
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
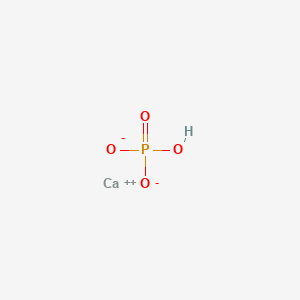

1. Anhydrous Dibasic Calcium Phosphate
2. Calcium Phosphate (cahpo4)
3. Calcium Phosphate, Dibasic, Anhydrous
4. Dibasic Calcium Phosphate, Anhydrous
5. Monetite
1. 7757-93-9
2. Calcium Phosphate Dibasic
3. Dicalcium Phosphate
4. Calcium Phosphate, Dibasic
5. Dibasic Calcium Phosphate
6. Phosphoric Acid, Calcium Salt (1:1)
7. Secondary Calcium Phosphate
8. Calcium Phosphate (1:1)
9. Dicalcium Orthophosphate
10. Calcium Hydrogen Orthophosphate
11. Monocalcium Acid Phosphate
12. Dicalcium Phosphate Anhydrous
13. Calcium Monohydrogen Phosphate
14. Calciumphosphate,dibasic
15. Calcium Phosphate Dibasic Anhydrous
16. Anhydrous Dibasic Calcium Phosphate
17. Calcium Phosphate,dibasic
18. Mfcd00010909
19. Calcium Phosphate (dibasic)
20. Chebi:32596
21. Ins-341(ii)
22. L11k75p92j
23. Calcium Phosphate, Dibasic, Anhydrous
24. E-341(ii)
25. Biofos
26. Fujicalin S
27. Dicafos An
28. 7557-93-9
29. Anhydrous Emcompress
30. Ipifosc 20
31. A-tab
32. D-glucopyranosiduronicacid, 1-(2,4-dichlorophenyl)ethyl
33. Calcium Acid Phosphate
34. Calcium Dibasic Phosphate
35. Calcium Secondary Phosphate
36. Calcium Phosphate (cahpo4)
37. Monohydrogen Calcium Phosphate
38. Ccris 1337
39. Hsdb 992
40. Cahpo4
41. Calcium Phosphate, Dibasic [jan]
42. Einecs 231-826-1
43. Calcium Hydrogenorthophosphate
44. Calcium Phosphate, Dibasic, Dental Grade
45. Unii-l11k75p92j
46. Calstar (tn)
47. Calcium Monohydrogen Phosphate Anhydrous
48. Calcii Hydrogenophosphas
49. Calcium;hydrogen Phosphate
50. Ec 231-826-1
51. Calcium Monohydrogenphosphate
52. Dicalcium Phosphate, Anhydrous
53. Calcium (as Phosphate,dibasic)
54. Ins No.341(ii)
55. Chembl2107567
56. Dtxsid20872529
57. Dicalcium Phosphate [inci]
58. Phosphoric Acid Calcium Salt
59. Calcium Hydrogen Monophosphate
60. Calcium Hydrogen Phosphate, Anhydrous
61. Akos015965054
62. Calcium Hydrogenorthophosphate Anhydrous
63. Calcium Phosphate, Dibasic [mi]
64. Calcium Phosphate,dibasic [vandf]
65. Calcium Hydrogenorthophosphate, With A Fluorine Content Of Less Than 0,005 Per Cent By Weight On The Dry Anhydrous Product
66. Calcium Phosphate, Dibasic [hsdb]
67. Dibasic Calcium Phosphate Anhydrous
68. Calcium Hydrogen Phosphate Anhydrous
69. Calcium Phosphate Dibasic [who-dd]
70. Calcium Phosphate, Dibasic (99.95%-ca)
71. Phosphorus (as Dicalcium Phosphate)
72. Calcium Phosphate (1:1) [who-ip]
73. Calcium Phosphate Dibasic, 98.0-105.0%
74. Calcium Phosphate, Dibasic Anhydrous
75. Dibasic Calcium Phosphate, Anhydrous (jan)
76. Calcium Phosphate Dibasic, Puriss., 98.0%
77. Ft-0623397
78. Calcium (calcium Phosphate (dibasic))
79. Calcium Hydrogen Phosphate (anhydrous)
80. Calcii Hydrogenophosphas [who-ip Latin]
81. Calcium (as Phosphate,dibasic) [vandf]
82. D03302
83. Anhydrous Dibasic Calcium Phosphate [jan]
84. Calcium Hydrogen Phosphate [ep Monograph]
85. Calcium Phosphate Dibasic Anhydrous, Usp Fcc Pe
86. Calcium Phosphate Dibasic, Usp, 98.0-103.0%
87. Calcium Phosphate, Dibasic, Anhydrous [ii]
88. Q414619
89. Phosphorus (as Dicalcium Phosphate) [vandf]
90. Calcium Hydrogen Phosphate, Anhydrous [who-ip]
91. Anhydrous Dibasic Calcium Phosphate [usp Monograph]
92. Dibasic Calcium Phosphate, Meets Usp Testing Specifications
93. Phosphorus (as Calcium Phosphate,dibasic) [vandf]
94. Calcium Phosphate Dibasic, Puriss. P.a., Anhydrous, >98.5% (kt)
| Molecular Weight | 136.06 g/mol |
|---|---|
| Molecular Formula | CaHO4P |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Exact Mass | 135.9238364 g/mol |
| Monoisotopic Mass | 135.9238364 g/mol |
| Topological Polar Surface Area | 83.4 Ų |
| Heavy Atom Count | 6 |
| Formal Charge | 0 |
| Complexity | 46.5 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
USES--EXCELLENT SOURCE OF CALCIUM & PHOSPHORUS DURING PREGNANCY, LACTATION, OR MILD TO MODERATE HYPOCALCEMIA CHARACTERIZED BY LOW DEGREE OF TETANY. ... IF TETANY IS SEVERE, IV CALCIUM MEDICATION IS ADMIN. ... DOSE--1 TO 5 G/DAY IN 2 OR MORE DIVIDED DOSES.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 770
IT IS NOT MARKETED IN SINGLE ENTITY FORM, SO THAT DOSE OF OTHER INGREDIENTS MAY HAVE TO BE CONSIDERED IN ARRIVING @ SUITABLE SCHEDULE. CALCIUM IS BETTER ABSORBED IF TAKEN IN SMALL DOSES @ FREQUENT INTERVALS.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 770
MEDICATION (VET): FOR PREVENTION & TREATMENT OF RICKETS ALONG WITH VITAMIN D THERAPY, & AS NUTRIENT SOURCE OF CALCIUM & PHOSPHORUS IN FEEDSTUFFS. ... OPTIMUM CALCIUM:PHOSPHORUS RATIO IN DIET MUST ALSO BE MAINTAINED.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 71
DIBASIC CALCIUM PHOSPHATE, NATIONAL FORMULARY ... /IS AMONG CMPD/ CLASSIFIED AS NONSYSTEMIC GASTRIC ANTACIDS, BUT THEIR EFFICACY IS LOW & THEY ARE SELDOM EMPLOYED.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 967
Medication (VET): Has been used as a dietary supplement, and as an antacid.
Budavari, S. (ed.). The Merck Index - Encyclopedia of Chemicals, Drugs and Biologicals. Rahway, NJ: Merck and Co., Inc., 1989., p. 256
BECAUSE OF PHOSPHATE CONTENT, IT IS CONTRAINDICATED IN /PERSONS WITH/ HYPOPARATHYROIDISM.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 770
... ITS ELEMENTS ARE SLOWLY ABSORBED FROM SMALL INTESTINE.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 71
Related Excipient Companies

Excipients by Applications
Market Place

ABOUT THIS PAGE
12
PharmaCompass offers a list of Calcium Acid Phosphate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Calcium Acid Phosphate manufacturer or Calcium Acid Phosphate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Calcium Acid Phosphate manufacturer or Calcium Acid Phosphate supplier.
A Biofos manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Biofos, including repackagers and relabelers. The FDA regulates Biofos manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Biofos API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Biofos manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Biofos supplier is an individual or a company that provides Biofos active pharmaceutical ingredient (API) or Biofos finished formulations upon request. The Biofos suppliers may include Biofos API manufacturers, exporters, distributors and traders.
click here to find a list of Biofos suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Biofos DMF (Drug Master File) is a document detailing the whole manufacturing process of Biofos active pharmaceutical ingredient (API) in detail. Different forms of Biofos DMFs exist exist since differing nations have different regulations, such as Biofos USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Biofos DMF submitted to regulatory agencies in the US is known as a USDMF. Biofos USDMF includes data on Biofos's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Biofos USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Biofos suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Biofos as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Biofos API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Biofos as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Biofos and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Biofos NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Biofos suppliers with NDC on PharmaCompass.
Biofos Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Biofos GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Biofos GMP manufacturer or Biofos GMP API supplier for your needs.
A Biofos CoA (Certificate of Analysis) is a formal document that attests to Biofos's compliance with Biofos specifications and serves as a tool for batch-level quality control.
Biofos CoA mostly includes findings from lab analyses of a specific batch. For each Biofos CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Biofos may be tested according to a variety of international standards, such as European Pharmacopoeia (Biofos EP), Biofos JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Biofos USP).

