
Synopsis
Synopsis
0
VMF
0
Australia
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Amineurin
2. Amitrip
3. Amitriptylin Beta
4. Amitriptylin Desitin
5. Amitriptylin Neuraxpharm
6. Amitriptylin Rph
7. Amitriptylin-neuraxpharm
8. Amitriptyline
9. Amitrol
10. Anapsique
11. Apo Amitriptyline
12. Apo-amitriptyline
13. Damilen
14. Desitin, Amitriptylin
15. Domical
16. Elavil
17. Endep
18. Laroxyl
19. Lentizol
20. Novoprotect
21. Rph, Amitriptylin
22. Saroten
23. Sarotex
24. Syneudon
25. Triptafen
26. Tryptanol
27. Tryptine
28. Tryptizol
1. 549-18-8
2. Amitriptyline Hcl
3. Annoyltin
4. Tryptizol
5. Domical
6. Syneudon
7. Endep
8. Lentizol
9. Novoprotect
10. Etravil
11. Saroten
12. Elavil Hydrochloride
13. Amineurin
14. Amitril
15. Amitrip
16. Anapsique
17. Tryptine
18. Amavil
19. Amitid
20. Ami-anelun
21. Apo-amitriptyline
22. Daprimen
23. Elatrolet
24. Maxivalet
25. Miketorin
26. Mitaptyline
27. Rantoron
28. Sarotena
29. Trepiline
30. Triptizol
31. Trytomer
32. Vanatrip
33. Yamanouchi
34. Amilit
35. Amiplin
36. Amiprin
37. Amyline
38. Amyzol
39. Belpax
40. Elatrol
41. Enafon
42. Etrafon
43. Kyliran
44. Larozyl
45. Levate
46. Pinsanu
47. Pinsaun
48. Teperin
49. Triavil
50. Tridep
51. Tripta
52. Trynol
53. Damilen Hydrochloride
54. Amitriptyline Chloride
55. Saroten Retard
56. Tryptizol Retard
57. Limbitrol Ds
58. Tryptacap Hydrochloride
59. Adt-zimaia
60. Oasil-m
61. Redomex
62. Proheptadien Monohydrochloride
63. Amitriptyline (hydrochloride)
64. Sk-amitriptyline Chloride
65. Limbitrol
66. Sarotex
67. Nih 10794
68. Ccris 7092
69. Amitryptiline Hydrochloride
70. Nsc-104210
71. 26lud4jo9k
72. Annolytin
73. Mls000028437
74. Component Of Etrafon
75. Component Of Triavil
76. 5-(3-(dimethylamino)propylidene) Dibenzosuberane Hcl
77. 3-(10,11-dihydro-5h-dibenzo[a,d][7]annulen-5-ylidene)-n,n-dimethylpropan-1-amine Hydrochloride
78. 1-propanamine, 3-(10,11-dihydro-5h-dibenzo(a,d)cyclohepten-5-ylidene)-n,n-dimethyl-, Hydrochloride
79. Novotriptyn
80. Nornaln
81. Smr000058368
82. 549-18-8 (hcl)
83. Amitryptyline Hydrochloride
84. 5-(3-dimethylaminopropylidene)dibenzo(a,d)(1,4)cycloheptadiene Hydrochloride
85. Dsstox_cid_13187
86. Dsstox_rid_79055
87. Dsstox_gsid_33187
88. Amitriptyline Hydrochloride 100 Microg/ml In Acetonitrile
89. 1-propanamine, 3-(10,11-dihydro-5h-dibenzo[a,d]cyclohepten-5-ylidene)-n,n-dimethyl-, Hydrochloride
90. 3-(10,11-dihydro-5h-dibenzo[a,d][7]annulen-5-ylidene)-n,n-dimethyl-1-propanamine Hydrochloride
91. 3-(10,11-dihydro-5h-dibenzo[a,d]cyclohepten-5-ylidene)-n,n-dimethyl-1-propanamine Hydrochloride
92. Nsc169910
93. Wln: L C676 By&t&j Bu3n1&1 &gh
94. Amitriptyline Hydrochloride [jan]
95. Sr-01000003046
96. Ncgc00015095-09
97. Cas-549-18-8
98. Einecs 208-964-6
99. Mfcd00012537
100. Amitryptylline Hydrochloride
101. Unii-26lud4jo9k
102. Nsc 104210
103. Amilent
104. Sylvemid
105. Amicen
106. Euplit
107. Uxen
108. 5-(3-dimethylaminopropylidene)dibenzo[a,4]cycloheptadiene Hydrochloride
109. 5-[3-(dimethylamino)propylidene]dibenzo[a,4]cycloheptadiene Hydrochloride
110. 1-propanamine,11-dihydro-5h-dibenzo[a,d]cyclohepten-5-ylidene)-n,n-dimethyl-, Hydrochloride
111. 5-(3-dimethylaminopropylidene)dibenzo[a,d][1,4]cycloheptadiene Hydrochloride
112. 5h-dibenzo[a,.gamma.-propylamine, 10,11-dihydro-n,n-dimethyl-, Hydrochloride
113. 5h-dibenzo[a,.gamma.-propylamine, 10,11-dihydro-n,n-dimethyl-,hydrochloride
114. Prestwick_20
115. 3-(5,6-dihydrodibenzo[2,1-b:1',2'-e][7]annulen-11-ylidene)-n,n-dimethylpropan-1-amine;hydrochloride
116. Drg-0169
117. Elavil (tn)
118. Amitriptyline Hydrochloride [usp:jan]
119. Cpd000058368
120. Opera_id_618
121. Nortriptyline Impurity F
122. Amitriptylini Hydrochloridum
123. C20h23n.hcl
124. Schembl41079
125. 10,11-dihydro-n,n-dimethyl-5h-dibenzo(a,d)cycloheptene-delta(sup 5,gamma)-propylamine Hydrochloride
126. Mls001055393
127. Mls001074176
128. Mls002222152
129. Spectrum1500117
130. Regid_for_cid_11065
131. Chebi:2667
132. Chembl1200964
133. Dtxsid9033187
134. Hy-b0527a
135. Amitriptyline Hydrochloride ,(s)
136. Amitriptyline Hydrochloride, 99%
137. Hms1568o09
138. Hms1920c13
139. Pharmakon1600-01500117
140. Bcp31573
141. Amitriptyline-[13c3] Hydrochloride
142. Tox21_113122
143. Tox21_201278
144. Tox21_303612
145. Tox21_500112
146. Ccg-38919
147. Nsc104210
148. Nsc755869
149. S3183
150. Akos015994716
151. Tox21_113122_1
152. Ks-1291
153. Lp00112
154. Nc00497
155. Nsc-169910
156. Nsc-755869
157. Amitriptyline Hydrochloride (jp17/usp)
158. Amitriptyline Hydrochloride [mi]
159. Ncgc00015095-15
160. Ncgc00024433-03
161. Ncgc00024433-05
162. Ncgc00093609-01
163. Ncgc00093609-02
164. Ncgc00093609-03
165. Ncgc00093609-04
166. Ncgc00093609-05
167. Ncgc00257393-01
168. Ncgc00258830-01
169. Ncgc00260797-01
170. 5h-dibenzo(a,d)cycloheptene-delta(sup 5),gamma-propylamine, 10,11-dihydro-n,n-dimethyl-, Hydrochloride
171. 5h-dibenzo(a,d)cycloheptene-delta5,gamma-propylamine, 10,11-dihydro-n,n-dimethyl-, Hydrochloride
172. Amitriptyline Hydrochloride [hsdb]
173. Ba164159
174. N,n-dimethyl-3-(2-tricyclo[9.4.0.03,8]pentadeca-1(15),3,5,7,11,13-hexaenylidene)propan-1-amine;hydrochloride
175. Amitriptyline Hcl;nortriptyline Impurity F
176. Amitriptyline Hydrochloride [mart.]
177. Amitriptyline Hydrochloride [vandf]
178. Amitriptyline Hydrochloride [usp-rs]
179. Amitriptyline Hydrochloride [who-dd]
180. Amitriptyline Hydrochloride [who-ip]
181. A0908
182. Eu-0100112
183. Ft-0662108
184. Ft-0662109
185. Sw196337-3
186. A 8404
187. A16404
188. D00809
189. D81789
190. Amitriptyline Hydrochloride [ep Impurity]
191. A854879
192. Amitriptyline Hydrochloride [ep Monograph]
193. Amitriptyline Hydrochloride [usp Impurity]
194. Amitriptyline Hydrochloride [usp Monograph]
195. Amitriptylini Hydrochloridum [who-ip Latin]
196. Amitriptyline Hydrochloride, >=98% (tlc), Powder
197. Sr-01000003046-2
198. Sr-01000003046-6
199. W-105596
200. Etrafon-a Component Amitriptyline Hydrochloride
201. Limbitrol Component Amitriptyline Hydrochloride
202. Q27254112
203. Amitriptyline Hydrochloride Component Of Etrafon-a
204. Amitriptyline Hydrochloride Component Of Limbitrol
205. F0001-1443
206. Limbitrol Ds Component Amitriptyline Hydrochloride
207. Amitriptyline Hydrochloride Component Of Limbitrol Ds
208. 5-[3-(dimethylamino)propylidene]dibenzosuberane Hydrochloride
209. Amitriptyline Hydrochloride 1.0 Mg/ml In Methanol (as Free Base)
210. Amitriptyline Hydrochloride, Drug Standard, 1.0 Mg/ml In Methanol
211. Amitriptyline Hydrochloride, British Pharmacopoeia (bp) Reference Standard
212. Amitriptyline Hydrochloride, European Pharmacopoeia (ep) Reference Standard
213. Amitriptyline Hydrochloride, Pharmaceutical Secondary Standard; Certified Reference Material
214. Amitriptyline Hydrochloride, United States Pharmacopeia (usp) Reference Standard
215. 1-propanamine, 3-(10,11-dihydro-5h-dibenzo(a,d)cyclohepten-5-ylidene)-n,n-dimethyl-, Hydrochloride (1:1)
216. 10,11-dihydro-n,n-dimethyl-5h-dibenzo[a,d]cycloheptene-delta5,gamma-propylamine Hydrochloride
217. 3-(10,11-dihydro-5h-dibenzo[a,d]cyc Lohepten-5-ylidene)-n,n-dimethyl-1-propanamine Hydrochloride
218. 5h-dibenzo[a,d]cycloheptene-delta(sup5),gamma-propylamine, 10,11-dihydro-n,n-dimethyl-, Hydrochloride
219. 5h-dibenzo[a,d]cycloheptene-delta5,gamma-propylamine, 10,11-dihydro-n,n-dimethyl-, Hydrochloride (6ci,8ci)
220. Amitriptyline Hydrochloride Solution, 1.0 Mg/ml In Methanol, Ampule Of 1 Ml, Certified Reference Material
221. N,n-dimethyl-3-(2-tricyclo[9.4.0.03,8]pentadeca-1(15),3,5,7,11,13-hexaenylidene)propan-1-amine;hydron;chloride
| Molecular Weight | 313.9 g/mol |
|---|---|
| Molecular Formula | C20H24ClN |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 3 |
| Exact Mass | 313.1597275 g/mol |
| Monoisotopic Mass | 313.1597275 g/mol |
| Topological Polar Surface Area | 3.2 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 331 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Amitriptyline hydrochloride |
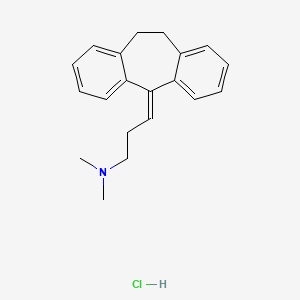
| Drug Label | DESCRIPTIONAmitriptyline HCl is 3-(10,11-dihydro-5H-dibenzo [a,d] cycloheptene-5-ylidene)-N,N-dimethyl-1-propanamine hydrochloride. Its empirical formula is C20H23NHCl, and its structural formula isAmitriptyline HCl, a dibenzocycloheptadiene deriv... |
| Active Ingredient | Amitriptyline hydrochloride |
| Dosage Form | Tablet |
| Route | oral; Oral |
| Strength | 75mg; 100mg; 25mg; 50mg; 10mg; 150mg |
| Market Status | Prescription |
| Company | Vintage Pharms; Mutual Pharm; Sun Pharm Inds; Sandoz; Mylan |
| 2 of 2 | |
|---|---|
| Drug Name | Amitriptyline hydrochloride |
| Drug Label | DESCRIPTIONAmitriptyline HCl is 3-(10,11-dihydro-5H-dibenzo [a,d] cycloheptene-5-ylidene)-N,N-dimethyl-1-propanamine hydrochloride. Its empirical formula is C20H23NHCl, and its structural formula isAmitriptyline HCl, a dibenzocycloheptadiene deriv... |
| Active Ingredient | Amitriptyline hydrochloride |
| Dosage Form | Tablet |
| Route | oral; Oral |
| Strength | 75mg; 100mg; 25mg; 50mg; 10mg; 150mg |
| Market Status | Prescription |
| Company | Vintage Pharms; Mutual Pharm; Sun Pharm Inds; Sandoz; Mylan |
Analgesics, Non-Narcotic
A subclass of analgesic agents that typically do not bind to OPIOID RECEPTORS and are not addictive. Many non-narcotic analgesics are offered as NONPRESCRIPTION DRUGS. (See all compounds classified as Analgesics, Non-Narcotic.)
Antidepressive Agents, Tricyclic
Substances that contain a fused three-ring moiety and are used in the treatment of depression. These drugs block the uptake of norepinephrine and serotonin into axon terminals and may block some subtypes of serotonin, adrenergic, and histamine receptors. However, the mechanism of their antidepressant effects is not clear because the therapeutic effects usually take weeks to develop and may reflect compensatory changes in the central nervous system. (See all compounds classified as Antidepressive Agents, Tricyclic.)
Adrenergic Uptake Inhibitors
Drugs that block the transport of adrenergic transmitters into axon terminals or into storage vesicles within terminals. The tricyclic antidepressants (ANTIDEPRESSIVE AGENTS, TRICYCLIC) and amphetamines are among the therapeutically important drugs that may act via inhibition of adrenergic transport. Many of these drugs also block transport of serotonin. (See all compounds classified as Adrenergic Uptake Inhibitors.)
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
29
PharmaCompass offers a list of Amitriptyline Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Amitriptyline Hydrochloride manufacturer or Amitriptyline Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Amitriptyline Hydrochloride manufacturer or Amitriptyline Hydrochloride supplier.
A Amyline manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Amyline, including repackagers and relabelers. The FDA regulates Amyline manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Amyline API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Amyline manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Amyline supplier is an individual or a company that provides Amyline active pharmaceutical ingredient (API) or Amyline finished formulations upon request. The Amyline suppliers may include Amyline API manufacturers, exporters, distributors and traders.
click here to find a list of Amyline suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Amyline DMF (Drug Master File) is a document detailing the whole manufacturing process of Amyline active pharmaceutical ingredient (API) in detail. Different forms of Amyline DMFs exist exist since differing nations have different regulations, such as Amyline USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Amyline DMF submitted to regulatory agencies in the US is known as a USDMF. Amyline USDMF includes data on Amyline's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Amyline USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Amyline suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Amyline Drug Master File in Japan (Amyline JDMF) empowers Amyline API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Amyline JDMF during the approval evaluation for pharmaceutical products. At the time of Amyline JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Amyline suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Amyline Drug Master File in Korea (Amyline KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Amyline. The MFDS reviews the Amyline KDMF as part of the drug registration process and uses the information provided in the Amyline KDMF to evaluate the safety and efficacy of the drug.
After submitting a Amyline KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Amyline API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Amyline suppliers with KDMF on PharmaCompass.
A Amyline CEP of the European Pharmacopoeia monograph is often referred to as a Amyline Certificate of Suitability (COS). The purpose of a Amyline CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Amyline EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Amyline to their clients by showing that a Amyline CEP has been issued for it. The manufacturer submits a Amyline CEP (COS) as part of the market authorization procedure, and it takes on the role of a Amyline CEP holder for the record. Additionally, the data presented in the Amyline CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Amyline DMF.
A Amyline CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Amyline CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Amyline suppliers with CEP (COS) on PharmaCompass.
A Amyline written confirmation (Amyline WC) is an official document issued by a regulatory agency to a Amyline manufacturer, verifying that the manufacturing facility of a Amyline active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Amyline APIs or Amyline finished pharmaceutical products to another nation, regulatory agencies frequently require a Amyline WC (written confirmation) as part of the regulatory process.
click here to find a list of Amyline suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Amyline as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Amyline API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Amyline as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Amyline and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Amyline NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Amyline suppliers with NDC on PharmaCompass.
Amyline Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Amyline GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Amyline GMP manufacturer or Amyline GMP API supplier for your needs.
A Amyline CoA (Certificate of Analysis) is a formal document that attests to Amyline's compliance with Amyline specifications and serves as a tool for batch-level quality control.
Amyline CoA mostly includes findings from lab analyses of a specific batch. For each Amyline CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Amyline may be tested according to a variety of international standards, such as European Pharmacopoeia (Amyline EP), Amyline JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Amyline USP).

