
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
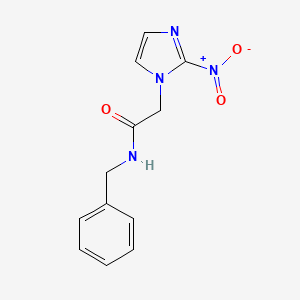

1. 2-nitroimidazole Benznidazole
2. Benzonidazole
3. Radanil
4. Ro 7-1051
1. 22994-85-0
2. Radanil
3. Benznidazol
4. Benzonidazole
5. Benzonidazol
6. Benznidazolum
7. Rochagan
8. Ro 07-1051
9. N-benzyl-2-(2-nitroimidazol-1-yl)acetamide
10. 1h-imidazole-1-acetamide, 2-nitro-n-(phenylmethyl)-
11. N-benzyl-2-(2-nitro-1h-imidazol-1-yl)acetamide
12. N-benzyl-2-nitroimidazole-1-acetamide
13. N-benzyl-2-nitro-1h-imidazole-1-acetamide
14. N-benzyl-2-nitroimidazol-1-yl-acetamide
15. 2-nitro-n-(phenylmethyl)-1h-imidazole-1-acetamide
16. Ro 71051
17. Imidazole-1-acetamide, N-benzyl-2-nitro-
18. Yc42nrj1zd
19. Chembl110
20. Nsc-299972
21. Ro-7-1051
22. Mmv688773
23. Nsc299972
24. Ncgc00166238-01
25. Ro 07-1051;ro 71051
26. Ro-07-1051
27. Dsstox_cid_26570
28. Dsstox_rid_81729
29. Dsstox_gsid_46570
30. Benznidazole [inn]
31. Benznidazol [inn-spanish]
32. Benznidazolum [inn-latin]
33. Smr000857153
34. Nsc 299972
35. Cas-22994-85-0
36. Ccris 2200
37. Sr-01000841264
38. Unii-yc42nrj1zd
39. N-benzyl-2-nitro-1-imidazoleacetamide
40. Brn 0551486
41. Ragonil
42. Acetamide, N-benzyl-2-(nitroimidazol-1-yl)-
43. Benznidazole (tn)
44. Rochagan (tn)
45. Radanil (tn)
46. Benznidazole [mi]
47. Benznidazole (usan/inn)
48. Benznidazole [usan:inn]
49. Benznidazole [usan]
50. Schembl45081
51. Benznidazole [mart.]
52. Mls001332409
53. Mls001332410
54. Mls001360496
55. Benznidazole [who-dd]
56. Benznidazole [who-ip]
57. Dtxsid9046570
58. Schembl22493029
59. Zinc56949
60. Chebi:133833
61. Benznidazole [orange Book]
62. Hms2233g13
63. Hms3369c11
64. Hy-b1548
65. Tox21_112364
66. Bdbm50089916
67. Benznidazolum [who-ip Latin]
68. Mfcd00243089
69. S3741
70. Akos015916722
71. Akos024283499
72. Tox21_112364_1
73. Ccg-267054
74. Db11989
75. Ncgc00166238-02
76. As-68694
77. Cs-0013411
78. Ft-0662547
79. Benznidazol (ro 07-1051; Ro 71051)
80. C74184
81. D02489
82. N-benzyl-2-(2-nitro-imidazol-1-yl)-acetamide
83. N-benzyl-2-(2-nitro-1himidazol-1-yl)acetamide
84. A912716
85. N-benzyl-2-(2-nitro-1h-imidazol-5-yl)acetamide
86. N-benzyl-2-nitro-1h-imidazole-1-acetamide, 97%
87. Q425300
88. 2-(2-nitroimidazol-1-yl)-n-(phenylmethyl)acetamide
89. J-014932
90. Sr-01000841264-3
91. Sr-01000841264-4
92. 1h-imidazole-1-acetamide,2-nitro-n-(phenylmethyl)-
93. Brd-k56156805-001-05-4
94. 1h-imidazole-1-acetamide, 2-nitro-n-(phenylmethyl)- [
95. N-benzyl-2-(2-nitro-imidazol-1-yl)-acetamide (benznidazole)
96. Pyridinium,1-dodecyl-2-[(hydroxyimino)methyl]-,iodide(1:1)
| Molecular Weight | 260.25 g/mol |
|---|---|
| Molecular Formula | C12H12N4O3 |
| XLogP3 | 0.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 260.09094026 g/mol |
| Monoisotopic Mass | 260.09094026 g/mol |
| Topological Polar Surface Area | 92.7 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 325 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
For use in the treatment of Chagas disease in children 2-12 years of age.
Benznidazole is a trypanocidal agent which kills the causative organism in Chagas disease, *Trypanosoma cruzi*.
Mutagens
Chemical agents that increase the rate of genetic mutation by interfering with the function of nucleic acids. A clastogen is a specific mutagen that causes breaks in chromosomes. (See all compounds classified as Mutagens.)
Trypanocidal Agents
Agents destructive to the protozoal organisms belonging to the suborder TRYPANOSOMATINA. (See all compounds classified as Trypanocidal Agents.)
Immunosuppressive Agents
Agents that suppress immune function by one of several mechanisms of action. Classical cytotoxic immunosuppressants act by inhibiting DNA synthesis. Others may act through activation of T-CELLS or by inhibiting the activation of HELPER CELLS. While immunosuppression has been brought about in the past primarily to prevent rejection of transplanted organs, new applications involving mediation of the effects of INTERLEUKINS and other CYTOKINES are emerging. (See all compounds classified as Immunosuppressive Agents.)
P - Antiparasitic products, insecticides and repellents
P01 - Antiprotozoals
P01C - Agents against leishmaniasis and trypanosomiasis
P01CA - Nitroimidazole derivatives
P01CA02 - Benznidazole
Absorption
Benznidazole has a bioavailability of 91.7% and a Tmax of 2.93 h.
Route of Elimination
The metabolites of benznidazole appear to be primarily exreted in the urine.
Volume of Distribution
The apparent volume of distribution is 39.19 L.
Clearance
The apparent oral clearance is 2.04 L/h.
Benznidazole is metabolized by nitroreductases in *Trypanosoma cruzi* and by cytochrome P450 enzymes.
The half life of elimination is 13.27 h.
Benznidazole is thought to be reduced to various electrophilic metabolites by nitroreductases present in *Trypanosoma cruzi*. These metabolites likely bind to proteins, lipids, DNA, and RNA resulting in damage to these macromolecules. Benznidazole has been found to increase trypanosomal death through interferon- which is likely present in increased amounts due to inflammation caused by macromolecule damage. DNA in parasites affected by benznidazole has been found to undergo extensive unpacking with overexpression of DNA repair proteins supporting the idea of DNA damage contributing to the mechanism of the drug.
Related Excipient Companies

Excipients by Applications
ABOUT THIS PAGE
24
PharmaCompass offers a list of Benznidazole API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Benznidazole manufacturer or Benznidazole supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Benznidazole manufacturer or Benznidazole supplier.
A Benznidazole manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Benznidazole, including repackagers and relabelers. The FDA regulates Benznidazole manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Benznidazole API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Benznidazole manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Benznidazole supplier is an individual or a company that provides Benznidazole active pharmaceutical ingredient (API) or Benznidazole finished formulations upon request. The Benznidazole suppliers may include Benznidazole API manufacturers, exporters, distributors and traders.
click here to find a list of Benznidazole suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Benznidazole DMF (Drug Master File) is a document detailing the whole manufacturing process of Benznidazole active pharmaceutical ingredient (API) in detail. Different forms of Benznidazole DMFs exist exist since differing nations have different regulations, such as Benznidazole USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Benznidazole DMF submitted to regulatory agencies in the US is known as a USDMF. Benznidazole USDMF includes data on Benznidazole's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Benznidazole USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Benznidazole suppliers with USDMF on PharmaCompass.
Benznidazole Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Benznidazole GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Benznidazole GMP manufacturer or Benznidazole GMP API supplier for your needs.
A Benznidazole CoA (Certificate of Analysis) is a formal document that attests to Benznidazole's compliance with Benznidazole specifications and serves as a tool for batch-level quality control.
Benznidazole CoA mostly includes findings from lab analyses of a specific batch. For each Benznidazole CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Benznidazole may be tested according to a variety of international standards, such as European Pharmacopoeia (Benznidazole EP), Benznidazole JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Benznidazole USP).

