
Synopsis
Synopsis
0
KDMF
0
VMF
Annual Reports
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
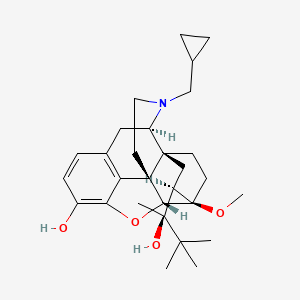

1. 6029 M
2. 6029-m
3. 6029m
4. Buprenex
5. Buprenorphine Hydrochloride
6. Buprex
7. Hydrochloride, Buprenorphine
8. Prefin
9. Rx 6029 M
10. Rx-6029-m
11. Rx6029m
12. Subutex
13. Temgsic
14. Temgesic
1. Temgesic
2. Probuphine
3. Buprenex
4. 52485-79-7
5. Buprenophine
6. Buprenorfina
7. Buprenorphinum
8. Butrans
9. Buprenorfina [inn-spanish]
10. Buprenorphinum [inn-latin]
11. (-)-buprenorphine
12. Buprenorphin
13. Sublocade
14. Chebi:3216
15. Dea No. 9064
16. 40d3scr4gz
17. Cam2038
18. 21-cyclopropyl-7alpha-[(s)-1-hydroxy-1,2,2-trimethylpropyl]-6,14-endo-ethano-6,7,8,14-tetrahydrooripavine
19. Rx 6029m
20. Brixadi
21. 17-cyclopropylmethyl-4,5alpha-epoxy-7alpha-((s)-1-hydroxy-1,2,2-trimethylpropyl)-6-methoxy-6,14-endo-ethanomorphinan-3-ol
22. 2-(n-cyclopropylmethyl-4,5alpha-epoxy-3-hydroxy-6-methoxy-6,14-endo-ethanomorphinan-6alpha-yl)-3,3-dimethyl-2-butanol
23. Probuphenine
24. Temgesic (tn)
25. (1s,2s,6r,14r,15r,16r)-5-(cyclopropylmethyl)-16-[(2s)-2-hydroxy-3,3-dimethylbutan-2-yl]-15-methoxy-13-oxa-5-azahexacyclo[13.2.2.12,8.01,6.02,14.012,20]icosa-8(20),9,11-trien-11-ol
26. 2-[3-cyclopropylmethyl-11-hydroxy-15-methoxy-(14r)-13-oxa-3-azahexacyclo[13.2.2.12,8.01,6.06,14.07,12]icosa-7,9,11-trien-16-yl]-3,3-dimethyl-2-butanol
27. 6029-m
28. Buprenorphine (jan/inn)
29. Chembl560511
30. Unii-40d3scr4gz
31. Buvidal
32. Buprenorphine [inn:ban:jan]
33. Alks-5461 Component Buprenorphine
34. Einecs 257-950-6
35. Bema
36. [5alpha,7alpha(s)]-
37. Buprenorphine [mi]
38. Buprenorphine [inn]
39. Buprenorphine [jan]
40. Schembl15821
41. Buprenorphine [vandf]
42. (5alpha,6beta,14beta,18r)-17-(cyclopropylmethyl)-18-[(2s)-2-hydroxy-3,3-dimethylbutan-2-yl]-6-methoxy-18,19-dihydro-4,5-epoxy-6,14-ethenomorphinan-3-ol
43. 17-cyclopropylmethyl-4,5alpha-epoxy-7alpha-((s)-1-hydroxy-1,2,2-trimethylpropyl-6-methoxy-6,14-endo-ethanomorphinan-3-ol
44. 2-(n-cyclopropylmethyl-4,5alpha-epoxy-3-hydroxy-6-methoxy-6,14-endo-ethanomorphinan-7alpha-yl)-3,3-dimethyl-2-butanol
45. 21-(cyclopropyl-7alpha-((s)-1-hydroxy-1,2,2-trimethylpropyl-6,14-endo-ethano-6,7,8,14-tetrahydrooripavine
46. Buprenorphine [mart.]
47. Buprenorphine [who-dd]
48. Gtpl1670
49. Buprenorphine [ema Epar]
50. Dtxsid2022705
51. Buprenorphine [ep Impurity]
52. Buprenorphine [orange Book]
53. Sixmo (buprenorphine Hydrochloride)
54. Rbp-6000
55. Zinc1319780
56. Buprenorphine [ep Monograph]
57. Bdbm50026603
58. Db00921
59. [5alpha,7alpha(s)]-17-(cyclopropylmethyl)-alpha-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-alpha-methyl-6,14-ethenomorphinan-7-methanol
60. 21-cyclopropyl-7alpha-(2-hydroxy-3,3-dimethyl-2-butyl)-6,14-endo-ethano-6,7,8,14-tetrahydrooripavine
61. 6,14-ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)-.alpha.-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-.alpha.-methyl-, (.alpha.s,5.alpha.,7.alpha.)-
62. C08007
63. D07132
64. Q407721
65. (1s,2r,6s,14r,15r,16r)-3-(cyclopropylmethyl)-16-[(2s)-2-hydroxy-3,3-dimethylbutan-2-yl]-15-methoxy-13-oxa-3-azahexacyclo[13.2.2.1^{2,8}.0^{1,6}.0^{6,14}.0^{7,12}]icosa-7(12),8,10-trien-11-ol
66. (1s,2r,6s,14r,15r,16r)-3-(cyclopropylmethyl)-16-[(2s)-2-hydroxy-3,3-dimethylbutan-2-yl]-15-methoxy-13-oxa-3-azahexacyclo[13.2.2.1^{2,8}.0^{1,6}.0^{6,14}.0^{7,12}]icosa-7,9,11-trien-11-ol
67. (2s)-2-[(5r,6r,7r,14s)-9alpha-cyclopropylmethyl-3-hydroxy-6-methoxy-4,5-epoxy-6,14-ethanomorphinan-7-yl]-3,3-dimethylbutan-2-ol
68. 6,14-ethenomorphinan-3-ol, 17-(cyclopropylmethyl)-4,5-epoxy-18,19-dihydro-7-[(1s)-1-hydroxy-1,2,2-trimethylpropyl]-6-methoxy-, (5alpha,7alpha)-
69. 6,14-ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)-alpha-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-alpha-methyl-,
70. 6,14-ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)-alpha-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-alpha-methyl-, (5-alpha,7-alpha-(s))-
71. 6,14-ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)-alpha-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-alpha-methyl-, (alphas,5alpha,7alpha)- (9ci)
| Molecular Weight | 467.6 g/mol |
|---|---|
| Molecular Formula | C29H41NO4 |
| XLogP3 | 5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Exact Mass | 467.30355879 g/mol |
| Monoisotopic Mass | 467.30355879 g/mol |
| Topological Polar Surface Area | 62.2 Ų |
| Heavy Atom Count | 34 |
| Formal Charge | 0 |
| Complexity | 869 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 7 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Buprenex |
| PubMed Health | Buprenorphine (Injection) |
| Drug Classes | Analgesic, Anesthetic Adjunct |
| Drug Label | Buprenex (buprenorphine hydrochloride) is a narcotic under the Controlled Substances Act due to its chemical derivation from thebaine. Chemically, it is 17-(cyclopropylmethyl)--(1,1-dimethylethyl)-4, 5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy--met... |
| Active Ingredient | Buprenorphine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | eq 0.3mg base/ml; 0.3mg |
| Market Status | Prescription |
| Company | Reckitt Benckiser; Norwich-eaton Pharma |
| 2 of 4 | |
|---|---|
| Drug Name | Butrans |
| PubMed Health | Buprenorphine (Absorbed through the skin) |
| Drug Classes | Analgesic |
| Drug Label | Butrans is a transdermal system providing systemic delivery of buprenorphine, a mu opioid partial agonist analgesic, continuously for 7 days. The chemical name of buprenorphine is 6,14-ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)- -(1,1-dimet... |
| Active Ingredient | Buprenorphine |
| Dosage Form | Film, extended release |
| Route | Transdermal |
| Strength | 10mcg/hr; 20mcg/hr; 15mcg/hr; 5mcg/hr; 7.5mcg/hr |
| Market Status | Prescription |
| Company | Purdue Pharma |
| 3 of 4 | |
|---|---|
| Drug Name | Buprenex |
| PubMed Health | Buprenorphine (Injection) |
| Drug Classes | Analgesic, Anesthetic Adjunct |
| Drug Label | Buprenex (buprenorphine hydrochloride) is a narcotic under the Controlled Substances Act due to its chemical derivation from thebaine. Chemically, it is 17-(cyclopropylmethyl)--(1,1-dimethylethyl)-4, 5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy--met... |
| Active Ingredient | Buprenorphine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | eq 0.3mg base/ml; 0.3mg |
| Market Status | Prescription |
| Company | Reckitt Benckiser; Norwich-eaton Pharma |
| 4 of 4 | |
|---|---|
| Drug Name | Butrans |
| PubMed Health | Buprenorphine (Absorbed through the skin) |
| Drug Classes | Analgesic |
| Drug Label | Butrans is a transdermal system providing systemic delivery of buprenorphine, a mu opioid partial agonist analgesic, continuously for 7 days. The chemical name of buprenorphine is 6,14-ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)- -(1,1-dimet... |
| Active Ingredient | Buprenorphine |
| Dosage Form | Film, extended release |
| Route | Transdermal |
| Strength | 10mcg/hr; 20mcg/hr; 15mcg/hr; 5mcg/hr; 7.5mcg/hr |
| Market Status | Prescription |
| Company | Purdue Pharma |
Buprenorphine is indicated for the management of pain severe enough to require an opioid analgesic and for which alternative treatments are inadequate. Buprenorphine is also used in combination with [naloxone] in a fixed-dose combination product for the treatment of moderate to severe opioid use disorder.
FDA Label
Treatment of opioid dependence within a framework of medical, social and psychological treatment. Treatment is intended for use in adults and adolescents aged 16 years or over.
Buprenorphine interacts predominately with the opioid mu-receptor. These mu-binding sites are discretely distributed in the human brain, spinal cord, and other tissues. In clinical settings, buprenorphine exerts its principal pharmacologic effects on the central nervous system. Its primary actions of therapeutic value are analgesia and sedation. In addition to analgesia, alterations in mood, euphoria and dysphoria, and drowsiness commonly occur. Buprenorphine depresses the respiratory centers, depresses the cough reflex, and constricts the pupils. **Dependence** Buprenorphine is a partial agonist at the mu-opioid receptor and chronic administration produces physical dependence of the opioid type, characterized by withdrawal signs and symptoms upon abrupt discontinuation or rapid taper. The withdrawal syndrome is typically milder than seen with full agonists and may be delayed in onset. Buprenorphine can be abused in a manner similar to other opioids. This should be considered when prescribing or dispensing buprenorphine in situations when the clinician is concerned about an increased risk of misuse, abuse, or diversion. **Withdrawal** Abrupt discontinuation of treatment is not recommended as it may result in an opioid withdrawal syndrome that may be delayed in onset. Signs and symptoms may include body aches, diarrhea, gooseflesh, loss of appetite, nausea, nervousness or restlessness, anxiety, runny nose, sneezing, tremors or shivering, stomach cramps, tachycardia, trouble with sleeping, unusual increase in sweating, palpitations, unexplained fever, weakness and yawning. **Risk of Respiratory and Central Nervous System (CNS) Depression and Overdose** Buprenorphine has been associated with life-threatening respiratory depression and death. Many, but not all, post-marketing reports regarding coma and death involved misuse by self-injection or were associated with the concomitant use of buprenorphine and benzodiazepines or other CNS depressant, including alcohol. Use buprenorphine and naloxone sublingual tablets with caution in patients with compromised respiratory function (e.g., chronic obstructive pulmonary disease, cor pulmonale, decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression). **Risk of Overdose in Opioid Nave Patients** There have been reported deaths of opioid-nave individuals who received a 2 mg dose of buprenorphine as a sublingual tablet for analgesia. Buprenorphine and naloxone sublingual tablets are not appropriate as an analgesic in opioid-nave patients. **Precipitation of Opioid Withdrawal Signs and Symptoms** If buprenorphine is started in opioid-dependent individuals, it will displace the other opioids and cause a phenomenon known as "precipitated withdrawal" which is characterized by a rapid and intense onset of withdrawal symptoms. Individuals must therefore be in a state of mild to moderate withdrawal before starting therapy with buprenorphine. Because it contains naloxone, buprenorphine and naloxone sublingual tablets are also highly likely to produce marked and intense withdrawal signs and symptoms if misused parenterally by individuals dependent on full opioid agonists such as heroin, morphine, or methadone. **Gastrointestinal Effects** Buprenorphine and other morphine-like opioids have been shown to decrease bowel motility and cause constipation. Buprenorphine may obscure the diagnosis or clinical course of patients with acute abdominal conditions and should be administered with caution to patients with dysfunction of the biliary tract. **Effects on the Endocrine System** Opioids inhibit the secretion of adrenocorticotropic hormone (ACTH), cortisol, and luteinizing hormone (LH) in humans. They also stimulate prolactin, growth hormone (GH) secretion, and pancreatic secretion of insulin and glucagon. Chronic use of opioids may influence the hypothalamic-pituitary-gonadal axis, leading to androgen deficiency that may manifest as low libido, impotence, erectile dysfunction, amenorrhea, or infertility. The causal role of opioids in the clinical syndrome of hypogonadism is unknown because the various medical, physical, lifestyle, and psychological stressors that may influence gonadal hormone levels have not been adequately controlled for in studies conducted to date. Patients presenting with symptoms of androgen deficiency should undergo laboratory evaluation. **Adrenal Insufficiency** Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use. Presentation of adrenal insufficiency may include non-specific symptoms and signs including nausea, vomiting, anorexia, fatigue, weakness, dizziness, and low blood pressure. If adrenal insufficiency is suspected, confirm the diagnosis with diagnostic testing as soon as possible. If adrenal insufficiency is diagnosed, treat with physiologic replacement doses of corticosteroids. Wean the patient off of the opioid to allow adrenal function to recover and continue corticosteroid treatment until adrenal function recovers. Other opioids may be tried as some cases reported use of a different opioid without recurrence of adrenal insufficiency. The information available does not identify any particular opioids as being more likely to be associated with adrenal insufficiency. **Use in Patients With Impaired Hepatic Function** Buprenorphine/naloxone products are not recommended in patients with severe hepatic impairment and may not be appropriate for patients with moderate hepatic impairment. The doses of buprenorphine and naloxone in this fixed-dose combination product cannot be individually titrated, and hepatic impairment results in a reduced clearance of naloxone to a much greater extent than buprenorphine. Therefore, patients with severe hepatic impairment will be exposed to substantially higher levels of naloxone than patients with normal hepatic function. This may result in an increased risk of precipitated withdrawal at the beginning of treatment (induction) and may interfere with buprenorphines efficacy throughout treatment. In patients with moderate hepatic impairment, the differential reduction of naloxone clearance compared to buprenorphine clearance is not as great as in subjects with severe hepatic impairment. However, buprenorphine/naloxone products are not recommended for initiation of (treatment induction) in patients with moderate hepatic impairment due to the increased risk of precipitated withdrawal. Buprenorphine/naloxone products may be used with caution for maintenance treatment in patients with moderate hepatic impairment who have initiated treatment on a buprenorphine product without naloxone. However, patients should be carefully monitored and consideration given to the possibility of naloxone interfering with buprenorphines efficacy. **Risk of Hepatitis, Hepatic Events** Cases of cytolytic hepatitis and hepatitis with jaundice have been observed in individuals receiving buprenorphine in clinical trials and through post-marketing adverse event reports. The spectrum of abnormalities ranges from transient asymptomatic elevations in hepatic transaminases to case reports of death, hepatic failure, hepatic necrosis, hepatorenal syndrome, and hepatic encephalopathy. In many cases, the presence of pre-existing liver enzyme abnormalities, infection with hepatitis B or hepatitis C virus, concomitant usage of other potentially hepatotoxic drugs, and ongoing injecting drug use may have played a causative or contributory role. In other cases, insufficient data were available to determine the etiology of the abnormality. Withdrawal of buprenorphine has resulted in amelioration of acute hepatitis in some cases; however, in other cases no dose reduction was necessary. The possibility exists that buprenorphine had a causative or contributory role in the development of the hepatic abnormality in some cases. Liver function tests, prior to initiation of treatment is recommended to establish a baseline. Periodic monitoring of liver function during treatment is also recommended. A biological and etiological evaluation is recommended when a hepatic event is suspected. Depending on the case, buprenorphine and naloxone sublingual tablets may need to be carefully discontinued to prevent withdrawal signs and symptoms and a return by the patient to illicit drug use, and strict monitoring of the patient should be initiated. **Orthostatic Hypotension** Like other opioids, buprenorphine and naloxone sublingual tablets may produce orthostatic hypotension in ambulatory patients. **Elevation of Cerebrospinal Fluid Pressure** Buprenorphine, like other opioids, may elevate cerebrospinal fluid pressure and should be used with caution in patients with head injury, intracranial lesions, and other circumstances when cerebrospinal pressure may be increased. Buprenorphine can produce miosis and changes in the level of consciousness that may interfere with patient evaluation. **Elevation of Intracholedochal Pressure** Buprenorphine has been shown to increase intracholedochal pressure, as do other opioids, and thus should be administered with caution to patients with dysfunction of the biliary tract.
Analgesics, Opioid
Compounds with activity like OPIATE ALKALOIDS, acting at OPIOID RECEPTORS. Properties include induction of ANALGESIA or NARCOSIS. (See all compounds classified as Analgesics, Opioid.)
Narcotics
Agents that induce NARCOSIS. Narcotics include agents that cause somnolence or induced sleep (STUPOR); natural or synthetic derivatives of OPIUM or MORPHINE or any substance that has such effects. They are potent inducers of ANALGESIA and OPIOID-RELATED DISORDERS. (See all compounds classified as Narcotics.)
Narcotic Antagonists
Agents inhibiting the effect of narcotics on the central nervous system. (See all compounds classified as Narcotic Antagonists.)
N07BC01
N07BC01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
N - Nervous system
N02 - Analgesics
N02A - Opioids
N02AE - Oripavine derivatives
N02AE01 - Buprenorphine
N - Nervous system
N07 - Other nervous system drugs
N07B - Drugs used in addictive disorders
N07BC - Drugs used in opioid dependence
N07BC01 - Buprenorphine
Absorption
Bioavailablity of buprenorphine/naloxone is very high following intravenous or subcutaneous administration, lower by the sublingual or buccal route, and very low when administered by the oral route. It is therefore provided as a sublingual tablet that is absorbed from the oral mucosa directly into systemic circulation. Clinical pharmacokinetic studies found that there was wide inter-patient variability in the sublingual absorption of buprenorphine and naloxone, but within subjects the variability was low. Both Cmax and AUC of buprenorphine increased in a linear fashion with the increase in dose (in the range of 4 to 16 mg), although the increase was not directly dose-proportional. Buprenorphine combination with naloxone (2mg/0.5mg) provided in sublingual tablets demonstrated a Cmax of 0.780 ng/mL with a Tmax of 1.50 hr and AUC of 7.651 ng.hr/mL. Coadministration with naloxone does not effect the pharmacokinetics of buprenorphine.
Route of Elimination
Buprenorphine, like morphine and other phenolic opioid analgesics, is metabolized by the liver and its clearance is related to hepatic blood flow. It is primarily eliminated via feces (as free forms of buprenorphine and norbuprenorphine) while 10 - 30% of the dose is excreted in urine (as conjugated forms of buprenorphine and norbuprenorphine). The overall mean elimination half-life of buprenorphine in plasma ranges from 31 to 42 hours, although the levels are very low 10 hours after dosing (majority of AUC of buprenorphine is captured within 10 hours), indicating that the effective half-life may be shorter.
Volume of Distribution
Buprenorphine is highly lipophilic, and therefore extensively distributed, with rapid penetration through the blood-brain barrier. The estimated volume of distribution is 188 - 335 L when given intravenously. It is able to cross into the placenta and breast milk.
Clearance
Clearance may be higher in children than in adults. Plasma clearance rate, IV administration, anaesthetized patients = 901.2 39.7 mL/min; Plasma clearance rate, IV administration, healthy subjects = 1042 - 1280 mL/min.
Buprenorphine is metabolized to norbuprenorphine via Cytochrome P450 3A4/3A5-mediated N-dealkylation. Buprenorphine and norbuprenorphine both also undergo glucuronidation to the inactive metabolites buprenorphine-3-glucuronide and norbuprenorphine-3-glucuronide, respectively. While norbuprenorphine has been found to bind to opioid receptors in-vitro, brain concentrations are very low which suggests that it does not contribute to the clinical effects of buprenorphine. [Naloxone] undergoes direct glucuronidation to naloxone-3-glucuronide as well as N-dealkylation, and reduction of the 6-oxo group.
Buprenorphine has known human metabolites that include Buprenorphine glucuronide and Norbuprenorphine.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
Buprenorphine demonstrates slow dissociation kinetics (~166 min), which contributes to its long duration of action and allows for once-daily or even every-second-day dosing. In clinical trial studies, the half-life of sublingually administered buprenorphine/naloxone 2mg/0.5mg was found to be 30.75 hours.
Buprenorphine is a partial agonist at the mu-opioid receptor and an antagonist at the kappa-opioid receptor. It demonstrates a high affinity for the mu-opioid receptor but has lower intrinsic activity compared to other full mu-opioid agonists such as [heroin], [oxycodone], or [methadone]. This means that buprenorphine preferentially binds the opioid receptor and displaces lower affinity opioids without activating the receptor to a comparable degree. Clinically, this results in a slow onset of action and a clinical phenomenon known as the "ceiling effect" where once a certain dose is reached buprenorphine's effects plateau. This effect can be beneficial, however, as dose-related side effects such as respiratory depression, sedation, and intoxication also plateau at around 32mg, resulting in a lower risk of overdose compared to [methadone] and other full agonist opioids. It also means that opioid-dependent patients do not experience sedation or euphoria at the same rate that they might experience with more potent opioids, improving quality of life for patients with severe pain and reducing the reinforcing effects of opioids which can lead to drug-seeking behaviours. Buprenorphine's high affinity, but low intrinsic activity for the mu-opioid receptor also means that if it is started in opioid-dependent individuals, it will displace the other opioids without creating an equal opioid effect and cause a phenomenon known as "precipitated withdrawal" which is characterized by a rapid and intense onset of withdrawal symptoms (i.e. anxiety, restlessness, gastrointestinal distress, diaphoresis, intense drug cravings, and tachycardia). Individuals must therefore be in a state of mild to moderate withdrawal before starting therapy with buprenorphine. Buprenorphine is commercially available as the brand name product Suboxone which is formulated in a 4:1 fixed-dose combination product along with [naloxone], a non-selective competitive opioid receptor antagonist. Combination of an opioid agonist with an opioid antagonist may seem counterintuitive, however this combination with naloxone is intended to reduce the abuse potential of Suboxone, as naloxone is poorly absorbed by the oral route (and has no effect when taken orally), but would reverse the opioid agonist effects of buprenorphine if injected intravenously.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
84
PharmaCompass offers a list of Buprenorphine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Buprenorphine manufacturer or Buprenorphine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Buprenorphine manufacturer or Buprenorphine supplier.
A Buprenorphine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Buprenorphine, including repackagers and relabelers. The FDA regulates Buprenorphine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Buprenorphine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Buprenorphine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Buprenorphine supplier is an individual or a company that provides Buprenorphine active pharmaceutical ingredient (API) or Buprenorphine finished formulations upon request. The Buprenorphine suppliers may include Buprenorphine API manufacturers, exporters, distributors and traders.
click here to find a list of Buprenorphine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Buprenorphine DMF (Drug Master File) is a document detailing the whole manufacturing process of Buprenorphine active pharmaceutical ingredient (API) in detail. Different forms of Buprenorphine DMFs exist exist since differing nations have different regulations, such as Buprenorphine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Buprenorphine DMF submitted to regulatory agencies in the US is known as a USDMF. Buprenorphine USDMF includes data on Buprenorphine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Buprenorphine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Buprenorphine suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Buprenorphine Drug Master File in Japan (Buprenorphine JDMF) empowers Buprenorphine API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Buprenorphine JDMF during the approval evaluation for pharmaceutical products. At the time of Buprenorphine JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Buprenorphine suppliers with JDMF on PharmaCompass.
A Buprenorphine CEP of the European Pharmacopoeia monograph is often referred to as a Buprenorphine Certificate of Suitability (COS). The purpose of a Buprenorphine CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Buprenorphine EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Buprenorphine to their clients by showing that a Buprenorphine CEP has been issued for it. The manufacturer submits a Buprenorphine CEP (COS) as part of the market authorization procedure, and it takes on the role of a Buprenorphine CEP holder for the record. Additionally, the data presented in the Buprenorphine CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Buprenorphine DMF.
A Buprenorphine CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Buprenorphine CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Buprenorphine suppliers with CEP (COS) on PharmaCompass.
A Buprenorphine written confirmation (Buprenorphine WC) is an official document issued by a regulatory agency to a Buprenorphine manufacturer, verifying that the manufacturing facility of a Buprenorphine active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Buprenorphine APIs or Buprenorphine finished pharmaceutical products to another nation, regulatory agencies frequently require a Buprenorphine WC (written confirmation) as part of the regulatory process.
click here to find a list of Buprenorphine suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Buprenorphine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Buprenorphine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Buprenorphine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Buprenorphine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Buprenorphine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Buprenorphine suppliers with NDC on PharmaCompass.
Buprenorphine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Buprenorphine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Buprenorphine GMP manufacturer or Buprenorphine GMP API supplier for your needs.
A Buprenorphine CoA (Certificate of Analysis) is a formal document that attests to Buprenorphine's compliance with Buprenorphine specifications and serves as a tool for batch-level quality control.
Buprenorphine CoA mostly includes findings from lab analyses of a specific batch. For each Buprenorphine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Buprenorphine may be tested according to a variety of international standards, such as European Pharmacopoeia (Buprenorphine EP), Buprenorphine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Buprenorphine USP).

