydrochloride
454. N-(3-dimethylamino-propyl)-n'-ethyl-carbodiimide-hydrochloride
455. N-(3-dimethylamino-propyl)-n'-ethylcarbodiimid Hydrochloride
456. N-(3-dimethylamino-propyl)-n'-ethylcarbodiimide Hydrochloride
457. N-(3-dimethylamino-propyl)-n'-ethylcarbodiimide, Hydrochloride
458. N-(3-dimethylaminopropyl) -n'-ethylcarbodiimide Hydrochloride
459. N-(3-dimethylaminopropyl) N'-ethylcarbodiimide Hydrochloride
460. N-(3-dimethylaminopropyl)- N'-ethylcarbodiimide Hydrochloride
461. N-(3-dimethylaminopropyl)-n '-ethylcarbodiimide Hydrochloride
462. N-(3-dimethylaminopropyl)-n' Ethylcarbodiimide Hydrochloride
463. N-(3-dimethylaminopropyl)-n'-ethyl Carbodiimide Hydrochloride
464. N-(3-dimethylaminopropyl)-n'-ethyl-carbodiimid Hydrochloride
465. N-(3-dimethylaminopropyl)-n'-ethyl-carbodiimid-hydrochloride
466. N-(3-dimethylaminopropyl)-n'-ethyl-carbodiimide Hydrochloride
467. N-(3-dimethylaminopropyl)-n'-ethyl-carbodiimide-hydrochloride
468. N-(3-dimethylaminopropyl)-n'-ethylcarbo-diimide Hydrochloride
469. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydro-chloride
470. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide, Hydrochloride
471. N-(3-dimethylaminopropyl)-n'-ethylcarbodimide Hydro Chloride
472. N-(3dimethylaminopropyl)-n'- Ethylcarbodiimide Hydrochloride
473. N-(dimethylamino-propyl)-n'-ethyl-carbodiimide Hydrochloride
474. N-[(dimethylamino)propyl]-n'-ethylcarbodiimide Hydrochloride
475. N-[3-(dimethylamino)-propyl]-n'-ethylcarbodiimide.hydrochloride
476. N-[3-(dimethylamino)propyl]-n'-ethylcarbodiimid Hydrochloride
477. N-[3-(dimethylamino)propyl]-n'-ethylcarbodiimide-hydrochloride
478. N-[3-(dimethylamino)propyl]-n'-ethylcarbodiimide.hydrochloride
479. N-[3-(dimethylamino)propyl]-n'-ethylcarbodiimidehydrochloride
480. N-[3-(dimethylamino)propyl]n'-ethylcarbodiimide Hydrochloride
481. N-ethyl-/v-(3-dimethylaminopropyl)carbodiimide Hydrochloride
482. N-ethyl-n'-(3-dimethylamino-propyl)-carbodiimide Hydrochloride
483. N-ethyl-n'-(3-dimethylaminopro Pyl)carbodiimide Hydrochloride
484. N-ethyl-n'-(3-dimethylaminoprop-1-yl)carbodiimide Hydrochloride
485. N-ethyl-n'-(3-dimethylaminopropyl) Carbodiimide Hydrochloride
486. N-ethyl-n'-(3-dimethylaminopropyl)-carbo Diimide Hydrochloride
487. N-ethyl-n'-(3-dimethylaminopropyl)-carbodiimid Hydrochloride
488. N-ethyl-n'-(3-dimethylaminopropyl)-carbodiimide Hydrochloride
489. N-ethyl-n'-(3-dimethylaminopropyl)-carbodimide Hydrochloride
490. N-ethyl-n'-(3-dimethylaminopropyl)carbodimide, Hydrochloride
491. N-ethyl-n'-3-(n,n-dimethylamino)propylcarbodimide Hydrochloride
492. 1-(3-dimethylamino-propyl)-3-ethylcarbodiimide Monohydrochloride
493. 1-(3-dimethylaminopropyl)-3-ethyl Carbodiimide Hydrochloride Salt
494. 1-(3-dimethylaminopropyl)-3-ethyl-carbodiimide Hydrochloride Salt
495. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloric Acid Salt
496. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride (edci)
497. 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide Hydrochloride Salt
498. 1-[3-(dimethylamino) Propyl]-3-ethyl-carbodiimide Hydrochloride
499. 1-[3-(dimethylamino)-propyl]-3-ethyl-carbodiimide Hydrochloride
500. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydrochloride Salt
501. 1-[3-(dimethylamino)propyl]3-ethylcarbodiimide Monohydrochloride
502. 1-ethyl-3-(3 -dimethylaminopropyl)carbodiimide Hydrochloride Salt
503. 1-ethyl-3-(3'-dimethylaminopropyi)carbodiimide Hydrogen Chloride
504. 1-ethyl-3-(3'-dimethylaminopropyl) Carbodiimide Hydrogen Chloride
505. 1-ethyl-3-(3'-dimethylaminopropyl)-carbodiimide Monohydrochloride
506. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hydrochloric Acid
507. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hydrochloride Salt
508. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Hydrogen Chloride
509. 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide Mono-hydrochloride
510. 1-ethyl-3-(3-dimethylaminopropyl) Carbodiimide Hydrochloric Acid
511. 1-ethyl-3-(3-dimethylaminopropyl) Carbodiimide Hydrochloride Salt
512. 1-ethyl-3-(3-dimethylaminopropyl) Carbodiimide Monohydrochloride
513. 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide Hydrochloride Salt
514. 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide Hydrogen Chloride
515. 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide Monohydrochloride
516. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloric Acid Salt
517. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride Salt
518. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrogen Chloride Salt
519. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Mono-hydrochloride
520. 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Monohydro-chloride
521. 1-ethyl-3-[3-(dimethylamino)propyl]-carbodiimide Monohydrochloride
522. 1-ethyl-3-[3-(dimethylamino)propyl]carbodiimide Monohydrochloride
523. 1-ethyl-3-[3-(n,n-dimethylamino) Propyl]carbodiimide Hydrochloride
524. 1-ethyl-3-[3-(n,n-dimethylamino)propyl]carbodiimide Hydrochloride
525. 3-(3-dimethylaminopropyl)-1-ethylcarbodiimide Hydrochloric Acid Salt
526. L-ethyl-3-(3-dimethylaminopropyl)carbodiimide Hydrochloride Salt
527. N-(3-dimethylamino-1-propyl)-n'-ethylcarbodiimide Hydrochloride
528. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Monohydrochloride
529. N-(3-dimethylaminopropyl)-n\'-ethylcarbodiimide Hydrochloride
530. N-[3-(dimethylamino) Propyl]-n'-ethylcarbodiimide Hydrochloride
531. N-[3-(dimethylamino)propyl]-n'-ethylcarbodiimide Monohydrochloride
532. N-ethyl-n'-(3-dimethylaminopropyl)carbodiimide Hydrochloride, 98%
533. N-ethyl-n'-3-(n,n-dimethylamino)propylcarbodiimide Hydrochloride
534. N-ethyl-n'-3-(n,n-dimethylamino)propylcarbodiimide.hydrochloride
535. 1-(3-(n,n-dimethylamino)-prop-1-yl)-3-ethylcarbodiimide Hydrochloride
536. 1-[3-(dimethylamino) Propyl]-3-ethylcarbodiimide Hydrochloric Acid Salt
537. 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide Hydrochloric Acid Salt
538. 1-ethyl-3-(3-(dimethylamino)propyl)carbodiimide Hydrochloric Acid Salt
539. 3-(((ethylimino)methylene)amino)-n,n-dimethylpropan-l-aminium Chloride
540. 3-(ethyliminomethyleneamino)-n,n-dimethyl-propan-1-amine Hydrochloride
541. 3-(ethyliminomethylideneamino)-n,n-dimethyl-propane-1-aminehydrochloride
542. 3-{[(ethylimino)-methylene]amino}-n,n-dimethylpropan-1-aminium Chloride
543. 3-{[(ethylimino)methylene]amino}-n,n-dimethylpropan-1-aminium Chloride
544. 3-{[(ethylimino)methylene]amino}-n,n-dimethylpropane-1-aminium Chloride
545. N-((ethylimino)methylene)-n3,n3-dimethylpropane-1,3-diamine Hydrochloride
546. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloric Acid Salt
547. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride (edac Hcl)
548. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride, Bioxtra
549. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride, Crystalline
550. N1-((ethylimino)methylene)-n3, N3-dimethylpropane-1,3-diamine Hydrochloride
551. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride (edc) 98%, >=98%
552. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride, >=99.0% (at)
553. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride, Commercial Grade, Powder
554. N-(3-dimethylaminopropyl)-n'-ethylcarbodiimide Hydrochloride, Purum, >=98.0% (at)
555. N1-((ethylimino)-methylene)-n3,n3-dimethylpropane-1,3-diamine Hydrochloride
556. N1-((ethylimino)methylene)-n3 ,n3-dimethylpropane-1,3-diamine Hydrochloride
557. N1-((ethylimino)methylene)-n3,n3-dimethylpropane-1,3-di Amine Hydrochloride
558. N1-((ethylimino)methylene)-n3,n3-dimethylpropane-1,3-diamine Hydrogen Chloride Salt
559. 93128-40-6
560. Edac . Hcl;edc . Hcl;n-(3-dimethylaminopropyl)-n'-ethylcarbodiimide . Hcl;water-soluble Carbodiimide;
| Molecular Weight | 191.70 g/mol |
|---|---|
| Molecular Formula | C8H18ClN3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 5 |
| Exact Mass | 191.1189253 g/mol |
| Monoisotopic Mass | 191.1189253 g/mol |
| Topological Polar Surface Area | 28 Ų |
| Heavy Atom Count | 12 |
| Formal Charge | 0 |
| Complexity | 134 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
ABOUT THIS PAGE
62
PharmaCompass offers a list of Carbodiimide, Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Carbodiimide, Hydrochloride manufacturer or Carbodiimide, Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Carbodiimide, Hydrochloride manufacturer or Carbodiimide, Hydrochloride supplier.
A Carbodiimide, Hydrochloride manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Carbodiimide, Hydrochloride, including repackagers and relabelers. The FDA regulates Carbodiimide, Hydrochloride manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Carbodiimide, Hydrochloride API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Carbodiimide, Hydrochloride supplier is an individual or a company that provides Carbodiimide, Hydrochloride active pharmaceutical ingredient (API) or Carbodiimide, Hydrochloride finished formulations upon request. The Carbodiimide, Hydrochloride suppliers may include Carbodiimide, Hydrochloride API manufacturers, exporters, distributors and traders.
click here to find a list of Carbodiimide, Hydrochloride suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Carbodiimide, Hydrochloride DMF (Drug Master File) is a document detailing the whole manufacturing process of Carbodiimide, Hydrochloride active pharmaceutical ingredient (API) in detail. Different forms of Carbodiimide, Hydrochloride DMFs exist exist since differing nations have different regulations, such as Carbodiimide, Hydrochloride USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Carbodiimide, Hydrochloride DMF submitted to regulatory agencies in the US is known as a USDMF. Carbodiimide, Hydrochloride USDMF includes data on Carbodiimide, Hydrochloride's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Carbodiimide, Hydrochloride USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Carbodiimide, Hydrochloride suppliers with USDMF on PharmaCompass.
Carbodiimide, Hydrochloride Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Carbodiimide, Hydrochloride GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Carbodiimide, Hydrochloride GMP manufacturer or Carbodiimide, Hydrochloride GMP API supplier for your needs.
A Carbodiimide, Hydrochloride CoA (Certificate of Analysis) is a formal document that attests to Carbodiimide, Hydrochloride's compliance with Carbodiimide, Hydrochloride specifications and serves as a tool for batch-level quality control.
Carbodiimide, Hydrochloride CoA mostly includes findings from lab analyses of a specific batch. For each Carbodiimide, Hydrochloride CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Carbodiimide, Hydrochloride may be tested according to a variety of international standards, such as European Pharmacopoeia (Carbodiimide, Hydrochloride EP), Carbodiimide, Hydrochloride JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Carbodiimide, Hydrochloride USP).

